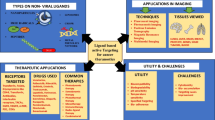
Abstract
Over recent years, theranostic nanosystems have emerged as promising tools in cancer therapy and diagnostics. Active-targeted theranostic nanosystems are based on develo** a nanocarrier modified with ligands or particular physical or chemical characteristics for specifically bind to surface markers of cancer cells and deliver therapeutic imaging and diagnostic agent or all of them to specific cancer cells and even organelles, providing personalized treatment for different tumors and patients. To now, different ligands were used to active targeting of nanocarriers. Antibodies, aptamers, transferrin, folic acid, and arginine-glycine-aspartic acid tripeptide are of the most studied ligands used for active targeting. Cancerous cells can internalize these systems by receptor-mediated endocytosis. However, therapeutic efficacy of single-targeted developed drug delivery systems is not yet satisfactory for clinical use because of the tumors complex microenvironment. To address mentioned challenge, dual and multi-targeted therapies have developed recently and demonstrate superior efficacy over single targeting in terms of cellular uptake, cell selectivity, and penetration into the tumor. Several dual-targeted nanocarriers introduced for cancer theranostic mainly belong to lipid-, polymer-, and carbon-based carriers. Herein, an overview of various developed dual-targeted nanomedicines used for cancer targeted theranostic will be reviewed; also challenges and outlook in designing dual-targeted nanomedicines will be discussed. Although these studies are only conducted in vitro and in vivo, in the upcoming years, it is highly likely that this field of treatment and diagnostics will be used in the clinical stage and help better treatment of rare diseases including cancer.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- BBB :
-
Blood-brain barrier
- BPA :
-
Borono phenylalanine
- BSH :
-
Sodium boro captate
- c(RGDyC):
-
Cyclic arginine-glycine-aspartic acid-tyrosine-cysteine
- CNT :
-
Carbon nanotubes
- DOX :
-
Doxorubicin
- EGFP :
-
Enhanced green fluorescent protein
- EGFR :
-
Epidermal growth factor receptor
- EPR :
-
Permeability and retention effect
- FA :
-
Folate
- FDA :
-
US Food and Drug Administration
- GBM :
-
Glioblastoma multiforme
- Glu-VC :
-
Glucose and vitamin C
- GO :
-
Graphene oxide
- HA :
-
Hyaluronic acid
- HAP :
-
Hydroxyapatite
- HepG2 :
-
Hepatocellular carcinoma cell line
- HIV :
-
Human immunodeficiency virus
- IL-4R :
-
Interleukin-4 receptor
- MGO :
-
Magnetic graphene oxide
- OA :
-
Oleanolic acid
- O-MWNT :
-
Oxidized multi-walled carbon nanotube
- PAMAM :
-
Poly(amidoamine)
- PCL :
-
Poly(3-caprolactone)
- PLA :
-
Polylactic acid
- PLGA :
-
Polylactic-co-glycolic acid
- PNAL :
-
Poly[(N isopropylacrylamide-r-acrylamide)-b-L-lactic acid]
- PTX :
-
Paclitaxel
- RAGE :
-
Receptors for advanced glycation end products
- RGD :
-
Arginine-glycine-aspartic acid
- Tf :
-
Transferrin
- TfR :
-
Tf receptor
- WGA :
-
Wheat germ agglutinin
References
Amoabediny, G., Haghiralsadat, F., Naderinezhad, S., Helder, M. N., Akhoundi Kharanaghi, E., Mohammadnejad Arough, J., & Zandieh-Doulabi, B. (2018a). Overview of preparation methods of polymeric and lipid-based (niosome, solid lipid, liposome) nanoparticles: A comprehensive review. International Journal of Polymeric Materials and Polymeric Biomaterials, 67(6), 383–400.
Amoabediny, G., Ochi, M. M., Rezayat, S. M., Akbarzadeh, A., & Ebrahimi, B. (2018b). Targeted nano-liposome co-entrap** anti-cancer drugs. Google patent US20160228362A1.
Anarjan, F. S. (2019). Active targeting drug delivery nanocarriers: Ligands. Nano-Structures & Nano-Objects, 19, 100370.
Belfiore, L., Saunders, D. N., Ranson, M., Thurecht, K. J., Storm, G., & Vine, K. L. (2018). Towards clinical translation of ligand-functionalized liposomes in targeted cancer therapy: Challenges and opportunities. Journal of Controlled Release, 277, 1–13.
