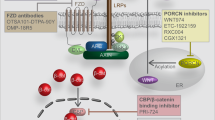
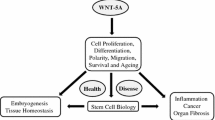
Abstract
WNT/β-catenin signaling plays fundamental roles in numerous developmental processes and in adult tissue homeostasis and repair after injury, by controlling cellular self-renewal, activation, division, differentiation, movement, genetic stability, and apoptosis. As such, it comes as no surprise that dysregulation of WNT/β-catenin signaling is associated with various diseases, including cancer, fibrosis, neurodegeneration, etc. Although multiple agents that specifically target the WNT/β-catenin signaling pathway have been studied preclinically and a number have entered clinical trials, none has been approved by the FDA to date. In this chapter, we provide our insights as to the reason(s) it has been so difficult to safely pharmacologically target the WNT/β-catenin signaling pathway and discuss the significant efforts undertaken towards this goal.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- 17-AAG:
-
17-Allylamino-geldanamycin
- ALDH1:
-
Aldehyde dehydrogenase 1 family member A1
- AML:
-
Acute myeloid leukemia
- APC:
-
Adenomatous polyposis coli
- ApcMin:
-
Mutant allele Min (multiple intestinal neoplasia) of the murine Apc locus which predisposes to intestinal adenoma formation
- Arg1:
-
Arginase 1
- Arg2:
-
Arginase 2
- ASCO:
-
American Society of Clinical Oncology
- BCL-2:
-
BCL2 apoptosis regulator
- BCR–ABL:
-
Mutation that is formed by the combination of two genes, known as BCR (BCR activator of RhoGEF and GTPase) and ABL (ABL proto-oncogene 1, non-receptor tyrosine kinase)
- BIRC5:
-
Baculoviral IAP repeat containing 5 (also known as survivin)
- BMP:
-
Bone morphogenetic protein
- BRAF:
-
B-Raf proto-oncogene, serine/threonine kinase
- CAR-T:
-
Chimeric antigen receptor (CAR) T cells
- CBP:
-
CREB-binding protein
- Ccnd1:
-
Cyclin D1
- Ccnd3:
-
Cyclin D3
- CD133:
-
Prominin 1
- CD27:
-
CD27 molecule
- CD40:
-
CD40 molecule
- CD44:
-
CD44 molecule (Indian blood group)
- CD8A:
-
CD8a molecule
- CDK1:
-
Cyclin dependent kinase 1
- CDK2:
-
Cyclin dependent kinase 2
- CDK4:
-
Cyclin dependent kinase 4
- CDK6:
-
Cyclin dependent kinase 6
- CK1α:
-
Casein kinase 1 alpha
- CK20:
-
Keratin 20
- CKO:
-
Conditional knockout
- CLK:
-
CDC-like kinase
- CLK2:
-
CDC like kinase 2
- CLK3:
-
CDC like kinase 3
- c-Myc:
-
MYC proto-oncogene, bHLH transcription factor
- COMP:
-
Cartilage oligomeric matrix protein
- COX:
-
Cyclooxygenase
- CRC:
-
Colorectal cancer
- Cre:
-
Cre recombinase
- CreERT2:
-
Cre recombinase (Cre) fused to a mutant estrogen ligand-binding domain (ERT2) that requires the presence of tamoxifen for activity
- CSC:
-
Cancer stem cells
- CTLA4:
-
Cytotoxic T-lymphocyte associated protein 4
- Cxcl1:
-
C-X-C motif chemokine ligand 1
- Cxcl2:
-
C-X-C motif chemokine ligand 2
- CYCA2:
-
Cyclin A2
- DAB2:
-
Disabled homolog 2
- DKK1:
-
Dickkopf WNT signaling pathway inhibitor 1
- DVL:
-
Dishevelled segment polarity protein
- DYRK1A:
-
Dual specificity tyrosine phosphorylation-regulated kinase 1A
- EC50:
-
Concentration of a drug that gives half-maximal response
- EGCG:
-
Epigallocatechin 3 gallate
- ELISA:
-
Enzyme-linked immunosorbent assay
- EMT:
-
Epithelial-mesenchymal transition
- ETS:
-
Erythroblast transformation-specific
- FDA:
-
US Food and Drug Administration
- FGF:
-
Fibroblast growth factor
- fl/fl:
-
Homozygous for allele floxed (flanked by loxP sites)
- FLT3:
-
Fms related receptor tyrosine kinase 3
- Foxp3:
-
Forkhead box P3
- FZD:
-
Frizzled class receptor
- FZD1:
-
Frizzled class receptor 1
- FZD2:
-
Frizzled class receptor 2
- FZD5:
-
Frizzled class receptor 5
- FZD7:
-
Frizzled class receptor 7
- FZD8:
-
Frizzled class receptor 8
- GSK3:
-
Glycogen synthase kinase 3
- GSK3β:
-
Glycogen synthase kinase 3 beta
- hDVL1:
-
Human dishevelled segment polarity protein 1
- HGF:
-
Hepatocyte growth factor
- HGK:
-
Hematopoietic progenitor kinase/germinal center kinase-like kinase (also known as MAP4K4)
- hMSC:
-
Human mesenchymal stem cells
- HSC:
-
Hepatic stellate cells
- IC50:
-
Half-maximal inhibitory concentration
- iCRT:
-
Inhibitor of catenin responsive transcription (CRT)
- Ifng:
-
Interferon gamma
- IL10:
-
Interleukin 10
- IWP:
-
Inhibitor of WNT production
- IWR:
-
Inhibitor of WNT response
- JAK:
-
Janus kinase
- JAK3:
-
Janus kinase 3
- Kat3:
-
Lysine acetyltransferases type 3
- KD:
-
Dissociation constant
- Lef:
-
Lymphoid enhancer factor
- Lgr4:
-
Leucine rich repeat containing G protein-coupled receptor 4
- Lgr5:
-
Leucine rich repeat containing G protein-coupled receptor 5
- Lgr6:
-
Leucine rich repeat containing G protein-coupled receptor 6
- Lkb1:
-
Liver kinase B1 (also known as Stk11)
- LRP5:
-
LDL receptor-related protein 5
- LRP6:
-
LDL receptor-related protein 6
- MAPK:
-
Mitogen-activated protein kinase
- MET:
-
MET proto-oncogene, receptor tyrosine kinase
