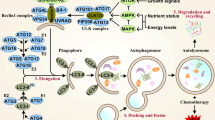
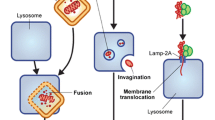
Abstract
Autophagy is a cellular survival mechanism influenced by a wide variety of intracellular and extracellular stresses including energy and oxygen deprivation, signaling aberrancies, ER stress, DNA damage, systemic cancer therapies, and radiotherapies. There is growing evidence that it may potentiate cancer survival. A number of clinical trials have been launched using autophagy inhibition in combination with standard cancer therapeutics. The information gleaned from these as well as ongoing in vitro and in vivo studies will allow us to better understand the role of autophagy in cancer.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ashford TP, Porter KR (1962) Cytoplasmic components in hepatic cell lysosomes. J Cell Biol 12:198–202
Watanabe E et al (2009) Sepsis induces extensive autophagic vacuolization in hepatocytes: a clinical and laboratory-based study. Lab Invest 89(5):549–561
Amaravadi RK et al (2007) Autophagy inhibition enhances therapy-induced apoptosis in a Myc-induced model of lymphoma. J Clin Invest 117(2):326–336
Klionsky DJ (2007) Autophagy: from phenomenology to molecular understanding in less than a decade. Nat Rev Mol Cell Biol 8(11):931–937
Kroemer G, Jaattela M (2005) Lysosomes and autophagy in cell death control. Nat Rev Cancer 5(11):886–897
Levine B, Deretic V (2007) Unveiling the roles of autophagy in innate and adaptive immunity. Nat Rev Immunol 7(10):767–777
Muller O et al (2000) Autophagic tubes: vacuolar invaginations involved in lateral membrane sorting and inverse vesicle budding. J Cell Biol 151(3):519–528
Bandyopadhyay U et al (2008) The chaperone-mediated autophagy receptor organizes in dynamic protein complexes at the lysosomal membrane. Mol Cell Biol 28(18):5747–5763
Chen HY, White E (2011) Role of autophagy in cancer prevention. Cancer Prev Res (Phila) 4(7):973–983
He C, Klionsky DJ (2009) Regulation mechanisms and signaling pathways of autophagy. Annu Rev Genet 43:67–93
Gu Y, Wang C, Cohen A (2004) Effect of IGF-1 on the balance between autophagy of dysfunctional mitochondria and apoptosis. FEBS Lett 577(3):357–360
Bjorkoy G et al (2005) p62/SQSTM1 forms protein aggregates degraded by autophagy and has a protective effect on huntingtin-induced cell death. J Cell Biol 171(4):603–614
Wild P, Dikic I (2010) Mitochondria get a Parkin’ ticket. Nat Cell Biol 12(2):104–106
Di Bartolomeo S, Nazio F, Cecconi F (2010) The role of autophagy during development in higher eukaryotes. Traffic 11(10):1280–1289
Mizushima N et al (2008) Autophagy fights disease through cellular self-digestion. Nature 451(7182):1069–1075
Wang CW, Klionsky DJ (2003) The molecular mechanism of autophagy. Mol Med 9(3–4):65–76
Kovacs AL, Zhang H (2010) Role of autophagy in Caenorhabditis elegans. FEBS Lett 584(7):1335–1341
Chang YY, Neufeld TP (2010) Autophagy takes flight in drosophila. FEBS Lett 584(7):1342–1349
Rong Y et al (2011) Spinster is required for autophagic lysosome reformation and mTOR reactivation following starvation. Proc Natl Acad Sci USA 108(19):7826–7831
Teter SA et al (2001) Degradation of lipid vesicles in the yeast vacuole requires function of Cvt17, a putative lipase. J Biol Chem 276(3):2083–2087
Epple UD et al (2001) Aut5/Cvt17p, a putative lipase essential for disintegration of autophagic bodies inside the vacuole. J Bacteriol 183(20):5942–5955
Yang Z et al (2006) Atg22 recycles amino acids to link the degradative and recycling functions of autophagy. Mol Biol Cell 17(12):5094–5104
Kabeya Y et al (2005) Atg17 functions in cooperation with Atg1 and Atg13 in yeast autophagy. Mol Biol Cell 16(5):2544–2553
Jung CH et al (2009) ULK-Atg13-FIP200 complexes mediate mTOR signaling to the autophagy machinery. Mol Biol Cell 20(7):1992–2003
Mizushima N (2010) The role of the Atg1/ULK1 complex in autophagy regulation. Curr Opin Cell Biol 22(2):132–9