Chen, D., Li, B., Cai, S., Wang, P., Peng, S., Sheng, Y., He, Y., Gu, Y., & Chen, H. (2016). Dual targeting luminescent gold nanoclusters for tumor imaging and deep tissue therapy. Biomaterials, 100, 1–16.
Danhier, F., Feron, O., & Préat, V. (2010). To exploit the tumor microenvironment: Passive and active tumor targeting of nanocarriers for anti-cancer drug delivery. Journal of Controlled Release, 148(2), 135–146.
Donahue, N. D., Acar, H., & Wilhelm, S. (2019). Concepts of nanoparticle cellular uptake, intracellular trafficking, and kinetics in nanomedicine. Advanced Drug Delivery Reviews, 143, 68–96.
Du, J., Li, Q., Chen, L., Wang, S., Zhang, L., Yu, S., Yang, Y., & Liu, X. (2020). In vitro cytotoxicity and antitumor activity of dual-targeting drug delivery system based on modified magnetic carbon by folate. Journal of Nanomaterials, 2020, 1.
Gao, H., **ong, Y., Zhang, S., Yang, Z., Cao, S., & Jiang, X. (2014). RGD and interleukin-13 peptide functionalized nanoparticles for enhanced glioblastoma cells and neovasculature dual targeting delivery and elevated tumor penetration. Molecular Pharmaceutics, 11(3), 1042–1052.
Haghiralsadat, F., Amoabediny, G., Naderinezhad, S., Zandieh-Doulabi, B., Forouzanfar, T., & Helder, M. N. (2018). Codelivery of doxorubicin and JIP1 siRNA with novel EphA2-targeted PEGylated cationic nanoliposomes to overcome osteosarcoma multidrug resistance. International Journal of Nanomedicine, 13, 3853.
He, H., Li, Y., Jia, X.-R., Du, J., Ying, X., Lu, W.-L., Lou, J.-N., & Wei, Y. (2011). PEGylated poly (amidoamine) dendrimer-based dual-targeting carrier for treating brain tumors. Biomaterials, 32(2), 478–487.
Huang, M., Pu, Y., Peng, Y., Fu, Q., Guo, L., Wu, Y., & Zheng, Y. (2020). Biotin and glucose dual-targeting, ligand-modified liposomes promote breast tumor-specific drug delivery. Bioorganic & Medicinal Chemistry Letters, 30(12), 127151.
Hussien, N. A., Işıklan, N., & Türk, M. (2018). Aptamer-functionalized magnetic graphene oxide nanocarrier for targeted drug delivery of paclitaxel. Materials Chemistry and Physics, 211, 479–488.
Kang, W., Svirskis, D., Saro**i, V., McGregor, A. L., Bevitt, J., & Wu, Z. (2017). Cyclic-RGDyC functionalized liposomes for dual-targeting of tumor vasculature and cancer cells in glioblastoma: An in vitro boron neutron capture therapy study. Oncotarget, 8(22), 36614.
Ke, X., Lin, W., Li, X., Wang, H., **ao, X., & Guo, Z. (2017). Synergistic dual-modified liposome improves targeting and therapeutic efficacy of bone metastasis from breast cancer. Drug Delivery, 24(1), 1680–1689.
Kluza, E., Jacobs, I., Hectors, S. J., Mayo, K. H., Griffioen, A. W., Strijkers, G. J., & Nicolay, K. (2012). Dual-targeting of αvβ3 and galectin-1 improves the specificity of paramagnetic/fluorescent liposomes to tumor endothelium in vivo. Journal of Controlled Release, 158(2), 207–214.
Kydd, J., Jadia, R., Velpurisiva, P., Gad, A., Paliwal, S., & Rai, P. (2017). Targeting strategies for the combination treatment of cancer using drug delivery systems. Pharmaceutics, 9(4), 46.
Lammers, T., Kiessling, F., Hennink, W. E., & Storm, G. (2012). Drug targeting to tumors: Principles, pitfalls and (pre-) clinical progress. Journal of Controlled Release, 161(2), 175–187.
Li, R., Wu, R. A., Zhao, L., Hu, Z., Guo, S., Pan, X., & Zou, H. (2011). Folate and iron difunctionalized multiwall carbon nanotubes as dual-targeted drug nanocarrier to cancer cells. Carbon, 49(5), 1797–1805.