- MMTV:
-
Mouse mammary tumor virus
- NCK:
-
NCK adaptor protein
- NF-κB:
-
Nuclear factor-κB
- NMR:
-
Nuclear magnetic resonance spectroscopy
- Nrp1:
-
Neuropilin 1
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs
- p300:
-
E1A binding protein p300
- p53:
-
Tumor protein p53
- PARP:
-
Poly(ADP-ribose) polymerase
- PARP 1:
-
Poly(ADP-ribose) polymerase 1
- PARP 2:
-
Poly(ADP-ribose) polymerase 2
- PD-1:
-
Programmed cell death 1 (also known as PDCD1)
- PDGFRα:
-
Platelet derived growth factor receptor alpha
- PD-L1:
-
Programmed cell death 1 ligand 1
- PDZ:
-
PDZ domain
- PGE2:
-
Prostaglandin E2
- PI3K:
-
Phosphatidylinositol 3-kinase
- Porcn:
-
Porcupine O-acyltransferase
- PTEN:
-
Phosphatase and tensin homolog
- RAR:
-
Retinoic acid receptor
- Rnf43:
-
Ring finger protein 43
- RSPO:
-
R-spondin
- RXR:
-
Retinoid X receptor
- SAM68:
-
SRC-associated in mitosis 68 kDa protein (also known as KHDRBS1)
- SRSF:
-
Serine and arginine rich splicing factor
- SSC:
-
Somatic stem cells
- Stat:
-
Signal transducer and activator of transcription
- Tcf:
-
T-cell factor
- TCF1:
-
T-cell factor 1
- TCF4:
-
T-cell factor 4
- Tcf7:
-
T-cell factor 7
- TGFβ:
-
Transforming growth factor beta
- TGFβ1:
-
Transforming growth factor beta 1
- THBS1:
-
Thrombospondin 1
- Tmax:
-
The time at which the maximum blood concentration of a drug is achieved after the drug has been administered and before the administration of a second dose
- TNIK:
-
TRAF2 and NCK-interacting kinase
- TNKS1:
-
Tankyrase 1
- TNKS2:
-
Tankyrase 2
- TRAF2:
-
TNF receptor associated factor 2
- Tregs:
-
Regulatory T cells
- TRKA:
-
Tropomyosin receptor kinase A
- US:
-
United States
- VDR:
-
Vitamin D receptor
- WNT1:
-
Wnt family member 1
- WNT3A:
-
Wnt family member 3A
- αSMA:
-
Actin alpha 2, smooth muscle
- β-CTX:
-
Beta C-terminal telopeptide
References
Akcora B, Storm G, Bansal R (2018) Inhibition of canonical WNT signaling pathway by β-catenin/CBP inhibitor ICG-001 ameliorates liver fibrosis in vivo through suppression of stromal CXCL12. Biochim Biophys Acta Mol basis Dis 1864(3):804–818. https://doi.org/10.1016/j.bbadis.2017.12.001
Albers J, Keller J, Baranowsky A, Beil FT, Catala-Lehnen P, Schulze J et al (2013) Canonical Wnt signaling inhibits osteoclastogenesis independent of osteoprotegerin. J Cell Biol 200(4):537–549. https://doi.org/10.1083/jcb.201207142
Anastas JN, Moon RT (2013) WNT signalling pathways as therapeutic targets in cancer. Nat Rev Cancer 13(1):11–26. https://doi.org/10.1038/nrc3419
Antelmann H, Helmann JD (2011) Thiol-based redox switches and gene regulation. Antioxid Redox Signal 14(6):1049–1063. https://doi.org/10.1089/ars.2010.3400
Baker NE (1987) Molecular cloning of sequences from wingless, a segment polarity gene in Drosophila: the spatial distribution of a transcript in embryos. EMBO J 6(6):1765–1773
Baron JA, Cole BF, Sandler RS, Haile RW, Ahnen D, Bresalier R et al (2003) A randomized trial of aspirin to prevent colorectal adenomas. N Engl J Med 348(10):891–899. https://doi.org/10.1056/NEJMoa021735
Bayle ED, Svensson F, Atkinson BN, Steadman D, Willis NJ, Woodward HL et al (2021) Carboxylesterase notum is a druggable target to modulate Wnt signaling. J Med Chem 64(8):4289–4311. https://doi.org/10.1021/acs.jmedchem.0c01974
Boon EM, Keller JJ, Wormhoudt TA, Giardiello FM, Offerhaus GJ, van der Neut R et al (2004) Sulindac targets nuclear beta-catenin accumulation and Wnt signalling in adenomas of patients with familial adenomatous polyposis and in human colorectal cancer cell lines. Br J Cancer 90(1):224–229. https://doi.org/10.1038/sj.bjc.6601505
Boulter L, Guest RV, Kendall TJ, Wilson DH, Wojtacha D, Robson AJ et al (2015) WNT signaling drives cholangiocarcinoma growth and can be pharmacologically inhibited. J Clin Invest 125(3):1269–1285. https://doi.org/10.1172/JCI76452
Cao H, Wang C, Chen X, Hou J, **ang Z, Shen Y et al (2018) Inhibition of Wnt/β-catenin signaling suppresses myofibroblast differentiation of lung resident mesenchymal stem cells and pulmonary fibrosis. Sci Rep 8(1):13644. https://doi.org/10.1038/s41598-018-28968-9
Chan KC, Chan LS, Ip JC, Lo C, Yip TT, Ngan RK et al (2015) Therapeutic targeting of CBP/β-catenin signaling reduces cancer stem-like population and synergistically suppresses growth of EBV-positive nasopharyngeal carcinoma cells with cisplatin. Sci Rep 5:9979. https://doi.org/10.1038/srep09979
Chee YC, Pahnke J, Bunte R, Adsool VA, Madan B, Virshup DM (2018) Intrinsic xenobiotic resistance of the intestinal stem cell niche. Dev Cell 46(6):681–95.e5. https://doi.org/10.1016/j.devcel.2018.07.023
Chen Z, Venkatesan AM, Dehnhardt CM, Dos Santos O, Delos Santos E, Ayral-Kaloustian S et al (2009a) 2,4-Diamino-quinazolines as inhibitors of beta-catenin/Tcf-4 pathway: potential treatment for colorectal cancer. Bioorg Med Chem Lett 19(17):4980–4983. https://doi.org/10.1016/j.bmcl.2009.07.070