Cheong H et al (2011) Ammonia-induced autophagy is independent of ULK1/ULK2 kinases. Proc Natl Acad Sci USA 108(27):11121–11126
Hailey DW et al (2010) Mitochondria supply membranes for autophagosome biogenesis during starvation. Cell 141(4):656–667
Ravikumar B et al (2010) Plasma membrane contributes to the formation of pre-autophagosomal structures. Nat Cell Biol 12(8):747–757
English L et al (2009) Autophagy enhances the presentation of endogenous viral antigens on MHC class I molecules during HSV-1 infection. Nat Immunol 10(5):480–487
Webber JL, Tooze SA (2010) New insights into the function of Atg9. FEBS Lett 584(7):1319–1326
Hayashi-Nishino M et al (2009) A subdomain of the endoplasmic reticulum forms a cradle for autophagosome formation. Nat Cell Biol 11(12):1433–1437
Yla-Anttila P et al (2009) 3D tomography reveals connections between the phagophore and endoplasmic reticulum. Autophagy 5(8):1180–1185
Funderburk SF, Wang QJ, Yue Z (2010) The Beclin 1-VPS34 complex–at the crossroads of autophagy and beyond. Trends Cell Biol 20(6):355–2
Itakura E et al (2008) Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol Biol Cell 19(12):5360–5372
Matsunaga K et al (2009) Two Beclin 1-binding proteins, Atg14L and Rubicon, reciprocally regulate autophagy at different stages. Nat Cell Biol 11(4):385–396
Kabeya Y et al (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19(21):5720–5728
Ichimura Y et al (2000) A ubiquitin-like system mediates protein lipidation. Nature 408(6811):488–492
Yoshimura K et al (2006) Effects of RNA interference of Atg4B on the limited proteolysis of LC3 in PC12 cells and expression of Atg4B in various rat tissues. Autophagy 2(3):200–208
Satoo K et al (2009) The structure of Atg4B-LC3 complex reveals the mechanism of LC3 processing and delipidation during autophagy. EMBO J 28(9):1341–1350
Tanida I et al (2002) Human Apg3p/Aut1p homologue is an authentic E2 enzyme for multiple substrates, GATE-16, GABARAP, and MAP-LC3, and facilitates the conjugation of hApg12p to hApg5p. J Biol Chem 277(16):13739–13744
Tanida I et al (2001) The human homolog of Saccharomyces cerevisiae Apg7p is a protein-activating enzyme for multiple substrates including human Apg12p, GATE-16, GABARAP, and MAP-LC3. J Biol Chem 276(3):1701–1706
Nair U et al (2011) SNARE proteins are required for macroautophagy. Cell 146(2):290–302
Pankiv S et al (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282(33):24131–24145
Schweers RL et al (2007) NIX is required for programmed mitochondrial clearance during reticulocyte maturation. Proc Natl Acad Sci USA 104(49):19500–19505
Kim PK et al (2008) Ubiquitin signals autophagic degradation of cytosolic proteins and peroxisomes. Proc Natl Acad Sci USA 105(52):20567–20574
Novak I et al (2010) Nix is a selective autophagy receptor for mitochondrial clearance. EMBO Rep 11(1):45–51
Sandoval H et al (2008) Essential role for nix in autophagic maturation of erythroid cells. Nature 454(7201):232–235
Jager S et al (2004) Role for Rab7 in maturation of late autophagic vacuoles. J Cell Sci 117(Pt 20):4837–4848
Djeddi A et al (2012) Induction of autophagy in ESCRT mutants is an adaptive response for cell survival in C. elegans. J Cell Sci 125(Pt 3):685–694
Fader CM, Colombo MI (2009) Autophagy and multivesicular bodies: two closely related partners. Cell Death Differ 16(1):70–78
Young AR et al (2006) Starvation and ULK1-dependent cycling of mammalian Atg9 between the TGN and endosomes. J Cell Sci 119(Pt 18):3888–3900
Manjithaya R et al (2010) Unconventional secretion of Pichia pastoris Acb1 is dependent on GRASP protein, peroxisomal functions, and autophagosome formation. J Cell Biol 188(4):537–46
Kabeya Y et al (2004) LC3, GABARAP and GATE16 localize to autophagosomal membrane depending on form-II formation. J Cell Sci 117(Pt 13):2805–2812
Tanida I et al (2003) GATE-16 and GABARAP are authentic modifiers mediated by Apg7 and Apg3. Biochem Biophys Res Commun 300(3):637–644
Tanida I, Ueno T, Kominami E (2004) LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 36(12):2503–2518
