Abstract
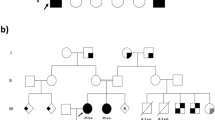
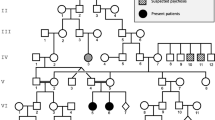
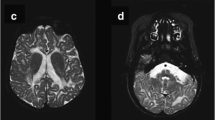
Krabbe disease is a lysosomal storage disease caused by galactosylceramidase deficiency, resulting in neurodegeneration with a rapid clinical downhill course within the first months of life in the classic infantile form. This process may be triggered by the accumulation of galactosylceramide (GalCer) in nervous tissues. Both the enzyme galactosylceramidase and its in vivo activator molecule, saposin A, are essential during GalCer degradation. A clinical manifestation almost identical to Krabbe disease is observed when, instead of the galactosylceramidase protein, the saposin A molecule is defective. Saposin A results from posttranslational processing of the precursor molecule, prosaposin, encoded by the PSAP gene. Clinical and neuroimaging findings in a 7-month-old child strongly suggested Krabbe disease, but this condition was excluded by enzymatic and genetic testing. However, at whole exome sequencing, the previously undescribed homozygous, obviously pathogenic PSAP gene NM_002778.3:c.209T>G(p.Val70Gly) variant was determined in the saposin A domain of the PSAP gene. Fibroblast studies showed GalCer accumulation and the activation of autophagy for the first time in a case of human saposin A deficiency. Our patient represents the second known case in the literature and provides new information concerning the pathophysiology of saposin A deficiency and its intralysosomal effects.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aflaki E, Moaven N, Borger DK, Lopez G, Westbroek W, Chae JJ et al (2016) Lysosomal storage and impaired autophagy lead to inflammasome activation in Gaucher macrophages. Aging Cell 15(1):77–88. https://doi.org/10.1111/acel.12409
Bradova V, Smid F, Ulrich-Bott B, Roggendorf W, Paton BC, Harzer K (1993) Prosaposin deficiency: further characterization of the sphingolipid activator protein-deficient sibs. Multiple glycolipid elevations (including lactosylceramidosis), partial enzyme deficiencies and ultrastructure of the skin in this generalized sphingolipid storage disease. Hum Genet 92(2):143–152
Deconinck N, Messaaoui A, Ziereisen F, Kadhim H, Sznajer Y, Pelc K et al (2008) Metachromatic leukodystrophy without arylsulfatase A deficiency: a new case of saposin-B deficiency. Eur J Paediatr Neurol 12(1):46–50. https://doi.org/10.1016/j.ejpn.2007.05.004
Filimonenko M, Stuffers S, Raiborg C, Yamamoto A, Malerod L, Fisher EM et al (2007) Functional multivesicular bodies are required for autophagic clearance of protein aggregates associated with neurodegenerative disease. J Cell Biol 179(3):485–500. https://doi.org/10.1083/jcb.200702115
Harzer K, Paton BC, Christomanou H, Chatelut M, Levade T, Hiraiwa M, O'Brien JS (1997) Saposins (sap) A and C activate the degradation of galactosylceramide in living cells. FEBS Lett 417(3):270–274
Harzer K, Knoblich R, Rolfs A, Bauer P, Eggers J (2002) Residual galactosylsphingosine (psychosine) beta-galactosidase activities and associated GALC mutations in late and very late onset Krabbe disease. Clin Chim Acta 317(1–2):77–84
Kishimoto Y, Hiraiwa M, O'Brien JS (1992) Saposins: structure, function, distribution, and molecular genetics. J Lipid Res 33(9):1255–1267
Kolter T, Sandhoff K (2005) Principles of lysosomal membrane digestion: stimulation of sphingolipid degradation by sphingolipid activator proteins and anionic lysosomal lipids. Annu Rev Cell Dev Biol 21:81–103. https://doi.org/10.1146/annurev.cellbio.21.122303.120013
Lieberman AP, Puertollano R, Raben N, Slaugenhaupt S, Walkley SU, Ballabio A (2012) Autophagy in lysosomal storage disorders. Autophagy 8(5):719–730. https://doi.org/10.4161/auto.19469
Lim SM, Choi BO, Oh SI, Choi WJ, Oh KW, Nahm M et al (2016) Patient fibroblasts-derived induced neurons demonstrate autonomous neuronal defects in adult-onset Krabbe disease. Oncotarget 7(46):74496–74509. https://doi.org/10.18632/oncotarget.12812
Matsuda J, Vanier MT, Saito Y, Tohyama J, Suzuki K, Suzuki K (2001) A mutation in the saposin A domain of the sphingolipid activator protein (prosaposin) gene results in a late-onset, chronic form of globoid cell leukodystrophy in the mouse. Hum Mol Genet 10(11):1191–1199
