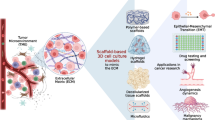
Abstract
Classic preclinical investigations on the mechanisms and effects of photodynamic therapy (PDT) are typically performed in two-dimensional cell cultures that have some, albeit limited, relevance to cancer biology. Bioengineered three-dimensional (3D) culture models of cancer are gaining traction in translational oncology as microtumors recapitulate the tumor architectures and cellular heterogeneity more faithfully than conventional 2D cultures. These 3D models bridge a gap between highly relevant but low-throughput in vivo animal models and high-throughput two-dimensional cultures with low clinical relevance, and thus hold promise as preclinical testing platforms in PDT research. Here, we discuss the potential applications of organotypic cancer models for PDT research and provide two well-established methodologies for generating 3D cultures of cancer: a liquid-suspended spheroid model and an adherent microtumor culture model grown on extracellular matrix scaffolds. Particular emphasis is given to harvesting the cultures for the purpose of immunoblotting and flow cytometry.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Sachs N, Clevers H (2014) Organoid cultures for the analysis of cancer phenotypes. Curr Opin Genet Dev 24:68–73
Tanner K, Gottesman MM (2015) Beyond 3D culture models of cancer. Sci Transl Med 7:283ps9
Pampaloni F, Reynaud EG, Stelzer EHK (2007) The third dimension bridges the gap between cell culture and live tissue. Nat Rev Mol Cell Biol 8:839–845
Clevers H (2016) Modeling development and disease with organoids. Cell 165:1586–1597
Rizvi I, Gurkan UA, Tasoglu S, Alagic N, Celli JP, Mensah LB, Mai Z, Demirci U, Hasan T (2013) Flow induces epithelial-mesenchymal transition, cellular heterogeneity and biomarker modulation in 3D ovarian cancer nodules. Proc Natl Acad Sci U S A 110:E1974–E1983
Li X, Nadauld L, Ootani A, Corney DC, Pai RK, Gevaert O, Cantrell MA, Rack PG, Neal JT, Chan CW-M et al (2014) Oncogenic transformation of diverse gastrointestinal tissues in primary organoid culture. Nat Med 20:769–777
Lee GY, Kenny PA, Lee EH, Bissell MJ (2007) Three-dimensional culture models of normal and malignant breast epithelial cells. Nat Methods 4:359–365
Boj SF, Hwang C-I, Baker LA, Chio IIC, Engle DD, Corbo V, Jager M, Ponz-Sarvise M, Tiriac H, Spector MS et al (2015) Organoid models of human and mouse ductal pancreatic cancer. Cell 160:324–338
Sachs N, de Ligt J, Kopper O, Gogola E, Bounova G, Weeber F, Balgobind AV, Wind K, Gracanin A, Begthel H et al (2018) A living biobank of breast cancer organoids captures disease heterogeneity. Cell 172(1–2):373–386.e10
van de Wetering M, Francies HE, Francis JM, Bounova G, Iorio F, Pronk A, van Houdt W, van Gorp J, Taylor-Weiner A, Kester L et al (2015) Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 161:933–945
Vinci M, Gowan S, Boxall F, Patterson L, Zimmermann M, Court W, Lomas C, Mendiola M, Hardisson D, Eccles SA (2012) Advances in establishment and analysis of three-dimensional tumor spheroid-based functional assays for target validation and drug evaluation. BMC Biol 10:29
Weeber F, Ooft SN, Dijkstra KK, Voest EE (2017) Tumor organoids as a pre-clinical cancer model for drug discovery. Cell Chem Biol 24:1092–1100
Celli JP, Rizvi I, Evans CL, Abu-Yousif AO, Hasan T (2010) Quantitative imaging reveals heterogeneous growth dynamics and treatment-dependent residual tumor distributions in a three-dimensional ovarian cancer model. J Biomed Opt 15:051603
Celli JP, Rizvi I, Blanden AR, Massodi I, Glidden MD, Pogue BW, Hasan T (2014) An imaging-based platform for high-content, quantitative evaluation of therapeutic response in 3D tumour models. Sci Rep 4:3751
Bulin A-L, Broekgaarden M, Hasan T (2017) Comprehensive high-throughput image analysis for therapeutic efficacy of architecturally complex heterotypic organoids. Sci Rep 7:16445
Rizvi I, Anbil S, Alagic N, Celli JP, Zheng LZ, Palanisami A, Glidden MD, Pogue BW, Hasan T (2013) PDT dose parameters impact tumoricidal durability and cell death pathways in a 3D ovarian cancer model. Photochem Photobiol 89:942–952
Anbil S, Rizvi I, Celli JP, Alagic N, Pogue BW, Hasan T (2013) Impact of treatment response metrics on photodynamic therapy planning and outcomes in a three-dimensional model of ovarian cancer. J Biomed Opt 18:098004
Glidden MD, Celli JP, Massodi I, Rizvi I, Pogue BW, Hasan T (2012) Image-based quantification of benzoporphyrin derivative uptake, localization, and Photobleaching in 3D tumor models, for optimization of PDT parameters. Theranostics 2:827–839
Kucinska M, Murias M, Nowak-Sliwinska P (2017) Beyond mouse cancer models: three-dimensional human-relevant in vitro and non-mammalian in vivo models for photodynamic therapy. Mutat Res 773:242–262