Weidberg H et al (2010) LC3 and GATE-16/GABARAP subfamilies are both essential yet act differently in autophagosome biogenesis. EMBO J 29(11):1792–1802
Marino G et al (2003) Human autophagins, a family of cysteine proteinases potentially implicated in cell degradation by autophagy. J Biol Chem 278(6):3671–3678
Behrends C et al (2010) Network organization of the human autophagy system. Nature 466(7302):68–76
Lee SJ et al (2011) Beclin 1 deficiency is associated with increased hypoxia-induced angiogenesis. Autophagy 7(8):829–839
Wirawan E et al (2012) Beclin1: a role in membrane dynamics and beyond. Autophagy 8(1):6–17
Nishida Y et al (2009) Discovery of Atg5/Atg7-independent alternative macroautophagy. Nature 461(7264):654–658
Klionsky DJ et al (2008) Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy 4(2):151–175
Tanida I et al (2005) Lysosomal turnover, but not a cellular level, of endogenous LC3 is a marker for autophagy. Autophagy 1(2):84–91
Ciechomska IA, Tolkovsky AM (2007) Non-autophagic GFP-LC3 puncta induced by saponin and other detergents. Autophagy 3(6):586–590
Kuma A, Matsui M, Mizushima N (2007) LC3, an autophagosome marker, can be incorporated into protein aggregates independent of autophagy: caution in the interpretation of LC3 localization. Autophagy 3(4):323–328
Kimura S, Noda T, Yoshimori T (2007) Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3(5):452–460
Sou YS et al (2008) The Atg8 conjugation system is indispensable for proper development of autophagic isolation membranes in mice. Mol Biol Cell 19(11):4762–4775
Mathew R et al (2009) Autophagy suppresses tumorigenesis through elimination of p62. Cell 137(6):1062–1075
Ma X et al (2011) Measurements of tumor cell autophagy predict invasiveness, resistance to chemotherapy, and survival in melanoma. Clin Cancer Res 17(10):3478–3489
Mendoza MC, Er EE, Blenis J (2011) The Ras-ERK and PI3 K-mTOR pathways: cross-talk and compensation. Trends Biochem Sci 36(6):320–328
Steelman LS et al (2011) Roles of the Raf/MEK/ERK and PI3 K/PTEN/Akt/mTOR pathways in controlling growth and sensitivity to therapy-implications for cancer and aging. Aging (Albany NY) 3(3):192–222
Blommaart EF et al (1995) Phosphorylation of ribosomal protein S6 is inhibitory for autophagy in isolated rat hepatocytes. J Biol Chem 270(5):2320–2326
Hosokawa N et al (2009) Nutrient-dependent mTORC1 association with the ULK1-Atg13-FIP200 complex required for autophagy. Mol Biol Cell 20(7):1981–1991
Sancak Y et al (2010) Ragulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141(2):290–303
Arico S et al (2001) The tumor suppressor PTEN positively regulates macroautophagy by inhibiting the phosphatidylinositol 3-kinase/protein kinase B pathway. J Biol Chem 276(38):35243–35246
Ueno T et al (2008) Loss of Pten, a tumor suppressor, causes the strong inhibition of autophagy without affecting LC3 lipidation. Autophagy 4(5):692–700
Laane E et al (2009) Cell death induced by dexamethasone in lymphoid leukemia is mediated through initiation of autophagy. Cell Death Differ 16(7):1018–1029
Kuo HP et al (2010) ARD1 stabilization of TSC2 suppresses tumorigenesis through the mTOR signaling pathway. Sci Signal 3(108):ra9
Zeng X, Kinsella TJ (2008) Mammalian target of rapamycin and S6 kinase 1 positively regulate 6-thioguanine-induced autophagy. Cancer Res 68(7):2384–2390
Yaswen P, Campisi J (2007) Oncogene-induced senescence pathways weave an intricate tapestry. Cell 128(2):233–234
Serrano M et al (1997) Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88(5):593–602
Elgendy M et al (2011) Oncogenic Ras-induced expression of Noxa and Beclin-1 promotes autophagic cell death and limits clonogenic survival. Mol Cell 42(1):23–35
Wu SY et al (2011) Ras-related tumorigenesis is suppressed by BNIP3-mediated autophagy through inhibition of cell proliferation. Neoplasia 13(12):1171–1182
Guo JY et al (2011) Activated Ras requires autophagy to maintain oxidative metabolism and tumorigenesis. Genes Dev 25(5):460–470