Morimoto S, Martin BM, Yamamoto Y, Kretz KA, O'Brien JS, Kishimoto Y (1989) Saposin A: second cerebrosidase activator protein. Proc Natl Acad Sci U S A 86(9):3389–3393
Orsini JJ, Martin MM, Showers AL, Bodamer OA, Zhang XK, Gelb MH, Caggana M (2012) Lysosomal storage disorder 4+1 multiplex assay for newborn screening using tandem mass spectrometry: application to a small-scale population study for five lysosomal storage disorders. Clin Chim Acta 413(15–16):1270–1273. https://doi.org/10.1016/j.cca.2012.04.012
Palmieri M, Impey S, Kang H, di Ronza A, Pelz C, Sardiello M, Ballabio A (2011) Characterization of the CLEAR network reveals an integrated control of cellular clearance pathways. Hum Mol Genet 20(19):3852–3866. https://doi.org/10.1093/hmg/ddr306
Pankiv S, Clausen TH, Lamark T, Brech A, Bruun JA, Outzen H et al (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282(33):24131–24145. https://doi.org/10.1074/jbc.M702824200
Ribbens JJ, Moser AB, Hubbard WC, Bongarzone ER, Maegawa GH (2014) Characterization and application of a disease-cell model for a neurodegenerative lysosomal disease. Mol Genet Metab 111(2):172–183. https://doi.org/10.1016/j.ymgme.2013.09.011
Sandhoff K (2016) Neuronal sphingolipidoses: membrane lipids and sphingolipid activator proteins regulate lysosomal sphingolipid catabolism. Biochimie 130:146–151. https://doi.org/10.1016/j.biochi.2016.05.004
Sandhoff R, Hepbildikler ST, Jennemann R, Geyer R, Gieselmann V, Proia RL et al (2002) Kidney sulfatides in mouse models of inherited glycosphingolipid disorders: determination by nano-electrospray ionization tandem mass spectrometry. J Biol Chem 277(23):20386–20398. https://doi.org/10.1074/jbc.M110641200
Seranova E, Connolly KJ, Zatyka M, Rosenstock TR, Barrett T, Tuxworth RI, Sarkar S (2017) Dysregulation of autophagy as a common mechanism in lysosomal storage diseases. Essays Biochem 61(6):733–749. https://doi.org/10.1042/ebc20170055
Settembre C, Fraldi A, Jahreiss L, Spampanato C, Venturi C, Medina D et al (2008) A block of autophagy in lysosomal storage disorders. Hum Mol Genet 17(1):119–129. https://doi.org/10.1093/hmg/ddm289
Spiegel R, Bach G, Sury V, Mengistu G, Meidan B, Shalev S et al (2005) A mutation in the saposin A coding region of the prosaposin gene in an infant presenting as Krabbe disease: first report of saposin A deficiency in humans. Mol Genet Metab 84(2):160–166
Sun Y, Grabowski GA (2013) Altered autophagy in the mice with a deficiency of saposin A and saposin B. Autophagy 9(7):1115–1116. https://doi.org/10.4161/auto.24919
Sun Y, Qi X, Grabowski GA (2003) Saposin C is required for normal resistance of acid beta-glucosidase to proteolytic degradation. J Biol Chem 278(34):31918–31923. https://doi.org/10.1074/jbc.M302752200
Sun Y, Zamzow M, Ran H, Zhang W, Quinn B, Barnes S et al (2013) Tissue-specific effects of saposin A and saposin B on glycosphingolipid degradation in mutant mice. Hum Mol Genet 22(12):2435–2450. https://doi.org/10.1093/hmg/ddt096
Ward C, Martinez-Lopez N, Otten EG, Carroll B, Maetzel D, Singh R et al (2016) Autophagy, lipophagy and lysosomal lipid storage disorders. Biochim Biophys Acta 1861(4):269–284. https://doi.org/10.1016/j.bbalip.2016.01.006
Acknowledgments
We thank Centogene AG for whole exome sequencing analyses.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Additional information
Communicated by: John Christodoulou, MB BS PhD FRACP FRCPA
Appendices
Conflict of Interest
No conflict of interest was declared by the authors.
Financial Disclosure
The authors declared that this study has received no financial support.
Ethics
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study.
Rights and permissions
Copyright information
© 2018 Society for the Study of Inborn Errors of Metabolism (SSIEM)
About this chapter
Cite this chapter
Kose, M. et al. (2018). The Second Case of Saposin A Deficiency and Altered Autophagy. In: Morava, E., Baumgartner, M., Patterson, M., Rahman, S., Zschocke, J., Peters, V. (eds) JIMD Reports, Volume 44. JIMD Reports, vol 44. Springer, Berlin, Heidelberg. https://doi.org/10.1007/8904_2018_114
Download citation
DOI: https://doi.org/10.1007/8904_2018_114
Received:
Revised:
Accepted:
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-58616-7
Online ISBN: 978-3-662-58617-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)