Abstract
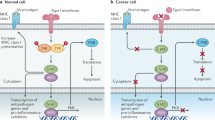
Oncolytic viruses are anticancer agents that selectively target and kill cancer cells by direct lysis, while at the same time stimulating a tumor antigen-specific adaptive immune response. These promising therapeutic agents target multiple cancers and have already proven to be an effective treatment option for solid malignancies. One such agent, T-Vec (Talimogene laherparepvec) has been licensed and is in routine clinical use for treatment of malignant melanoma.
Non-muscle invasive bladder cancer (NMIBC) is an ideal potential target for oncolytic immunotherapy as locally instilled live biological therapy using Bacille Calmette-Guerin (BCG) is already well established in the clinical setting. Coxsackievirus A21 (CVA21) is a novel intercellular adhesion molecule-1 (ICAM-1)-targeted immunotherapeutic virus. We have investigated CVA21-induced cytotoxicity in a panel of human bladder cancer cell lines, revealing a range of sensitivities largely correlating with expression of the viral receptor ICAM-1. CVA21 in combination with low doses of mitomycin-C enhanced CVA21 viral replication and oncolysis by increasing surface expression levels of ICAM-1. In addition to cell lines and an animal model a key component of our studies into oncolytic immunotherapy for bladder cancer was the use of a bladder tumor precision slice preclinical model system which represents tumor architecture, heterogeneity, and the complexity of a tumor in vitro. Results seen in cell lines were reflected in the tumor slice model whereby levels of virus protein expression and induction of apoptosis were enhanced with prior exposure to mitomycin-C. In this chapter we demonstrate the utility of the precision cut tumor slice model as a unique organotypic model to test oncolytic viruses. We will describe how to prepare and slice the tumor using a vibrating microtome together with the optimum culture and conditions for treatment.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Cancer Research UK (2016) Bladder cancer statistics. http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/bladder-cancer
Aldousari S, Kassouf W (2010) Update on the management of non-muscle invasive bladder cancer. Can Urol Assoc J 4:56–64
Fuge O, Vasdev N, Allchorne P, Green JSA (2015) Immunotherapy for bladder cancer. Res Rep Urol 7:65–79
Sanofi Pasteur (2016) Sanofi Pasteur statement on discontinuation of BCG. http://www.sanofipasteur.ca/node/50701
Davies B (2016) Sanofi shuts down bladder cancer drug production: inevitable drug shortage to harm patients. https://www.forbes.com/sites/benjamindavies/2016/11/17/sanofi-shuts-down-bladder-cancer-drug-production-inevitable-drug-shortage-to-harm-patients/#27d4a3abc132
Russell SJ, Peng KW, Bell JC (2012) Oncolytic virotherapy. Nat Biotechnol 30:658–670
Patel MR, Kratzke RA (2013) Oncolytic virus therapy for cancer: the first wave of translational clinical trials. Transl Res 161:355–364
Shafren DR, Au GG, Nguyen T et al (2004) Systemic therapy of malignant human melanoma tumours by a common cold-producing enterovirus, coxsackievirus A21. Clin Cancer Res 10:53–60
Au GG, Lindberg AM, Barry RD et al (2005) Oncolysis of vascular malignant human melanoma tumours by Coxsackievirus A21. Int J Oncol 26:1471–1476
Berry LJ, Au GG, Barry RD et al (2008) Potent oncolytic activity of human enteroviruses against human prostate cancer. Prostate 68:577–587
Skelding KA, Barry RD, Shafren DR (2009) Systemic targeting of metastatic human breast tumour xenografts by Coxsackievirus A21. Breast Cancer Res Treat 113:21–30
Au GG, Lincz LF, Enno A et al (2007) Oncolytic coxsackievirus A21 as a novel therapy for multiple myeloma. Br J Haematol 137:133–141
Andtbacka R, Curti B, Hallmeyer S et al (2015) Abstract CT214: phase II CALM study: changes in the tumour microenvironment induced by the immunotherapeutic agent coxsackievirus A21 delivered intratumorally in patients with advanced melanoma. Cancer Res 75:CT214
Annels NE, Arif M, Simpson GR et al (2018) Oncolytic immunotherapy for bladder cancer using Coxsackie A21 virus. Mol Ther Oncolytics 14(9):1–12
Hood CJ, Parham DM (1998) A simple method of tumour culture. Pathol Res Pract 194(3):177–181
Davies EJ, Dong M, Gutekunst M et al (2015) Capturing complex tumour biology in vitro: histological and molecular characterisation of precision cut slices. Sci Rep 9(5):17187
Gerlach MM, Merz F, Wichmann G et al (2014) Slice cultures from head and neck squamous cell carcinoma: a novel test system for drug susceptibility and mechanisms of resistance. Br J Cancer 110(2):479–488
Carranza-Torres IE, Guzmán-Delgado NE, Coronado-Martínez C et al (2015) Organotypic culture of breast tumour explants as a multicellular system for the screening of natural compounds with antineoplastic potential. Biomed Res Int 2015:618021
Acknowledgments
We would like to acknowledge and thank Thames West Unicorn and Topic of Cancer charities for funding the vibratome. We would also like to thank Guy Simpson for his editing and reviewing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Relph, K., Annels, N., Smith, C., Kostalas, M., Pandha, H. (2020). Oncolytic Immunotherapy for Bladder Cancer Using Coxsackie A21 Virus: Using a Bladder Tumor Precision-Cut Slice Model System to Assess Viral Efficacy. In: Engeland, C. (eds) Oncolytic Viruses. Methods in Molecular Biology, vol 2058. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9794-7_16
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9794-7_16
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9793-0
Online ISBN: 978-1-4939-9794-7
eBook Packages: Springer Protocols