Yang S et al (2011) Pancreatic cancers require autophagy for tumor growth. Genes Dev 25(7):717–729
Maddodi N et al (2010) Induction of autophagy and inhibition of melanoma growth in vitro and in vivo by hyperactivation of oncogenic BRAF. J Invest Dermatol 130(6):1657–1667
Zhou W et al (2011) Small interfering RNA targeting mcl-1 enhances proteasome inhibitor-induced apoptosis in various solid malignant tumors. BMC Cancer 11:485
Pattingre S et al (2005) Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 122(6):927–939
Ku B et al (2008) An insight into the mechanistic role of Beclin 1 and its inhibition by prosurvival Bcl-2 family proteins. Autophagy 4(4):519–520
Hoyer-Hansen M, Jaattela M (2007) AMP-activated protein kinase: a universal regulator of autophagy? Autophagy 3(4):381–383
Liang J et al (2007) The energy sensing LKB1-AMPK pathway regulates p27(kip1) phosphorylation mediating the decision to enter autophagy or apoptosis. Nat Cell Biol 9(2):218–224
Egan D et al (2011) The autophagy initiating kinase ULK1 is regulated via opposing phosphorylation by AMPK and mTOR. Autophagy 7(6):643–644
Kim J, Guan KL (2011) Regulation of the autophagy initiating kinase ULK1 by nutrients: Roles of mTORC1 and AMPK. Cell Cycle 10(9):1337–1338
Alexander A, Walker CL (2010) Differential localization of ATM is correlated with activation of distinct downstream signaling pathways. Cell Cycle 9(18):3685–3686
Herrero-Martin G et al (2009) TAK1 activates AMPK-dependent cytoprotective autophagy in TRAIL-treated epithelial cells. EMBO J 28(6):677–685
Tzatsos A, Tsichlis PN (2007) Energy depletion inhibits phosphatidylinositol 3-kinase/Akt signaling and induces apoptosis via AMP-activated protein kinase-dependent phosphorylation of IRS-1 at Ser-794. J Biol Chem 282(25):18069–18082
Xu J et al (2012) MicroRNAs in autophagy and their emerging roles in crosstalk with apoptosis. Autophagy 8(6):873–882
Baldwin AS (2012) Regulation of cell death and autophagy by IKK and NF-kappaB: critical mechanisms in immune function and cancer. Immunol Rev 246(1):327–345
** Y et al (2012) Autophagic proteins: New facets of the oxygen paradox. Autophagy 8(3):426–428
Wang RC, Levine B (2010) Autophagy in cellular growth control. FEBS Lett 584(7):1417–1426
Chaturvedi A, Pierce SK (2009) Autophagy in immune cell regulation and dysregulation. Curr Allergy Asthma Rep 9(5):341–346
Wang L et al (2012) The roles of integrin beta4 in vascular endothelial cells. J Cell Physiol 227(2):474–478
Lee J, Giordano S, Zhang J (2012) Autophagy, mitochondria and oxidative stress: cross-talk and redox signalling. Biochem J 441(2):523–540
Yang Z, Klionsky DJ (2010) Eaten alive: a history of macroautophagy. Nat Cell Biol 12(9):814–822
Levine B, Yuan J (2005) Autophagy in cell death: an innocent convict? J Clin Invest 115(10):2679–2688
Platini F et al (2010) Understanding autophagy in cell death control. Curr Pharm Des 16(1):101–113
Maiuri MC et al (2007) Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol 8(9):741–752
Chen Y, Azad MB, Gibson SB (2010) Methods for detecting autophagy and determining autophagy-induced cell death. Can J Physiol Pharmacol 88(3):285–295
Malhotra JD, Kaufman RJ (2007) The endoplasmic reticulum and the unfolded protein response. Semin Cell Dev Biol 18(6):716–731
Ron D, Walter P (2007) Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol 8(7):519–529
Bertolotti A et al (2000) Dynamic interaction of BiP and ER stress transducers in the unfolded- protein response. Nat Cell Biol 2(6):326–332
Bernales S, Schuck S, Walter P (2007) ER-phagy: selective autophagy of the endoplasmic reticulum. Autophagy 3(3):285–287
Yorimitsu T et al (2006) Endoplasmic reticulum stress triggers autophagy. J Biol Chem 281(40):30299–30304
Kruse KB, Brodsky JL, McCracken AA (2006) Autophagy: an ER protein quality control process. Autophagy 2(2):135–137
