Abstract
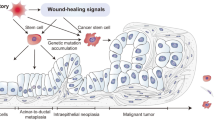
The hierarchical model of the origin of cancer from stem cells posits that cancer cells arise from undifferentiated (in the case of germinal tumors) or tissue-determined stem cells during the process of tissue renewal due to a block in the ability of the cells of the lineage to differentiate. The state of differentiation of the cancer depends on the level of maturation at which the arrest occurs. If arrest occurs early in the maturation process the tumors will be poorly differentiated; if later, they will be more differentiated. Examples presented in this review are included for each of the major mechanisms by which cancers arise: field theory (teratocarcinoma); chemical exposure (hepatocellular carcinoma, epithelial (skin) cancer); mutations (leukemia, colon cancer); and epigenetic change (gastric cancer). In each of these examples there is a block in maturation whereby the tissue stem cell produces cancerous progeny that express a phenotype that allows for continued proliferation and inhibition of differentiation. Differentiation therapy is based on the concept of removing the block to maturation arrest.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Pierce GB, Shikes R, Fink LM. Cancer: a problem of developmental biology. Englewood Cliffs, NJ: Prentice Hall; 1978. p. 1–242.
Pierce GB, Spears WC. Tumors as caricatures of the process of tissue renewal: prospects for therapy by directing differentiation. Cancer Res. 1988;48:1196–204.
Pierce BB, Wallace C. Differentiation of malignant to benign cells. Cancer Res. 1971;31:127–34.
Sell S, Pierce GB. Biology of disease: maturation arrest of stem cell differentiation is a common pathway for the cellular origin of teratocarcinomas and epithelial cancers. Lab Invest. 1994;70:6–21.
Shimkin M. Contrary to nature. Washington, DC: NIH, USDOH; 1977.
Bainbridge WS. The cancer problem. New York: Macmillan; 1914.
Recamier JCA. Recherches sur the traitement du cancer: par la compression methodique simple ou combinee, et sur l’histoire General de la meme maladie, vol. 2. Paris: Gabon; 1829.
Remak R. Ein beitrag zur entwickelungsgeschichte der krebshaften geschwulste. Deut Klin. 1854;6:70–174.
Durante F. Nesso fisio-pathologico tra la struttura dei nei materni e la genesi di alcuni tumori maligni. Arch Memor Osser Chir Pract. 1874;11:217–26.
Cohnheim J. Congenitales, quergestreiftes muskelsarkon der nireren. Virchows Arch. 1875;65:64.
Osler W, McCrea T. Modern medicine: it’s theory and practice. Philadelphia: Lean and Febiger; 1913. p. 5254.
Virchow R. Dir krankhoften geschwulste, vol. II (pt. 1). Berlin: Onkologie; 1863.
Dixon FJ, Moore RA. Testicular tumors: a clinicopathological study. Cancer. 1953;6:417–43.
Damjanov I. Pathobiology of human germ cell tumors. Recent Results Cancer Res. 1991;123:1–34.
Solter D, Damjanov I. Teratocarcinoma and the expression of oncodevelopmental genes. Methods Cancer Res. 1979;18: 277–98.
Chan D, Sell S. Tumor markers, chapter 23. In: Burtis CA, Ashwood ER, editors. Tietz textbook of clinical chemistry. 3rd ed. Philadelphia, PA: Saunders; 1999. p. 722–49.
Peyron A. Sur la presence des cellules genitales primordiales dans les boutons embryononnaires des embryomes parthenogenetiques chez l’homme. C R Acad Sci. 1938;206:1680–3.
Stevens LC. Experimental production of testicular teratomas in mice. Proc Natl Acad Sci USA. 1964;52:654–61.
Stevens LC. Origin of testicular teratomas from primordial germ cells in mice. J Natl Cancer Inst. 1967;38:549–52.
Brinster RL. The effect of cells transferred into the mouse blastocyst on subsequent development. J Exp Med. 1974;140:1049–56.
Mintz B, Illmensee K. Normal genetically mosaic mice produced from malignant teratocarcinoma cells. Proc Natl Acad Sci USA. 1975;72:3583–9.
Ilmmenesee K. Reversion of malignancy and normalized differentiation of teratocarcinoma cells in mammals. In: Russel LC, editor. Generic mosaics and chimeras in mammals. New York: Plenum; 1978. p. 3–25.
Papaionnou VE, McBurney MW, Gardner RL, Evans RL. Fate of teratocarcinoma cells injected into early mouse embryos. Nature. 1975;258:70–3.
Papaioannou VE. Ontogen, pathology, oncology. Int J Dev Biol. 1993;37:33–7.