Chen B, Dodge ME, Tang W, Lu J, Ma Z, Fan CW et al (2009b) Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer. Nat Chem Biol 5(2):100–107. https://doi.org/10.1038/nchembio.137
Cheng Y, Phoon YP, ** X, Chong SY, Ip JC, Wong BW et al (2015) Wnt-C59 arrests stemness and suppresses growth of nasopharyngeal carcinoma in mice by inhibiting the Wnt pathway in the tumor microenvironment. Oncotarget 6(16):14428–14439. https://doi.org/10.18632/oncotarget.3982
Cicalese A, Bonizzi G, Pasi CE, Faretta M, Ronzoni S, Giulini B et al (2009) The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell 138(6):1083–1095. https://doi.org/10.1016/j.cell.2009.06.048
Coluccia AM, Vacca A, Duñach M, Mologni L, Redaelli S, Bustos VH et al (2007) Bcr-Abl stabilizes beta-catenin in chronic myeloid leukemia through its tyrosine phosphorylation. EMBO J 26(5):1456–1466. https://doi.org/10.1038/sj.emboj.7601485
de Lau WB, Snel B, Clevers HC (2012) The R-spondin protein family. Genome Biol 13(3):242. https://doi.org/10.1186/gb-2012-13-3-242
de Lau W, Peng WC, Gros P, Clevers H (2014) The R-spondin/Lgr5/Rnf43 module: regulator of Wnt signal strength. Genes Dev 28(4):305–316. https://doi.org/10.1101/gad.235473.113
Deshmukh V, Hu H, Barroga C, Bossard C, Kc S, Dellamary L et al (2018) A small-molecule inhibitor of the Wnt pathway (SM04690) as a potential disease modifying agent for the treatment of osteoarthritis of the knee. Osteoarthr Cartil 26(1):18–27. https://doi.org/10.1016/j.joca.2017.08.015
Deshmukh V, O’Green AL, Bossard C, Seo T, Lamangan L, Ibanez M et al (2019) Modulation of the Wnt pathway through inhibition of CLK2 and DYRK1A by lorecivivint as a novel, potentially disease-modifying approach for knee osteoarthritis treatment. Osteoarthr Cartil 27(9):1347–1360. https://doi.org/10.1016/j.joca.2019.05.006
Deshmukh V, Seo T, O’Green AL, Ibanez M, Hofilena B, Kc S et al (2020) SM04755, a small-molecule inhibitor of the Wnt pathway, as a potential topical treatment for tendinopathy. J Orthop Res. https://doi.org/10.1002/jor.24898
DeVito NC, Sturdivant M, Thievanthiran B, **ao C, Plebanek MP, Salama AKS et al (2021) Pharmacological Wnt ligand inhibition overcomes key tumor-mediated resistance pathways to anti-PD-1 immunotherapy. Cell Rep 35(5):109071. https://doi.org/10.1016/j.celrep.2021.109071
Dihlmann S, Siermann A, von Knebel Doeberitz M (2001) The nonsteroidal anti-inflammatory drugs aspirin and indomethacin attenuate beta-catenin/TCF-4 signaling. Oncogene 20(5):645–653. https://doi.org/10.1038/sj.onc.1204123
El-Khoueiry A, Ning Y, Yang D, Cole S, Kahn M et al (2013) A phase I first-in-human study of PRI-724 in patients (pts) with advanced solid tumors. J Clin Oncol Abstr 31(15):2501
Emami KH, Nguyen C, Ma H, Kim DH, Jeong KW, Eguchi M et al (2004) A small molecule inhibitor of beta-catenin/CREB-binding protein transcription [corrected]. Proc Natl Acad Sci U S A 101(34):12682–12687. https://doi.org/10.1073/pnas.0404875101
Fagotto F (2013) Looking beyond the Wnt pathway for the deep nature of β-catenin. EMBO Rep 14(5):422–433. https://doi.org/10.1038/embor.2013.45
Feng Y, Ren J, Gui Y, Wei W, Shu B, Lu Q et al (2018) Wnt/β-catenin-promoted macrophage alternative activation contributes to kidney fibrosis. J Am Soc Nephrol 29(1):182–193. https://doi.org/10.1681/ASN.2017040391
Fujii N, You L, Xu Z, Uematsu K, Shan J, He B et al (2007) An antagonist of dishevelled protein-protein interaction suppresses beta-catenin-dependent tumor cell growth. Cancer Res 67(2):573–579. https://doi.org/10.1158/0008-5472.CAN-06-2726
Funato Y, Michiue T, Asashima M, Miki H (2006) The thioredoxin-related redox-regulating protein nucleoredoxin inhibits Wnt-beta-catenin signalling through dishevelled. Nat Cell Biol 8(5):501–508. https://doi.org/10.1038/ncb1405
Funck-Brentano T, Nilsson KH, Brommage R, Henning P, Lerner UH, Koskela A et al (2018) Porcupine inhibitors impair trabecular and cortical bone mass and strength in mice. J Endocrinol 238(1):13–23. https://doi.org/10.1530/JOE-18-0153
Gabata R, Harada K, Mizutani Y, Ouchi H, Yoshimura K, Sato Y et al (2020) Anti-tumor activity of the small molecule inhibitor PRI-724 against β-catenin-activated hepatocellular carcinoma. Anticancer Res 40(9):5211–5219. https://doi.org/10.21873/anticanres.14524
Gang EJ, Hsieh YT, Pham J, Zhao Y, Nguyen C, Huantes S et al (2014) Small-molecule inhibition of CBP/catenin interactions eliminates drug-resistant clones in acute lymphoblastic leukemia. Oncogene 33(17):2169–2178. https://doi.org/10.1038/onc.2013.169
Giardiello FM, Hamilton SR, Krush AJ, Piantadosi S, Hylind LM, Celano P et al (1993) Treatment of colonic and rectal adenomas with sulindac in familial adenomatous polyposis. N Engl J Med 328(18):1313–1316. https://doi.org/10.1056/NEJM199305063281805