Younce CW, Kolattukudy PE (2010) MCP-1 causes cardiomyoblast death via autophagy resulting from ER stress caused by oxidative stress generated by inducing a novel zinc-finger protein, MCPIP. Biochem J 426(1):43–53
Rodriguez-Rocha H et al (2011) DNA damage and autophagy. Mutat Res 711(1–2):158–166
Rieber M, Rieber MS (2008) Sensitization to radiation-induced DNA damage accelerates loss of bcl-2 and increases apoptosis and autophagy. Cancer Biol Ther 7(10):1561–1566
Feng Z et al (2005) The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci USA 102(23):8204–8209
Malzer E et al (2010) Impaired tissue growth is mediated by checkpoint kinase 1 (CHK1) in the integrated stress response. J Cell Sci 123(Pt 17):2892–2900
Crighton D et al (2006) DRAM, a p53-induced modulator of autophagy, is critical for apoptosis. Cell 126(1):121–134
Lorin S et al (2010) Evidence for the interplay between JNK and p53-DRAM signalling pathways in the regulation of autophagy. Autophagy 6(1):153–154
Feng Z, Levine AJ (2010) The regulation of energy metabolism and the IGF-1/mTOR pathways by the p53 protein. Trends Cell Biol 20(7):427–434
Wu H et al (2011) Central role of lactic acidosis in cancer cell resistance to glucose deprivation-induced cell death. J Pathol 227(2):189–199
Takagi H et al (2007) AMPK mediates autophagy during myocardial ischemia in vivo. Autophagy 3(4):405–407
Mihaylova MM, Shaw RJ (2011) The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol 13(9):1016–1023
Egan DF et al (2011) Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science 331(6016):456–461
Persons DA et al (1989) Increased expression of glycolysis-associated genes in oncogene-transformed and growth-accelerated states. Mol Carcinog 2(2):88–94
Kawauchi K et al (2008) p53 regulates glucose metabolism through an IKK-NF-kappaB pathway and inhibits cell transformation. Nat Cell Biol 10(5):611–618
Kondoh H et al (2005) Glycolytic enzymes can modulate cellular life span. Cancer Res 65(1):177–185
Matoba S et al (2006) p53 regulates mitochondrial respiration. Science 312(5780):1650–1653
Noman MZ et al (2011) Blocking hypoxia-induced autophagy in tumors restores cytotoxic T-cell activity and promotes regression. Cancer Res 71(18):5976–5986
Rouschop KM et al (2010) The unfolded protein response protects human tumor cells during hypoxia through regulation of the autophagy genes MAP1LC3B and ATG5. J Clin Invest 120(1):127–141
Semenza GL (2011) Hypoxia-inducible factor 1: regulator of mitochondrial metabolism and mediator of ischemic preconditioning. Biochim Biophys Acta 1813(7):1263–1268
Djavaheri-Mergny M et al (2006) NF-kappaB activation represses tumor necrosis factor-alpha-induced autophagy. J Biol Chem 281(41):30373–30382
Chen JL et al (2008) Novel roles for protein kinase Cdelta-dependent signaling pathways in acute hypoxic stress-induced autophagy. J Biol Chem 283(49):34432–34444
Huang J, Brumell JH (2009) NADPH oxidases contribute to autophagy regulation. Autophagy 5(6):887–889
Mortimore GE, Schworer CM (1977) Induction of autophagy by amino-acid deprivation in perfused rat liver. Nature 270(5633):174–176
Dodd KM, Tee AR (2012) Leucine and mTORC1: a complex relationship. Am J Physiol Endocrinol Metab 302(11):E1329–1342
Liu XM et al (2004) Platelet-derived growth factor stimulates LAT1 gene expression in vascular smooth muscle: role in cell growth. FASEB J 18(6):768–770
Kashiwagi H et al (2009) Regulatory mechanisms of SNAT2, an amino acid transporter, in L6 rat skeletal muscle cells by insulin, osmotic shock and amino acid deprivation. Amino Acids 36(2):219–230
Williams GS, Molinelli EJ, Smith GD (2008) Modeling local and global intracellular calcium responses mediated by diffusely distributed inositol 1,4,5-trisphosphate receptors. J Theor Biol 253(1):170–188
Eng CH et al (2010) Ammonia derived from glutaminolysis is a diffusible regulator of autophagy. Sci Signal 3(119):ra31
Kanzawa T et al (2004) Role of autophagy in temozolomide-induced cytotoxicity for malignant glioma cells. Cell Death Differ 11(4):448–457