Li, J., Liang, X., Zhang, J., Yin, Y., Zuo, T., Wang, Y., Yang, X., & Shen, Q. (2018a). Inhibiting pulmonary metastasis of breast cancer based on dual-targeting graphene oxide with high stability and drug loading capacity. Nanomedicine: Nanotechnology, Biology and Medicine, 14(4), 1237–1248.
Li, L., Wang, Q., Zhang, X., Luo, L., He, Y., Zhu, R., & Gao, D. (2018b). Dual-targeting liposomes for enhanced anticancer effect in somatostatin receptor II-positive tumor model. Nanomedicine, 13(17), 2155–2169.
Lin, M. M., Kang, Y. J., Sohn, Y., & Kim, D. K. (2015a). Dual targeting strategy of magnetic nanoparticle-loaded and RGD peptide-activated stimuli-sensitive polymeric micelles for delivery of paclitaxel. Journal of Nanoparticle Research, 17(6), 1–18.
Lin, M. M., Kang, Y. J., Sohn, Y., & Kim, D. K. (2015b). Dual targeting strategy of magnetic nanoparticle-loaded and RGD peptide-activated stimuli-sensitive polymeric micelles for delivery of paclitaxel. Journal of Nanoparticle Research, 17(6), 248.
Liu, G.-X., Fang, G.-Q., & Xu, W. (2014). Dual targeting biomimetic liposomes for paclitaxel/DNA combination cancer treatment. International Journal of Molecular Sciences, 15(9), 15287–15303.
Liu, Y., Chen, Q., Xu, M., Guan, G., Hu, W., Liang, Y., Zhao, X., Qiao, M., Chen, D., & Liu, H. (2015). Single peptide ligand-functionalized uniform hollow mesoporous silica nanoparticles achieving dual-targeting drug delivery to tumor cells and angiogenic blood vessel cells. International Journal of Nanomedicine, 10, 1855.
Lu, Y.-J., Lin, P.-Y., Huang, P.-H., Kuo, C.-Y., Shalumon, K., Chen, M.-Y., & Chen, J.-P. (2018). Magnetic graphene oxide for dual targeted delivery of doxorubicin and photothermal therapy. Nanomaterials, 8(4), 193.
Luo, Y., Yang, H., Zhou, Y.-F., & Hu, B. (2020). Dual and multi-targeted nanoparticles for site-specific brain drug delivery. Journal of Controlled Release, 317, 195–215.
Obraztsov, A., Obraztsova, E., Tyurnina, A., & Zolotukhin, A. (2007). Chemical vapor deposition of thin graphite films of nanometer thickness. Carbon, 45(10), 2017–2021.
Peng, Y., Zhao, Y., Chen, Y., Yang, Z., Zhang, L., **ao, W., Yang, J., Guo, L., & Wu, Y. (2018). Dual-targeting for brain-specific liposomes drug delivery system: Synthesis and preliminary evaluation. Bioorganic & Medicinal Chemistry, 26(16), 4677–4686.
Qin, L., Wang, C. Z., Fan, H. J., Zhang, C. J., Zhang, H. W., Lv, M. H., & Cui, S. D. (2014). A dual-targeting liposome conjugated with transferrin and arginine-glycine-aspartic acid peptide for glioma-targeting therapy. Oncology Letters, 8(5), 2000–2006.
Ren, J., Shen, S., Wang, D., **, Z., Guo, L., Pang, Z., Qian, Y., Sun, X., & Jiang, X. (2012). The targeted delivery of anticancer drugs to brain glioma by PEGylated oxidized multi-walled carbon nanotubes modified with angiopep-2. Biomaterials, 33(11), 3324–3333.
Rezayan, A. H., Mousavi, M., Kheirjou, S., Amoabediny, G., Ardestani, M. S., & Mohammadnejad, J. (2016). Monodisperse magnetite (Fe3O4) nanoparticles modified with water soluble polymers for the diagnosis of breast cancer by MRI method. Journal of Magnetism and Magnetic Materials, 420, 210–217.
Sun, Z., Yan, X., Liu, Y., Huang, L., Kong, C., Qu, X., Wang, M., Gao, R., & Qin, H. (2017). Application of dual targeting drug delivery system for the improvement of anti-glioma efficacy of doxorubicin. Oncotarget, 8(35), 58823.
van der Meel, R., Vehmeijer, L. J., Kok, R. J., Storm, G., & van Gaal, E. V. (2013). Ligand-targeted particulate nanomedicines undergoing clinical evaluation: Current status. Advanced Drug Delivery Reviews, 65(10), 1284–1298.