Goldsberry WN, Londoño A, Randall TD, Norian LA, Arend RC (2019) A review of the role of Wnt in cancer immunomodulation. Cancers (Basel) 11(6). https://doi.org/10.3390/cancers11060771
Gonsalves FC, Klein K, Carson BB, Katz S, Ekas LA, Evans S et al (2011) An RNAi-based chemical genetic screen identifies three small-molecule inhibitors of the Wnt/wingless signaling pathway. Proc Natl Acad Sci U S A 108(15):5954–5963. https://doi.org/10.1073/pnas.1017496108
Grandy D, Shan J, Zhang X, Rao S, Akunuru S, Li H et al (2009) Discovery and characterization of a small molecule inhibitor of the PDZ domain of dishevelled. J Biol Chem 284(24):16256–16263. https://doi.org/10.1074/jbc.M109.009647
Groden J, Thliveris A, Samowitz W, Carlson M, Gelbert L, Albertsen H et al (1991) Identification and characterization of the familial adenomatous polyposis coli gene. Cell 66(3):589–600. https://doi.org/10.1016/0092-8674(81)90021-0
Grösch S, Tegeder I, Niederberger E, Bräutigam L, Geisslinger G (2001) COX-2 independent induction of cell cycle arrest and apoptosis in colon cancer cells by the selective COX-2 inhibitor celecoxib. FASEB J 15(14):2742–2744. https://doi.org/10.1096/fj.01-0299fje
Grossmann TN, Yeh JT, Bowman BR, Chu Q, Moellering RE, Verdine GL (2012) Inhibition of oncogenic Wnt signaling through direct targeting of β-catenin. Proc Natl Acad Sci U S A 109(44):17942–17947. https://doi.org/10.1073/pnas.1208396109
Gurney A, Axelrod F, Bond CJ, Cain J, Chartier C, Donigan L et al (2012) Wnt pathway inhibition via the targeting of frizzled receptors results in decreased growth and tumorigenicity of human tumors. Proc Natl Acad Sci U S A 109(29):11717–11722. https://doi.org/10.1073/pnas.1120068109
Hahne G, Grossmann TN (2013) Direct targeting of β-catenin: inhibition of protein-protein interactions for the inactivation of Wnt signaling. Bioorg Med Chem 21(14):4020–4026. https://doi.org/10.1016/j.bmc.2013.02.050
Hao S, He W, Li Y, Ding H, Hou Y, Nie J et al (2011) Targeted inhibition of β-catenin/CBP signaling ameliorates renal interstitial fibrosis. J Am Soc Nephrol 22(9):1642–1653. https://doi.org/10.1681/ASN.2010101079
Hao HX, **e Y, Zhang Y, Charlat O, Oster E, Avello M et al (2012) ZNRF3 promotes Wnt receptor turnover in an R-spondin-sensitive manner. Nature 485(7397):195–200. https://doi.org/10.1038/nature11019
Hasegawa K, Yasuda SY, Teo JL, Nguyen C, McMillan M, Hsieh CL et al (2012) Wnt signaling orchestration with a small molecule DYRK inhibitor provides long-term xeno-free human pluripotent cell expansion. Stem Cells Transl Med 1(1):18–28. https://doi.org/10.5966/sctm.2011-0033
He K, Xu T, Xu Y, Ring A, Kahn M, Goldkorn A (2014) Cancer cells acquire a drug resistant, highly tumorigenic, cancer stem-like phenotype through modulation of the PI3K/Akt/β-catenin/CBP pathway. Int J Cancer 134(1):43–54. https://doi.org/10.1002/ijc.28341
Henderson WR, Chi EY, Ye X, Nguyen C, Tien YT, Zhou B et al (2010) Inhibition of Wnt/beta-catenin/CREB binding protein (CBP) signaling reverses pulmonary fibrosis. Proc Natl Acad Sci U S A 107(32):14309–14314. https://doi.org/10.1073/pnas.1001520107
Herr P, Hausmann G, Basler K (2012) WNT secretion and signalling in human disease. Trends Mol Med 18(8):483–493. https://doi.org/10.1016/j.molmed.2012.06.008
Hori K, Ajioka K, Goda N, Shindo A, Takagishi M, Tenno T et al (2018) Discovery of potent disheveled/Dvl inhibitors using virtual screening optimized with NMR-based docking performance index. Front Pharmacol 9:983. https://doi.org/10.3389/fphar.2018.00983
Hou YC, Chao YJ, Hsieh MH, Tung HL, Wang HC, Shan YS (2019) Low CD8+ T cell infiltration and high PD-L1 expression are associated with level of CD44+/CD133+ cancer stem cells and predict an unfavorable prognosis in pancreatic cancer. Cancers (Basel) 11(4). https://doi.org/10.3390/cancers11040541
Huang SM, Mishina YM, Liu S, Cheung A, Stegmeier F, Michaud GA et al (2009) Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature 461(7264):614–620. https://doi.org/10.1038/nature08356
Huang T, Li F, Cheng X, Wang J, Zhang W, Zhang B et al (2021) Wnt inhibition sensitizes PD-L1 blockade therapy by overcoming bone marrow-derived myofibroblasts-mediated immune resistance in tumors. Front Immunol 12:619209. https://doi.org/10.3389/fimmu.2021.619209
Im DU, Kim SC, Chau GC, Um SH (2019) Carbamazepine enhances adipogenesis by inhibiting Wnt/β-catenin expression. Cell 8(11). https://doi.org/10.3390/cells8111460
Jacob LS, Wu X, Dodge ME, Fan CW, Kulak O, Chen B et al (2011) Genome-wide RNAi screen reveals disease-associated genes that are common to Hedgehog and Wnt signaling. Sci Signal 4(157):ra4. https://doi.org/10.1126/scisignal.2001225
Jaiswal AS, Marlow BP, Gupta N, Narayan S (2002) Beta-catenin-mediated transactivation and cell-cell adhesion pathways are important in curcumin (diferuylmethane)-induced growth arrest and apoptosis in colon cancer cells. Oncogene 21(55):8414–8427. https://doi.org/10.1038/sj.onc.1205947