Repnik U, Turk B (2010) Lysosomal-mitochondrial cross-talk during cell death. Mitochondrion 10(6):662–669
Resau JH et al (1985) Studies on the mechanisms of altered exocrine acinar cell differentiation and ductal metaplasia following nitrosamine exposure using hamster pancreatic explant organ culture. Carcinogenesis 6(1):29–35
Bae H, Guan JL (2011) Suppression of autophagy by FIP200 deletion impairs DNA damage repair and increases cell death upon treatments with anticancer agents. Mol Cancer Res 9(9):1232–1241
Gonzalez-Malerva L et al (2011) High-throughput ectopic expression screen for tamoxifen resistance identifies an atypical kinase that blocks autophagy. Proc Natl Acad Sci USA 108(5):2058–2063
de Medina P, Silvente-Poirot S, Poirot M (2009) Tamoxifen and AEBS ligands induced apoptosis and autophagy in breast cancer cells through the stimulation of sterol accumulation. Autophagy 5(7):1066–1067
Wu WK et al (2010) Macroautophagy modulates cellular response to proteasome inhibitors in cancer therapy. Drug Resist Updat 13(3):87–92
Ropero S, Esteller M (2007) The role of histone deacetylases (HDACs) in human cancer. Mol Oncol 1(1):19–25
Carew JS et al (2007) Targeting autophagy augments the anticancer activity of the histone deacetylase inhibitor SAHA to overcome Bcr-Abl-mediated drug resistance. Blood 110(1):313–322
Park JH et al (2011) A new synthetic HDAC inhibitor, MHY218, induces apoptosis or autophagy-related cell death in tamoxifen-resistant MCF-7 breast cancer cells. Invest New Drugs 30(5):1887–1898
Yamamoto S et al (2008) Suberoylanilide hydroxamic acid (SAHA) induces apoptosis or autophagy-associated cell death in chondrosarcoma cell lines. Anticancer Res 28(3A)1585–91
Hrzenjak A et al (2008) SAHA induces caspase-independent, autophagic cell death of endometrial stromal sarcoma cells by influencing the mTOR pathway. J Pathol 216(4):495–504
Saitoh T, Akira S (2010) Regulation of innate immune responses by autophagy-related proteins. J Cell Biol 189(6):925–935
Zhao Z et al (2008) Autophagosome-independent essential function for the autophagy protein Atg5 in cellular immunity to intracellular pathogens. Cell Host Microbe 4(5):458–469
Dreux M, Chisari FV (2010) Viruses and the autophagy machinery. Cell Cycle 9(7):1295–1307
Amaravadi RK et al (2011) Principles and current strategies for targeting autophagy for cancer treatment. Clin Cancer Res 17(4):654–666
Michaud M et al (2011) Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science 334(6062):1573–1577
Liang XH et al (1999) Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature 402(6762):672–676
Aita VM et al (1999) Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics 59(1):59–65
Qu X et al (2003) Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J Clin Invest 112(12):1809–1820
Yue Z et al (2003) Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci USA 100(25):15077–15082
Takamura A et al (2011) Autophagy-deficient mice develop multiple liver tumors. Genes Dev 25(8):795–800
Degenhardt K et al (2006) Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell 10(1):51–64
Karantza-Wadsworth V et al (2007) Autophagy mitigates metabolic stress and genome damage in mammary tumorigenesis. Genes Dev 21(13):1621–1635
Mathew R, White E (2007) Why sick cells produce tumors: the protective role of autophagy. Autophagy 3(5):502–505
Komatsu M et al (2010) The selective autophagy substrate p62 activates the stress responsive transcription factor Nrf2 through inactivation of Keap1. Nat Cell Biol 12(3):213–223
Lau A et al (2010) A noncanonical mechanism of Nrf2 activation by autophagy deficiency: direct interaction between Keap1 and p62. Mol Cell Biol 30(13):3275–3285
Inami Y et al (2011) Persistent activation of Nrf2 through p62 in hepatocellular carcinoma cells. J Cell Biol 193(2):275–284
Young AR et al (2009) Autophagy mediates the mitotic senescence transition. Genes Dev 23(7):798–803
Sinha S, Levine B (2008) The autophagy effector Beclin 1: a novel BH3-only protein. Oncogene 27(Suppl 1):S137–S148
