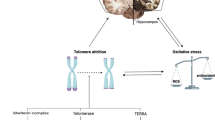
Abstract
Measuring the length of telomeres, repetitive nucleotide sequences cap** the chromosomes which shortens by each cell division, has become a popular way of attaining a marker of biological aging processes. Several observational studies have investigated the associations between telomere length and Alzheimer’s disease (AD) with the overall conclusion being shorter telomeres provide an increased risk for AD development. Here we present an alternative approach for addressing the topic where additional evidence on causality can be drawn. To do so, we include information on single nucleotide polymorphisms (SNPs) using nature’s own experiment with random segregation of alleles at conception. The protocol describes the full process of the so-called Mendelian Randomization by selecting appropriate SNPs for the analysis, discussing different data sources that can be used and inform about methods, assumptions and suitable software packages including Stata code.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Needham BL, Rehkopf D, Adler N et al (2015) Leukocyte telomere length and mortality in the National Health and Nutrition Examination Survey, 1999–2002. Epidemiology 26(4):528–535. https://doi.org/10.1097/EDE.0000000000000299
Hochstrasser T, Marksteiner J, Humpel C (2012) Telomere length is age-dependent and reduced in monocytes of Alzheimer patients. Exp Gerontol 47(2):160–163. https://doi.org/10.1016/j.exger.2011.11.012
Honig LS, Kang MS, Schupf N et al (2012) Association of shorter leukocyte telomere repeat length with dementia and mortality. Arch Neurol 69(10):1332–1339. https://doi.org/10.1001/archneurol.2012.1541
Thomas P, NJ OC, Fenech M (2008) Telomere length in white blood cells, buccal cells and brain tissue and its variation with ageing and Alzheimer’s disease. Mech Ageing Dev 129(4):183–190. https://doi.org/10.1016/j.mad.2007.12.004
Lukens JN, Van Deerlin V, Clark CM et al (2009) Comparisons of telomere lengths in peripheral blood and cerebellum in Alzheimer’s disease. Alzheimers Dement 5(6):463–469. https://doi.org/10.1016/j.jalz.2009.05.666
Boccardi V, Pelini L, Ercolani S et al (2015) From cellular senescence to Alzheimer’s disease: the role of telomere shortening. Ageing Res Rev 22:1–8. https://doi.org/10.1016/j.arr.2015.04.003
Lawlor DA, Harbord RM, Sterne JA et al (2008) Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat Med 27(8):1133–1163. https://doi.org/10.1002/sim.3034
Zhan Y, Song C, Karlsson R et al (2015) Telomere length shortening and Alzheimer disease—a Mendelian randomization study. JAMA Neurol 72(October):1202–1203
Lawlor DA (2016) Commentary: two-sample Mendelian randomization: opportunities and challenges. Int J Epidemiol 45(3):908–915. https://doi.org/10.1093/ije/dyw127
Burgess S (2014) Sample size and power calculations in Mendelian randomization with a single instrumental variable and a binary outcome. Int J Epidemiol 43(3):922–929. https://doi.org/10.1093/ije/dyu005
Smith GD, Ebrahim S (2003) ‘Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol 32(1):1–22
Palmer TM, Sterne JA, Harbord RM et al (2011) Instrumental variable estimation of causal risk ratios and causal odds ratios in Mendelian randomization analyses. Am J Epidemiol 173(12):1392–1403. https://doi.org/10.1093/aje/kwr026. kwr026 [pii]
Fall T, Hagg S, Magi R et al (2013) The role of adiposity in cardiometabolic traits: a Mendelian randomization analysis. PLoS Med 10(6):e1001474. https://doi.org/10.1371/journal.pmed.1001474
Codd V, Nelson CP, Albrecht E et al (2013) Identification of seven loci affecting mean telomere length and their association with disease. Nat Genet 45(4):422–427., 427e421-422. https://doi.org/10.1038/ng.2528
Johnson T (2013) Gtx: Genetics Toolbox. 0.0.8 edn
Lambert JC, Ibrahim-Verbaas CA, Harold D et al (2013) Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat Genet 45(12):1452–1458. https://doi.org/10.1038/ng.2802
Burgess S, Scott RA, Timpson NJ et al (2015) Using published data in Mendelian randomization: a blueprint for efficient identification of causal risk factors. Eur J Epidemiol 30(7):543–552. https://doi.org/10.1007/s10654-015-0011-z
Burgess S, Butterworth A, Thompson SG (2013) Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol 37(7):658–665. https://doi.org/10.1002/gepi.21758
Bowden J, Davey Smith G, Burgess S (2015) Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 44(2):512–525. https://doi.org/10.1093/ije/dyv080
Burgess S, Davies NM, Thompson SG et al (2014) Instrumental variable analysis with a nonlinear exposure-outcome relationship. Epidimiology 25(6):877–885. https://doi.org/10.1097/EDE.0000000000000161
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Zhan, Y., Hägg, S. (2018). Telomere Length Shortening in Alzheimer’s Disease: Procedures for a Causal Investigation Using Single Nucleotide Polymorphisms in a Mendelian Randomization Study. In: Perneczky, R. (eds) Biomarkers for Alzheimer’s Disease Drug Development. Methods in Molecular Biology, vol 1750. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7704-8_20
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7704-8_20
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7703-1
Online ISBN: 978-1-4939-7704-8
eBook Packages: Springer Protocols