Pierce GB, Dixon FJ. The demonstration of teratogenesis by metamorphosis of multipotential cells. Cancer. 1959;12:573–83.
Pierce GM, Dixon FJ, Verney E. Teratocarcinogenic and tissue forming potentials of the cell types comprising neoplastic embryoid bodies. Lab Invest. 1960;9:583–602.
Sell S. Stem cell origin of cancer and differentiation therapy. Crit Rev Oncol Hematol. 2004;51:1–28.
Sun SY, Lotan R. Retinoids and their receptors in cancer development and chemoprevention. Crit Rev Oncol Hematol. 2002;41:41–55.
Sell S. On the stem cell origin of cancer. Am J Pathol. 2010;176:2584–94.
Einhorm L. Are there factors preventing cancer development during embryonic life? Oncodev Biol Med. 1982;4:219–29.
Rl B. Radiation teratogenesis. Teratology. 1980;21:281–98.
Pierce GB. The cancer cell and its control by the embryo. Am J Pathol. 1983;113:116–24.
Pierce GB, Lewis SH, Miller GJ, Morits E, Miller P. Tumorigenicity of embryonal carcinoma as an assay to study control of malignancy by the murine blastocyst. Proc Natl Acad Sci USA. 1979;76:6649–51.
Pierce GB, Podesta A, Wells RS. The role of the blastocyst trophoderm in control of colony formation. In: Silver S, Strickland S, Martin G, editors. Teratocarcinoma stem cells, Cold Spring Harbor Symposium. New York; 1983. p. 15–22.
Pierce GB, Pantazis CG, Caldwell JE, Wells RS. Specificity of the control of tumor formation by blastocysts. Cancer Res. 1982;42(3):1082–7.
Gershenson M, Graves K, Carson D, Wells RS, Pierce GB. Regulation of melanoma by the embryonic skin. Proc Natl Acad Sci USA. 1986;83:7307–10.
Webb CW, Gootwine E, Sachs L. Developmental potential of myeloid leukemia cells injected into rat midgestation embryos. Dev Biol. 1984;101:221–4.
Coleman WB, Wennerberg AE, Smith GJ, Grisham JW. Regulation of differentiation of diploid and some aneuploid rat liver epithelial (stemlike) cells by the hepatic microenvironment. Am J Pathol. 1993;142:1373–82.
Weaver V, Petersen O, Wang F, Larabell C, Briand P, Damsky C, Bissel M. Reversion of the malignant phenotype of human breast cancer in three-dimensional culture and in vivo by integrin blocking bodies. J Cell Biol. 1997;137:231–45.
Postovit LM, Maragaryan NV, Seftor EA, et al. Human embryonic stem cell microenvironment suppresses the tumorigenic phenotype of aggressive cancer cells. Proc Natl Acad Sci USA. 2008;18:105–11.
Hendrix MJ, Seftor EA, Seftor REB, Kaisermeier-Kulesa J, Duleas PM, Postovit LM. Reprogramming metastatic tumor cells with the embryonic microenvironment. Nat Rev Cancer. 2007;7:246–55.
Kulesa PM, Kasemeier-Kulesa JC, Teddy JM, Margaryan NV, Seftor EA, Seftor RE, Hendrix MJ. Reprogramming metastatic tumor cells to assume a neural crest-like phenotype in an embryonic microenvironment. Proc Natl Acad Sci USA. 2006;103:3752–7.
Gootwine E, Webb CG, Sachs L. Participation of myeloid leukemia cells injected into embryos in haematopoietic differentiation in adult mice. Nature. 1982;299:63–5.
Biava PM, Fiorito A, Negro C, Mariani M. Effect of treatment with embryonic and uterine tissue homogenates on Lewis lung carcinoma development. Cancer Lett. 1988;41:265–70.
Biava PM, Bonsignorio D, Hoxa M. Life-protecting factor (LPF): an anti-cancer low molecular weight fraction isolated from pregnant uterine mucosa during embryo organogenesis. J Tumor Marker Oncol. 2000;15:223–33.
Biava PM, Bonsignorio D, Hoxa M, Impagliazzo M, Frosi A, Larese F, Negro C. Mother-embryo cross-talk: the anti-cancer substances produced by mother and embryo during cell differentiation. A review of experimental data. J Tumor Marker Oncol. 2002;17:55–8.
McKinnel RG, Deggins BA, Labat DD. Transplantation of pluripotential nuclei from triploid from tumors. Science. 1996;165:294–396.