Jiang X, Hao HX, Growney JD, Woolfenden S, Bottiglio C, Ng N et al (2013) Inactivating mutations of RNF43 confer Wnt dependency in pancreatic ductal adenocarcinoma. Proc Natl Acad Sci U S A 110(31):12649–12654. https://doi.org/10.1073/pnas.1307218110
Jiang X, Mak PY, Mu H, Tao W, Mak DH, Kornblau S et al (2018) Disruption of Wnt/β-catenin exerts antileukemia activity and synergizes with FLT3 inhibition in. Clin Cancer Res 24(10):2417–2429. https://doi.org/10.1158/1078-0432.CCR-17-1556
Kahn M (2011) Symmetric division versus asymmetric division: a tale of two coactivators. Future Med Chem 3(14):1745–1763. https://doi.org/10.4155/fmc.11.126
Kahn M (2014) Can we safely target the WNT pathway? Nat Rev Drug Discov 13(7):513–532. https://doi.org/10.1038/nrd4233
Kasashima H, Duran A, Martinez-Ordoñez A, Nakanishi Y, Kinoshita H, Linares JF et al (2021) Stromal SOX2 upregulation promotes tumorigenesis through the generation of a SFRP1/2-expressing cancer-associated fibroblast population. Dev Cell 56(1):95–110.e10. https://doi.org/10.1016/j.devcel.2020.10.014
Kim J, Zhang X, Rieger-Christ KM, Summerhayes IC, Wazer DE, Paulson KE et al (2006) Suppression of Wnt signaling by the green tea compound (−)-epigallocatechin 3-gallate (EGCG) in invasive breast cancer cells. Requirement of the transcriptional repressor HBP1. J Biol Chem 281(16):10865–10875. https://doi.org/10.1074/jbc.M513378200
Kim YM, Gang EJ, Kahn M (2017) CBP/catenin antagonists: targeting LSCs’ Achilles heel. Exp Hematol. https://doi.org/10.1016/j.exphem.2017.04.010
Kimura K, Ikoma A, Shibakawa M, Shimoda S, Harada K, Saio M et al (2017) Safety, tolerability, and preliminary efficacy of the anti-fibrotic small molecule PRI-724, a CBP/β-catenin inhibitor, in patients with hepatitis C virus-related cirrhosis: a single-center, open-label, dose escalation phase 1 trial. EBioMedicine 23:79–87. https://doi.org/10.1016/j.ebiom.2017.08.016
Kinzler KW, Nilbert MC, Su LK, Vogelstein B, Bryan TM, Levy DB et al (1991) Identification of FAP locus genes from chromosome 5q21. Science 253(5020):661–665. https://doi.org/10.1126/science.1651562
Klein PS, Melton DA (1996) A molecular mechanism for the effect of lithium on development. Proc Natl Acad Sci U S A 93(16):8455–8459. https://doi.org/10.1073/pnas.93.16.8455
Komiya Y, Habas R (2008) Wnt signal transduction pathways. Organogenesis 4(2):68–75. https://doi.org/10.4161/org.4.2.5851
Koo BK, Spit M, Jordens I, Low TY, Stange DE, van de Wetering M et al (2012) Tumour suppressor RNF43 is a stem-cell E3 ligase that induces endocytosis of Wnt receptors. Nature 488(7413):665–669. https://doi.org/10.1038/nature11308
Kühl SJ, Kühl M (2013) On the role of Wnt/β-catenin signaling in stem cells. Biochim Biophys Acta 1830(2):2297–2306. https://doi.org/10.1016/j.bbagen.2012.08.010
Kung AL, Rebel VI, Bronson RT, Ch'ng LE, Sieff CA, Livingston DM et al (2000) Gene dose-dependent control of hematopoiesis and hematologic tumor suppression by CBP. Genes Dev 14(3):272–277
LaBarge MA (2010) The difficulty of targeting cancer stem cell niches. Clin Cancer Res 16(12):3121–3129. https://doi.org/10.1158/1078-0432.CCR-09-2933
Lafyatis R, Mantero JC, Gordon J, Kishore N, Carns M, Dittrich H et al (2017) Inhibition of β-catenin signaling in the skin rescues cutaneous adipogenesis in systemic sclerosis: a randomized, double-blind, placebo-controlled trial of C-82. J Invest Dermatol 137(12):2473–2483. https://doi.org/10.1016/j.jid.2017.06.032
Lai KKY, Nguyen C, Lee KS, Lee A, Lin DP, Teo JL et al (2019) Convergence of canonical and non-canonical Wnt signal: differential Kat3 coactivator usage. Curr Mol Pharmacol 12(3):167–183. https://doi.org/10.2174/1874467212666190304121131
Lai KKY, Hu X, Chosa K, Nguyen C, Lin DP, Lai KK et al (2021) p300 serine 89: a critical signaling integrator and its effects on intestinal homeostasis and repair. Cancers (Basel) 13(6). https://doi.org/10.3390/cancers13061288
Lau T, Chan E, Callow M, Waaler J, Boggs J, Blake RA et al (2013) A novel tankyrase small-molecule inhibitor suppresses APC mutation-driven colorectal tumor growth. Cancer Res 73(10):3132–3144. https://doi.org/10.1158/0008-5472.CAN-12-4562
Le NH, Franken P, Fodde R (2008) Tumour-stroma interactions in colorectal cancer: converging on beta-catenin activation and cancer stemness. Br J Cancer 98(12):1886–1893. https://doi.org/10.1038/sj.bjc.6604401
Lee JY, Nakada D, Yilmaz OH, Tothova Z, Joseph NM, Lim MS et al (2010) mTOR activation induces tumor suppressors that inhibit leukemogenesis and deplete hematopoietic stem cells after Pten deletion. Cell Stem Cell 7(5):593–605. https://doi.org/10.1016/j.stem.2010.09.015