Wang, X., Lin, X., Dravid, V. P., Ketterson, J. B., & Chang, R. P. (1995). Carbon nanotubes synthesized in a hydrogen arc discharge. Applied Physics Letters, 66(18), 2430–2432.
Wang, S., Zhao, C., Liu, P., Wang, Z., Ding, J., & Zhou, W. (2018). Facile construction of dual-targeting delivery system by using lipid capped polymer nanoparticles for anti-glioma therapy. RSC Advances, 8(1), 444–453.
**n, H., Jiang, X., Gu, J., Sha, X., Chen, L., Law, K., Chen, Y., Wang, X., Jiang, Y., & Fang, X. (2011). Angiopep-conjugated poly (ethylene glycol)-co-poly (ε-caprolactone) nanoparticles as dual-targeting drug delivery system for brain glioma. Biomaterials, 32(18), 4293–4305.
**ong, X.-B., Uludağ, H., & Lavasanifar, A. (2010). Virus-mimetic polymeric micelles for targeted siRNA delivery. Biomaterials, 31(22), 5886–5893.
Xu, Q., Liu, Y., Su, S., Li, W., Chen, C., & Wu, Y. (2012). Anti-tumor activity of paclitaxel through dual-targeting carrier of cyclic RGD and transferrin conjugated hyperbranched copolymer nanoparticles. Biomaterials, 33(5), 1627–1639.
Yang, X., Chen, Y., Yuan, R., Chen, G., Blanco, E., Gao, J., & Shuai, X. (2008). Folate-encoded and Fe3O4-loaded polymeric micelles for dual targeting of cancer cells. Polymer, 49(16), 3477–3485.
Yang, X., Wang, Y., Huang, X., Ma, Y., Huang, Y., Yang, R., Duan, H., & Chen, Y. (2011). Multi-functionalized graphene oxide based anticancer drug-carrier with dual-targeting function and pH-sensitivity. Journal of Materials Chemistry, 21(10), 3448–3454.
Yuan, M., Qiu, Y., Zhang, L., Gao, H., & He, Q. (2016). Targeted delivery of transferrin and TAT co-modified liposomes encapsulating both paclitaxel and doxorubicin for melanoma. Drug Delivery, 23(4), 1171–1183.
Zhang, W., Yu, Z.-L., Wu, M., Ren, J.-G., **a, H.-F., Sa, G.-L., Zhu, J.-Y., Pang, D.-W., Zhao, Y.-F., & Chen, G. (2017). Magnetic and folate functionalization enables rapid isolation and enhanced tumor-targeting of cell-derived microvesicles. ACS Nano, 11(1), 277–290.
Zhang, Y., Cao, J., & Yuan, Z. (2020). Strategies and challenges to improve the performance of tumor-associated active targeting. Journal of Materials Chemistry B, 8(18), 3959–3971.
Zhao, Z., Zhao, Y., **e, C., Chen, C., Lin, D., Wang, S., Cui, X., Guo, Z., & Zhou, J. (2019). Dual-active targeting liposomes drug delivery system for bone metastatic breast cancer: Synthesis and biological evaluation. Chemistry and Physics of Lipids, 223, 104785.
Zhu, Y., Feijen, J., & Zhong, Z. (2018). Dual-targeted nanomedicines for enhanced tumor treatment. Nano Today, 18, 65–85.
Zong, T., Mei, L., Gao, H., Shi, K., Chen, J., Wang, Y., Zhang, Q., Yang, Y., & He, Q. (2014). Enhanced glioma targeting and penetration by dual-targeting liposome co-modified with T7 and TAT. Journal of Pharmaceutical Sciences, 103(12), 3891–3901.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Amoabediny, G. et al. (2021). Dual Targeting Drug Delivery for Cancer Theranostics. In: Saravanan, M., Barabadi, H. (eds) Cancer Nanotheranostics. Nanotechnology in the Life Sciences. Springer, Cham. https://doi.org/10.1007/978-3-030-74330-7_2
Download citation
DOI: https://doi.org/10.1007/978-3-030-74330-7_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-74329-1
Online ISBN: 978-3-030-74330-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)