Guertin DA, Sabatini DM (2007) Defining the role of mTOR in cancer. Cancer Cell 12(1):9–22
Diaz-Troya S et al (2008) The role of TOR in autophagy regulation from yeast to plants and mammals. Autophagy 4(7):851–865
Maiuri MC et al (2007) BH3-only proteins and BH3 mimetics induce autophagy by competitively disrupting the interaction between Beclin 1 and Bcl-2/Bcl-X(L). Autophagy 3(4):374–376
Liu J et al (2011) Beclin1 controls the levels of p53 by regulating the deubiquitination activity of USP10 and USP13. Cell 147(1):223–234
Wei H et al (2011) Suppression of autophagy by FIP200 deletion inhibits mammary tumorigenesis. Genes Dev 25(14):1510–1527
Zhang HF et al (2009) ATG16L1 T300A polymorphism and Crohn’s disease susceptibility: evidence from 13,022 cases and 17,532 controls. Hum Genet 125(5–6):627–631
Kang C, Avery L (2008) To be or not to be, the level of autophagy is the question: dual roles of autophagy in the survival response to starvation. Autophagy 4(1):82–84
Park MA et al (2008) PERK-dependent regulation of HSP70 expression and the regulation of autophagy. Autophagy 4(3):364–367
Hsu KF et al (2009) Cathepsin L mediates resveratrol-induced autophagy and apoptotic cell death in cervical cancer cells. Autophagy 5(4):451–460
Bhoopathi P et al (2010) Cathepsin B facilitates autophagy-mediated apoptosis in SPARC overexpressed primitive neuroectodermal tumor cells. Cell Death Differ 17(10):1529–1539
Mathieu V et al (2007) Galectin-1 knockdown increases sensitivity to temozolomide in a B16F10 mouse metastatic melanoma model. J Invest Dermatol 127(10):2399–2410
Debnath J, Baehrecke EH, Kroemer G (2005) Does autophagy contribute to cell death? Autophagy 1(2):66–74
Lu Z et al (2008) The tumor suppressor gene ARHI regulates autophagy and tumor dormancy in human ovarian cancer cells. J Clin Invest 118(12):3917–3929
Lum JJ et al (2005) Growth factor regulation of autophagy and cell survival in the absence of apoptosis. Cell 120(2):237–248
O’Neill PM et al (1998) 4-Aminoquinolines–past, present, and future: a chemical perspective. Pharmacol Ther 77(1):29–58
Kremer JM (2001) Rational use of new and existing disease-modifying agents in rheumatoid arthritis. Ann Intern Med 134(8):695–706
Romanelli F, Smith KM, Hoven AD (2004) Chloroquine and hydroxychloroquine as inhibitors of human immunodeficiency virus (HIV-1) activity. Curr Pharm Des 10(21):2643–2648
Sotelo J, Briceno E, Lopez-Gonzalez MA (2006) Adding chloroquine to conventional treatment for glioblastoma multiforme: a randomized, double-blind, placebo-controlled trial. Ann Intern Med 144(5):337–343
Carmichael SJ, Charles B, Tett SE (2003) Population pharmacokinetics of hydroxychloroquine in patients with rheumatoid arthritis. Ther Drug Monit 25(6):671–681
Rosenfeld MRGS, Brem S, Mikkelson T, Wang D, Piao S, Davis L, O’Dwyer PJ, Amaravadi RK (2010) Pharmacokinetic analysis and pharmacodynamic evidence of autophagy inhibition in patients with newly diagnosed glioblastoma treated on a phase I trial of hydroxychloroquine in combination with adjuvant temozolomide and radiation (ABTC 0603). J Clin Oncol 28(15s):3086
Petiot A et al (2000) Distinct classes of phosphatidylinositol 3’-kinases are involved in signaling pathways that control macroautophagy in HT-29 cells. J Biol Chem 275(2):992–998
Wu YT et al (2010) Dual role of 3-methyladenine in modulation of autophagy via different temporal patterns of inhibition on class I and III phosphoinositide 3-kinase. J Biol Chem 285(14):10850–10861
Werner G et al (1984) Metabolic products of microorganisms. 224. Bafilomycins, a new group of macrolide antibiotics. Production, isolation, chemical structure and biological activity. J Antibiot (Tokyo) 37(2):110–117
Dassonneville L, Bailly C (1999) Stimulation of topoisomerase II-mediated DNA cleavage by an indazole analogue of lucanthone. Biochem Pharmacol 58(8):1307–1312