Lee HJ, Bao J, Miller A, Zhang C, Wu J, Baday YC et al (2015) Structure-based discovery of novel small molecule Wnt signaling inhibitors by targeting the cysteine-rich domain of frizzled. J Biol Chem 290(51):30596–30606. https://doi.org/10.1074/jbc.M115.673202
Lee RS, Zhang L, Berger A, Lawrence MG, Song J, Niranjan B et al (2019) Characterization of the ERG-regulated kinome in prostate cancer identifies TNIK as a potential therapeutic target. Neoplasia 21(4):389–400. https://doi.org/10.1016/j.neo.2019.02.005
Lee JH, Faderl S, Pagel JM, Jung CW, Yoon SS, Pardanani AD et al (2020) Phase 1 study of CWP232291 in patients with relapsed or refractory acute myeloid leukemia and myelodysplastic syndrome. Blood Adv 4(9):2032–2043. https://doi.org/10.1182/bloodadvances.2019000757
Lehtiö L, Chi NW, Krauss S (2013) Tankyrases as drug targets. FEBS J 280(15):3576–3593. https://doi.org/10.1111/febs.12320
Lepourcelet M, Chen YN, France DS, Wang H, Crews P, Petersen F et al (2004) Small-molecule antagonists of the oncogenic Tcf/beta-catenin protein complex. Cancer Cell 5(1):91–102. https://doi.org/10.1016/s1535-6108(03)00334-9
Li X, Placencio V, Iturregui JM, Uwamariya C, Sharif-Afshar AR, Koyama T et al (2008) Prostate tumor progression is mediated by a paracrine TGF-beta/Wnt3a signaling axis. Oncogene 27(56):7118–7130. https://doi.org/10.1038/onc.2008.293
Liu J, Pan S, Hsieh MH, Ng N, Sun F, Wang T et al (2013) Targeting Wnt-driven cancer through the inhibition of porcupine by LGK974. Proc Natl Acad Sci U S A 110(50):20224–20229. https://doi.org/10.1073/pnas.1314239110
Logan CY, Nusse R (2004) The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 20:781–810. https://doi.org/10.1146/annurev.cellbio.20.010403.113126
Lukaszewicz AI, Nguyen C, Melendez E, Lin DP, Teo JL, Lai KKY et al (2019) The mode of stem cell division is dependent on the differential interaction of β-catenin with the Kat3 coactivators CBP or p300. Cancers (Basel) 11(7). https://doi.org/10.3390/cancers11070962
Luke JJ, Bao R, Sweis RF, Spranger S, Gajewski TF (2019) WNT/β-catenin pathway activation correlates with immune exclusion across human cancers. Clin Cancer Res 25(10):3074–3083. https://doi.org/10.1158/1078-0432.CCR-18-1942
Madan B, Patel MB, Zhang J, Bunte RM, Rudemiller NP, Griffiths R et al (2016a) Experimental inhibition of porcupine-mediated Wnt O-acylation attenuates kidney fibrosis. Kidney Int 89(5):1062–1074. https://doi.org/10.1016/j.kint.2016.01.017
Madan B, Ke Z, Harmston N, Ho SY, Frois AO, Alam J et al (2016b) Wnt addiction of genetically defined cancers reversed by PORCN inhibition. Oncogene 35(17):2197–2207. https://doi.org/10.1038/onc.2015.280
Mahmoudi T, Li VS, Ng SS, Taouatas N, Vries RG, Mohammed S et al (2009) The kinase TNIK is an essential activator of Wnt target genes. EMBO J 28(21):3329–3340. https://doi.org/10.1038/emboj.2009.285
Maier TJ, Janssen A, Schmidt R, Geisslinger G, Grösch S (2005) Targeting the beta-catenin/APC pathway: a novel mechanism to explain the cyclooxygenase-2-independent anticarcinogenic effects of celecoxib in human colon carcinoma cells. FASEB J 19(10):1353–1355. https://doi.org/10.1096/fj.04-3274fje
Malta TM, Sokolov A, Gentles AJ, Burzykowski T, Poisson L, Weinstein JN et al (2018) Machine learning identifies stemness features associated with oncogenic dedifferentiation. Cell 173(2):338–54.e15. https://doi.org/10.1016/j.cell.2018.03.034
Manegold P, Lai KKY, Wu Y, Teo JL, Lenz HJ, Genyk YS et al (2018) Differentiation therapy targeting the β-catenin/CBP interaction in pancreatic cancer. Cancers (Basel) 10(4). https://doi.org/10.3390/cancers10040095
Marson A, Foreman R, Chevalier B, Bilodeau S, Kahn M, Young RA et al (2008) Wnt signaling promotes reprogramming of somatic cells to pluripotency. Cell Stem Cell 3(2):132–135. https://doi.org/10.1016/j.stem.2008.06.019
Masuda M, Uno Y, Ohbayashi N, Ohata H, Mimata A, Kukimoto-Niino M et al (2016) TNIK inhibition abrogates colorectal cancer stemness. Nat Commun 7:12586. https://doi.org/10.1038/ncomms12586
McMahon AP, Moon RT (1989) Ectopic expression of the proto-oncogene int-1 in Xenopus embryos leads to duplication of the embryonic axis. Cell 58(6):1075–1084. https://doi.org/10.1016/0092-8674(89)90506-0
McMillan M, Kahn M (2005) Investigating Wnt signaling: a chemogenomic safari. Drug Discov Today 10(21):1467–1474. https://doi.org/10.1016/S1359-6446(05)03613-5
Meng Q, Chen X, Sun L, Zhao C, Sui G, Cai L (2011) Carbamazepine promotes Her-2 protein degradation in breast cancer cells by modulating HDAC6 activity and acetylation of Hsp90. Mol Cell Biochem 348(1-2):165–171. https://doi.org/10.1007/s11010-010-0651-y
Menon M, Elliott R, Bowers L, Balan N, Rafiq R, Costa-Cabral S et al (2019) A novel tankyrase inhibitor, MSC2504877, enhances the effects of clinical CDK4/6 inhibitors. Sci Rep 9(1):201. https://doi.org/10.1038/s41598-018-36447-4
Merchant AA, Matsui W (2010) Targeting Hedgehog – a cancer stem cell pathway. Clin Cancer Res 16(12):3130–3140. https://doi.org/10.1158/1078-0432.CCR-09-2846