Li L, Connelly MC, Wetmore C, Currant T, Morgan JI. Mouse embryos cloned from brain tumors. Cancer Res. 2003;63:2733–7.
Biava PM, Carluccio A. Activation of anti-oncogene p53 produced by embryonic extracts in vitro tumor cells. J Tumor Marker Oncol. 1977;12:9–15.
Biava PM, Bonsignorio D, Hoxa M, Facco R, Ielapi T, Frati L, Bizzarri M. Post-translational modification of the retinoblastoma protein (pRb) induced by in vitro administration of Zebrafish embryonic extracts on kidney adenocarcinoma cell line. J Tumor Marker Oncol. 2002;17:59–64.
Cucina A, Biava PM, D’Anselmi F, Coluccia P, Conti F, Di Clemente R, Miccheli A, Frati L, Gulino A, Bizzarri M. Zebrafish embryo proteins induce apoptosis in human colon cancer cells (Caco2). Apoptosis. 2006;9:1617–28.
Biava PM, Basevi M, Biggiero L, Borgonovo A, Borgonovo E, Burigana F. Cancer cell reprogramming: stem cell differentiation stage factors and an agent based model to optimize cancer treatment. Curr Pharm Biotechnol. 2011;12:231–42.
Dobson, J. ‘Percivall Pott’ in Annals of The Royal College of Surgeons of England; 1972. vol. 50, p. 54–65.
Rehn L. Blasengeschwulse bei fuchsin-arbeitern. Arch Klin Chir. 1895;50:588–600.
Farber E. Similarities in the sequence of early histologic changes induced in the liver by ethionine, 2-acetylaminofluorene, and 3′-methyl−4-dimethylamionazobenzene. Cancer Res. 1956;16:142–8.
Farber E. The multistep nature of cancer development. Cancer Res. 1984;44:4217–23.
Abelev GI. Alphafetoprotein: the genesis. Oncodev Biol Med. 1983;4:371–81.
Sell S. Alpha-fetoprotein, stem cells, and cancer. The abbot award lecture. Tumor Biol. 2008;29:161–80. doi:10.1159/000143402.
Sell S, Leffert HL. An evaluation of cellular lineages in the pathogenesis of experimental hepatocellular carcinoma. Hepatology. 1982;2:77–86.
Sell S, Dunsford H. Evidence for the stem cell origin of hepatocellular carcinoma and cholangiocarcinoma. Am J Pathol. 1989;134:1347–63.
Sell S, Leffert HL. Liver cancer stem cells. J Clin Oncol. 2008;26:2800–5.
Sell S. Stem cells in hepatocarcinogenesis. Cell Science Reviews. ISN NO. 2003;3:1742–8130.
Guest I, Ilic Z, Sell S. Age dependence of oval cell responses and bile duct carcinomas in male Fischer 344 rats fed a cyclic choline-deficient, ethionine-supplemented diet. Hepatology. 2010;52: 1750–7.
Korsching E, Jeffrey SS, et al. Basal carcinoma of the breast revisited: an old entity with new interpretations. J Clin Pathol. 2008;61:553–60.
Pott P. Chirurgical observations relative to the cataract, the polypus of the nose, the cancer of the scrotum, the different kinds of ruptures, and the mortification of the toes and feet. London: Hawes, Clarke and Collins; 1775.
Yamagiwa K, Ichikawa K. Experimental study of the pathogenesis of carcinoma. J Cancer Res. 1918;3:1–29.
Rous P, Kidd JG. Conditional neoplasms and subthreshold neoplastic states. J Exp Med. 1942;73:365–72.
Berenblum I. The mechanism of carcinogenesis. A study of the significance of cocarcinogenic action and related phenomena. Cancer Res. 1941;1:807.
Boutwell RK. Some biological aspects of skin carcinogenesis. Prog Exp Tumor Res. 1964;4:207–50.
Van Duuren BL, Sivak A, Katz C, Seidman I, Melchionne S. The effect of ageing and interval between primary and secondary treatment in two-stage carcinogenesis on mouse skin. Cancer Res. 1975;35:502–5.
Potten CS, Morris RJ. Epithelial stem cells in vivo. J Cell Sci Suppl. 1988;10:45–62.
Nowell PC. Diagnostic and prognostic value of chromosome studies in cancer. Ann Clin Lab Sci. 1974;4:234–40.
Rowley JD. Nonrandom chromosomal abnormalities in hematologic disorders of man. Proc Natl Acad Sci USA. 1975;72:152–6.
Chalandon Y, Schwaller J. Targeting mutated protein tyrosine kinases and their signaling pathways in hematologic malignancies. Haematologica. 2005;90:949–68.