Luo M, Kelley MR (2004) Inhibition of the human apurinic/apyrimidinic endonuclease (APE1) repair activity and sensitization of breast cancer cells to DNA alkylating agents with lucanthone. Anticancer Res 24(4):2127–2134
Carew JS et al (2011) Lucanthone is a novel inhibitor of autophagy that induces cathepsin D-mediated apoptosis. J Biol Chem 286(8):6602–6613
Leu JI et al (2011) HSP70 inhibition by the small-molecule 2-phenylethynesulfonamide impairs protein clearance pathways in tumor cells. Mol Cancer Res 9(7):936–947
Nice DC et al (2002) Cooperative binding of the cytoplasm to vacuole targeting pathway proteins, Cvt13 and Cvt20, to phosphatidylinositol 3-phosphate at the pre-autophagosomal structure is required for selective autophagy. J Biol Chem 277(33):30198–30207
Guan J et al (2001) Cvt18/Gsa12 is required for cytoplasm-to-vacuole transport, pexophagy, and autophagy in Saccharomyces cerevisiae and Pichia pastoris. Mol Biol Cell 12(12):3821–3838
Stromhaug PE et al (2004) Atg21 is a phosphoinositide binding protein required for efficient lipidation and localization of Atg8 during uptake of aminopeptidase I by selective autophagy. Mol Biol Cell 15(8):3553–3566
Tanida I et al (2006) Atg8L/Apg8L is the fourth mammalian modifier of mammalian Atg8 conjugation mediated by human Atg4B, Atg7 and Atg3. FEBS J 273(11):2553–2562
Li M et al (2011) Kinetics comparisons of mammalian Atg4 homologues indicate selective preferences toward diverse Atg8 substrates. J Biol Chem 286(9):7327–7338
Mizushima N et al (1998) A protein conjugation system essential for autophagy. Nature 395(6700):395–398
Tanida I et al (1999) Apg7p/Cvt2p: A novel protein-activating enzyme essential for autophagy. Mol Biol Cell 10(5):1367–1379
Mizushima N, Noda T, Ohsumi Y (1999) Apg16p is required for the function of the Apg12p-Apg5p conjugate in the yeast autophagy pathway. EMBO J 18(14):3888–3896
Lynch-Day MA et al (2010) Trs85 directs a Ypt1 GEF, TRAPPIII, to the phagophore to promote autophagy. Proc Natl Acad Sci USA 107(17):7811–7816
Balderhaar HJ et al (2010) The Rab GTPase Ypt7 is linked to retromer-mediated receptor recycling and fusion at the yeast late endosome. J Cell Sci 123(Pt 23):4085–4094
Degtyarev M et al (2008) Akt inhibition promotes autophagy and sensitizes PTEN-null tumors to lysosomotropic agents. J Cell Biol 183(1):101–116
Bellodi C et al (2009) Targeting autophagy potentiates tyrosine kinase inhibitor-induced cell death in Philadelphia chromosome-positive cells, including primary CML stem cells. J Clin Invest 119(5):1109–1123
Gupta A et al (2010) Autophagy inhibition and antimalarials promote cell death in gastrointestinal stromal tumor (GIST). Proc Natl Acad Sci USA 107(32):14333–14338
Ding WX et al (2009) Oncogenic transformation confers a selective susceptibility to the combined suppression of the proteasome and autophagy. Mol Cancer Ther 8(7):2036–2045
Rouschop KM et al (2009) Autophagy is required during cycling hypoxia to lower production of reactive oxygen species. Radiother Oncol 92(3):411–416
Fan QW et al (2010) Akt and autophagy cooperate to promote survival of drug-resistant glioma. Sci Signal 3(147):81
Pan Y et al (2011) Targeting autophagy augments in vitro and in vivo antimyeloma activity of DNA-damaging chemotherapy. Clin Cancer Res 17(10):3248–3258
Sheen JH et al (2011) Defective regulation of autophagy upon leucine deprivation reveals a targetable liability of human melanoma cells in vitro and in vivo. Cancer Cell 19(5):613–628
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Rangwala, R., Amaravadi, R. (2013). The Role of Autophagy in Drug Resistance and Potential for Therapeutic Targeting. In: Johnson, D. (eds) Cell Death Signaling in Cancer Biology and Treatment. Cell Death in Biology and Diseases. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4614-5847-0_4
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5847-0_4
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4614-5846-3
Online ISBN: 978-1-4614-5847-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)