Miller RK, Hong JY, Muñoz WA, McCrea PD (2013) Beta-catenin versus the other armadillo catenins: assessing our current view of canonical Wnt signaling. Prog Mol Biol Transl Sci 116:387–407. https://doi.org/10.1016/B978-0-12-394311-8.00017-0
Miranda A, Hamilton PT, Zhang AW, Pattnaik S, Becht E, Mezheyeuski A et al (2019) Cancer stemness, intratumoral heterogeneity, and immune response across cancers. Proc Natl Acad Sci U S A 116(18):9020–9029. https://doi.org/10.1073/pnas.1818210116
Miyabayashi T, Teo JL, Yamamoto M, McMillan M, Nguyen C, Kahn M (2007) Wnt/beta-catenin/CBP signaling maintains long-term murine embryonic stem cell pluripotency. Proc Natl Acad Sci U S A 104(13):5668–5673. https://doi.org/10.1073/pnas.0701331104
Moore KN, Gunderson CC, Sabbatini P, McMeekin DS, Mantia-Smaldone G, Burger RA et al (2019) A phase 1b dose escalation study of ipafricept (OMP54F28) in combination with paclitaxel and carboplatin in patients with recurrent platinum-sensitive ovarian cancer. Gynecol Oncol 154(2):294–301. https://doi.org/10.1016/j.ygyno.2019.04.001
Morishita EC, Murayama K, Kato-Murayama M, Ishizuka-Katsura Y, Tomabechi Y, Hayashi T et al (2011) Crystal structures of the armadillo repeat domain of adenomatous polyposis coli and its complex with the tyrosine-rich domain of Sam68. Structure 19(10):1496–1508. https://doi.org/10.1016/j.str.2011.07.013
Ng M, Tan DS, Subbiah V, Weekes CD, Teneggi V, Diermayr V et al (2017) First-in-human phase 1 study of ETC-159 an oral PORCN inhibitor in patients with advanced solid tumours. J Clin Oncol 35(15_suppl):2584
Niehrs C (2012) The complex world of WNT receptor signalling. Nat Rev Mol Cell Biol 13(12):767–779. https://doi.org/10.1038/nrm3470
Nusse R, Varmus HE (1982) Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell 31(1):99–109. https://doi.org/10.1016/0092-8674(82)90409-3
Okazaki H, Sato S, Koyama K, Morizumi S, Abe S, Azuma M et al (2019) The novel inhibitor PRI-724 for Wnt/β-catenin/CBP signaling ameliorates bleomycin-induced pulmonary fibrosis in mice. Exp Lung Res 45(7):188–199. https://doi.org/10.1080/01902148.2019.1638466
Ono M, Lai KKY, Wu K, Nguyen C, Lin DP, Murali R et al (2018) Nuclear receptor/Wnt beta-catenin interactions are regulated via differential CBP/p300 coactivator usage. PLoS One 13(7):e0200714. https://doi.org/10.1371/journal.pone.0200714
Osawa Y, Oboki K, Imamura J, Kojika E, Hayashi Y, Hishima T et al (2015) Inhibition of cyclic adenosine monophosphate (cAMP)-response element-binding protein (CREB)-binding protein (CBP)/β-catenin reduces liver fibrosis in mice. EBioMedicine 2(11):1751–1758. https://doi.org/10.1016/j.ebiom.2015.10.010
Osawa Y, Kojika E, Nishikawa K, Kimura M, Osakaya S, Miyauchi H et al (2019) Programmed cell death ligand 1 (PD-L1) blockade attenuates metastatic colon cancer growth in cAMP-response element-binding protein (CREB)-binding protein (CBP)/β-catenin inhibitor-treated livers. Oncotarget 10(32):3013–3026. https://doi.org/10.18632/oncotarget.26892
Otero JJ, Fu W, Kan L, Cuadra AE, Kessler JA (2004) Beta-catenin signaling is required for neural differentiation of embryonic stem cells. Development 131(15):3545–3557. https://doi.org/10.1242/dev.01218
Pálmer HG, González-Sancho JM, Espada J, Berciano MT, Puig I, Baulida J et al (2001) Vitamin D(3) promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and the inhibition of beta-catenin signaling. J Cell Biol 154(2):369–387. https://doi.org/10.1083/jcb.200102028
Pannuti A, Foreman K, Rizzo P, Osipo C, Golde T, Osborne B et al (2010) Targeting notch to target cancer stem cells. Clin Cancer Res 16(12):3141–3152. https://doi.org/10.1158/1078-0432.CCR-09-2823
Park CH, Chang JY, Hahm ER, Park S, Kim HK, Yang CH (2005) Quercetin, a potent inhibitor against beta-catenin/Tcf signaling in SW480 colon cancer cells. Biochem Biophys Res Commun 328(1):227–234. https://doi.org/10.1016/j.bbrc.2004.12.151
Pendás-Franco N, Aguilera O, Pereira F, González-Sancho JM, Muñoz A (2008) Vitamin D and Wnt/beta-catenin pathway in colon cancer: role and regulation of DICKKOPF genes. Anticancer Res 28(5A):2613–2623
Phillips RK, Wallace MH, Lynch PM, Hawk E, Gordon GB, Saunders BP et al (2002) A randomised, double blind, placebo controlled study of celecoxib, a selective cyclooxygenase 2 inhibitor, on duodenal polyposis in familial adenomatous polyposis. Gut 50(6):857–860. https://doi.org/10.1136/gut.50.6.857
Pode-Shakked N, Harari-Steinberg O, Haberman-Ziv Y, Rom-Gross E, Bahar S, Omer D et al (2011) Resistance or sensitivity of Wilms’ tumor to anti-FZD7 antibody highlights the Wnt pathway as a possible therapeutic target. Oncogene 30(14):1664–1680. https://doi.org/10.1038/onc.2010.549
Polakis P (2012) Drugging Wnt signalling in cancer. EMBO J 31(12):2737–2746. https://doi.org/10.1038/emboj.2012.126