Chudy M, Tokarska K, Jastrzębska E, Bułka M, Drozdek S, Lamch Ł, Wilk KA, Brzózka Z (2018) Lab-on-a-chip systems for photodynamic therapy investigations. Biosens Bioelectron 101:37–51
Rahmanzadeh R, Rai P, Celli JP, Rizvi I, Baron-Lühr B, Gerdes J, Hasan T (2010) Ki-67 as a molecular target for therapy in an in vitro three-dimensional model for ovarian cancer. Cancer Res 70:9234–9242
Rizvi I, Celli JP, Evans CL, Abu-Yousif AO, Muzikansky A, Pogue BW, Finkelstein D, Hasan T (2010) Synergistic enhancement of carboplatin efficacy with photodynamic therapy in a three-dimensional model for micrometastatic ovarian cancer. Cancer Res 70:9319–9328
Cramer GM, Jones DP, El-Hamidi H, Celli JP (2017) ECM composition and rheology regulate growth, motility, and response to photodynamic therapy in 3D models of pancreatic ductal adenocarcinoma. Mol Cancer Res 15:15–25
Broekgaarden M, Rizvi I, Bulin A-L, Petrovic L, Goldschmidt R, Celli JP, Hasan T (2018) Neoadjuvant photodynamic therapy augments immediate and prolonged oxaliplatin efficacy in metastatic pancreatic cancer organoids. Oncotarget 9:13009–13022
Broekgaarden M, Anbil S, Bulin A-L, Obaid G, Mai Z, Baglo Y, Rizvi I, Hasan T (2019) Modulation of redox metabolism negates cancer-associated fibroblasts-induced treatment resistance in a heterotypic 3D culture platform of pancreatic cancer. Biomaterials 222:119421
Broekgaarden M, Bulin A-L, Frederick J, Mai Z, Hasan T (2019) Tracking photodynamic- and chemotherapy-induced redox state perturbations in 3D culture models of pancreatic cancer: a tool for identifying therapy-induced metabolic changes. J Clin Med 8:1399
Bulin A-L, Broekgaarden M, Simeone D, Hasan T (2019) Low dose photodynamic therapy harmonizes with radiation therapy to induce beneficial effects on pancreatic heterocellular spheroids. Oncotarget 10:2625–2643
Mohammad-Hadi L, MacRobert AJ, Loizidou M, Yaghini E (2018) Photodynamic therapy in 3D cancer models and the utilisation of nanodelivery systems. Nanoscale 10:1570–1581
Klein OJ, Yuan H, Nowell NH, Kaittanis C, Josephson L, Evans CL (2017) An integrin-targeted, highly diffusive construct for photodynamic therapy. Sci Rep 7:13375
Kumari P, Jain S, Ghosh B, Zorin V, Biswas S (2017) Polylactide-based block copolymeric micelles loaded with chlorin e6 for photodynamic therapy: in vitro evaluation in monolayer and 3D spheroid models. Mol Pharm 14:3789–3800
Flak D, Yate L, Nowaczyk G, Jurga S (2017) Hybrid ZnPc@TiO2 nanostructures for targeted photodynamic therapy, bioimaging and doxorubicin delivery. Mater Sci Eng C Mater Biol Appl 78:1072–1085
Chiarante N, García Vior MC, Awruch J, Marino J, Roguin LP (2017) Phototoxic action of a zinc(II) phthalocyanine encapsulated into poloxamine polymeric micelles in 2D and 3D colon carcinoma cell cultures. J Photochem Photobiol B 170:140–151
Obaid G, Bano S, Mallidi S, Broekgaarden M, Kuriakose J, Silber Z, Bulin A-L, Wang Y, Mai Z, ** W et al (2019) Impacting pancreatic cancer therapy in heterotypic in vitro organoids and in vivo tumors with specificity-tuned, NIR-activable photoimmunonanoconjugates: towards conquering desmoplasia? Nano Lett 19:7375–7387
Obaid G, Broekgaarden M, Bulin A-L, Huang H-C, Kuriakose J, Liu J, Hasan T (2016) Photonanomedicine: a convergence of photodynamic therapy and nanotechnology. Nanoscale 8:12471–12503
Celli JP (2012) Stromal interactions as regulators of tumor growth and therapeutic response: a potential target for photodynamic therapy? Isr J Chem 52:757–766
Broekgaarden M, Rizvi I, Bulin A-L, Petrovic L, Goldschmidt R, Celli JP, Hasan T, Broekgaarden M, Rizvi I, Bulin A-L et al (2018) Neoadjuvant photodynamic therapy augments immediate and prolonged oxaliplatin efficacy in metastatic pancreatic cancer organoids. Oncotarget 9(16):13009–13022
Molla A, Couvet M, Coll J-L (2017) Unsuccessful mitosis in multicellular tumour spheroids. Oncotarget 8:28769–28784
Zanoni M, Piccinini F, Arienti C, Zamagni A, Santi S, Polico R, Bevilacqua A, Tesei A (2016) 3D tumor spheroid models for in vitro therapeutic screening: a systematic approach to enhance the biological relevance of data obtained. Sci Rep 6:19103
Bonnier F, Keating ME, Wróbel TP, Majzner K, Baranska M, Garcia-Munoz A, Blanco A, Byrne HJ (2015) Cell viability assessment using the Alamar blue assay: a comparison of 2D and 3D cell culture models. Toxicol In Vitro 29:124–131
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Broekgaarden, M., Coll, JL. (2022). Microtumor Models as a Preclinical Investigational Platform for Photodynamic Therapy. In: Broekgaarden, M., Zhang, H., Korbelik, M., Hamblin, M.R., Heger, M. (eds) Photodynamic Therapy. Methods in Molecular Biology, vol 2451. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2099-1_3
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2099-1_3
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2098-4
Online ISBN: 978-1-0716-2099-1
eBook Packages: Springer Protocols