Epstein MA, Achong BG, Barr YM. Virus particles in cultured lymphoblasts from Burkitt’s lymphoma. Lancet. 1964;1:702–3.
Cory S, Vaux DL, Harris AW, Adams JM. Insights from Bcl-2 and Myc: malignancy involves abrogation of apoptosis as well as sustained proliferation. Cancer Res. 1999;59(7 Suppl):1681s–92s.
Park SS, Kim JS, Tessarollo L, Owens JD, Peng L, Han SS, Tae Chung S, Torrey TA, Cheung WC, Plakiewica RD, McNeil N, Ried T, Buchinski JF, Morse 3rd HC, Janz S. Insertion of c-Myc into Igh induces B-cell and plasma-cell neoplasms in mice. Cancer Res. 2005;65:1306–15.
Sell S. Leukemia: stem cells, maturation arrest and differentiation therapy. Stem Cell Rev. 2005;1:197–205.
Drucker BJ, Ralpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM, Lydon NB, Kantarjian J, Capdeville R, Ohno-Jones S, Sawyers CL. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med. 2001;334: 1031–7.
Drucker BJ, Cuilhot F, O’Brien SG, et al. Five year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355:2408–17.
Melnick A, Licht JD. Deconstructing a disease: RARα, its fusion partners, and their roles in the pathogenesis of acute promyelocytic leukemia. Blood. 1999;93:3167–215.
Soignet S, Fleischauer A, Pollyak T, Heller G, Warrel Jr RP. All trans retinoic acid significantly increases 5-year survival in patients with acute promyelocytic leukemia: long term follow-up of the New York study. Cancer Chemother Pharmacol. 1997;40: S24–9.
Nowell PC. The clonal evolution of tumor cell populations. Science. 1976;194:23–8.
Fowler JF. Radiation biology as applied to radiotherapy. Curr Top Radiat Res. 1966;2:304–64.
Trott KR. Tumour stem cells: the biological concept and its application in cancer treatment. Radiother Oncol. 1994;30:1–5.
Denekamp J. Tumour stem cells: facts, interpretation and consequences. Radiother Oncol. 1994;30:6–10.
Eric R, Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61:759–67.
Zhang T, Otevrel T, Gao Z, Gao Z, Ehrlich SM, Fields JZ, Bowman BM. Evidence that APC regulates surviving expression: a possible mechanism contributing to the stem cell origin of colon cancer. Cancer Res. 2001;62:8664–7.
Brittan M, Wright NA. Stem cell origin of cell lineages, proliferative units, and cancer in the gastrointestinal tract. In: Sell S, editor. Stem cell handbook. Totowa, NJ: Humana; 2004. p. 329.
Slaughter DP, Southwick HW, Smejkal W. Field cancerization in oral stratified squamous epithelium. Clinical implications of multicentric origin. Cancer. 1953;6:963–8.
Carlson JA, Scott D, Wharton J, Sell S. Incidental histopathologic patterns: possible evidence of “field cancerization” surrounding skin tumors. Am J Dermatopathol. 2001;223:494–7.
Braakhuis JMB, Tabor MP, Kummer JA, Leemans CR, Brakenhoff RH. A genetic explanation of Slaughter’s concept of field cancerization: evidence and clinical implications. Cancer Res. 2003;63:1727–30.
Ushijima T, Nakajima T, Maekita T. DNA methylation as a marker for the past and future. J Gastroenterol. 2006;41:401–7. doi:10.1007/s00535-006-1846-6.
Ushijima T. Epigenetic field for cancerization. J Biochem Mol Biol. 2007;40:142–50.
Ushijima T. Detection and interpretation of altered methylation patterns in cancer cells. Nat Rev Cancer. 2005;5:223–31. doi:10.1038/nrc1571.
Godlblatt H, Cameron G. Induced malignancy in cells from rat myocardium subjected to intermittent anaerobiosis during long propagation in vitro. J Exp Med. 1953;97:525–62.
Warberg O, Wind F, Negelein E. The metabolism of tumors in the body. J Gen Physiol. 1927;8:519–30. PMCID: PMC2140820.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science + Business Media New York
About this chapter
Cite this chapter
Sell, S., Biava, P.M. (2013). Cancer Is a Problem of Developmental Biology: Maturation Arrest and the Hierarchical Model of the Origin of Cancer from Stem Cells. In: Sell, S. (eds) Stem Cells Handbook. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4614-7696-2_31
Download citation
DOI: https://doi.org/10.1007/978-1-4614-7696-2_31
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4614-7695-5
Online ISBN: 978-1-4614-7696-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)