Proffitt KD, Madan B, Ke Z, Pendharkar V, Ding L, Lee MA et al (2013) Pharmacological inhibition of the Wnt acyltransferase PORCN prevents growth of WNT-driven mammary cancer. Cancer Res 73(2):502–507. https://doi.org/10.1158/0008-5472.CAN-12-2258
Rao CV, Desai D, Rivenson A, Simi B, Amin S, Reddy BS (1995) Chemoprevention of colon carcinogenesis by phenylethyl-3-methylcaffeate. Cancer Res 55(11):2310–2315
Rasola A, Fassetta M, De Bacco F, D'Alessandro L, Gramaglia D, Di Renzo MF et al (2007) A positive feedback loop between hepatocyte growth factor receptor and beta-catenin sustains colorectal cancer cell invasive growth. Oncogene 26(7):1078–1087. https://doi.org/10.1038/sj.onc.1209859
Reya T, Morrison SJ, Clarke MF, Weissman IL (2001) Stem cells, cancer, and cancer stem cells. Nature 414(6859):105–111. https://doi.org/10.1038/35102167
Ring A, Kim YM, Kahn M (2014) Wnt/catenin signaling in adult stem cell physiology and disease. Stem Cell Rev 10(4):512–525. https://doi.org/10.1007/s12015-014-9515-2
Ring A, Nguyen C, Smbatyan G, Tripathy D, Yu M, Press M et al (2018) CBP/β-catenin/FOXM1 is a novel therapeutic target in triple negative breast cancer. Cancers (Basel) 10(12). https://doi.org/10.3390/cancers10120525
Roccaro AM, Leleu X, Sacco A, Moreau AS, Hatjiharissi E, Jia X et al (2008) Resveratrol exerts antiproliferative activity and induces apoptosis in Waldenström’s macroglobulinemia. Clin Cancer Res 14(6):1849–1858. https://doi.org/10.1158/1078-0432.CCR-07-1750
Roth JF, Shikama N, Henzen C, Desbaillets I, Lutz W, Marino S et al (2003) Differential role of p300 and CBP acetyltransferase during myogenesis: p300 acts upstream of MyoD and Myf5. EMBO J 22(19):5186–5196. https://doi.org/10.1093/emboj/cdg473
Salminen JK, Tammela TL, Auvinen A, Murtola TJ (2016) Antiepileptic drugs with histone deacetylase inhibition activity and prostate cancer risk: a population-based case-control study. Cancer Causes Control 27(5):637–645. https://doi.org/10.1007/s10552-016-0737-2
Sandler RS, Halabi S, Baron JA, Budinger S, Paskett E, Keresztes R et al (2003) A randomized trial of aspirin to prevent colorectal adenomas in patients with previous colorectal cancer. N Engl J Med 348(10):883–890. https://doi.org/10.1056/NEJMoa021633
Saraswati S, Alfaro MP, Thorne CA, Atkinson J, Lee E, Young PP (2010) Pyrvinium, a potent small molecule Wnt inhibitor, promotes wound repair and post-MI cardiac remodeling. PLoS One 5(11):e15521. https://doi.org/10.1371/journal.pone.0015521
Sasaki T, Hwang H, Nguyen C, Kloner RA, Kahn M (2013) The small molecule Wnt signaling modulator ICG-001 improves contractile function in chronically infarcted rat myocardium. PLoS One 8(9):e75010. https://doi.org/10.1371/journal.pone.0075010
Sato N, Meijer L, Skaltsounis L, Greengard P, Brivanlou AH (2004) Maintenance of pluripotency in human and mouse embryonic stem cells through activation of Wnt signaling by a pharmacological GSK-3-specific inhibitor. Nat Med 10(1):55–63. https://doi.org/10.1038/nm979
Schenke-Layland K, Nsair A, Van Handel B, Angelis E, Gluck JM, Votteler M et al (2011) Recapitulation of the embryonic cardiovascular progenitor cell niche. Biomaterials 32(11):2748–2756. https://doi.org/10.1016/j.biomaterials.2010.12.046
Schinzari V, Timperi E, Pecora G, Palmucci F, Gallerano D, Grimaldi A et al (2018) Wnt3a/β-catenin signaling conditions differentiation of partially exhausted T-effector cells in human cancers. Cancer Immunol Res 6(8):941–952. https://doi.org/10.1158/2326-6066.CIR-17-0712
Schreinemachers DM, Everson RB (1994) Aspirin use and lung, colon, and breast cancer incidence in a prospective study. Epidemiology 5(2):138–146. https://doi.org/10.1097/00001648-199403000-00003
Sekita T, Yamada T, Kobayashi E, Yoshida A, Hirozane T, Kawai A et al (2020) Feasibility of targeting Traf2-and-Nck-interacting kinase in synovial sarcoma. Cancers (Basel) 12(5). https://doi.org/10.3390/cancers12051258
Shah S, Hecht A, Pestell R, Byers SW (2003) Trans-repression of beta-catenin activity by nuclear receptors. J Biol Chem 278(48):48137–48145. https://doi.org/10.1074/jbc.M307154200
Shan J, Shi DL, Wang J, Zheng J (2005) Identification of a specific inhibitor of the dishevelled PDZ domain. Biochemistry 44(47):15495–15503. https://doi.org/10.1021/bi0512602
Shitashige M, Satow R, Jigami T, Aoki K, Honda K, Shibata T et al (2010) Traf2- and Nck-interacting kinase is essential for Wnt signaling and colorectal cancer growth. Cancer Res 70(12):5024–5033. https://doi.org/10.1158/0008-5472.CAN-10-0306
Shultz MD, Cheung AK, Kirby CA, Firestone B, Fan J, Chen CH et al (2013) Identification of NVP-TNKS656: the use of structure-efficiency relationships to generate a highly potent, selective, and orally active tankyrase inhibitor. J Med Chem 56(16):6495–6511. https://doi.org/10.1021/jm400807n
Smith DC, Rosen L, Chugh R, Goldman JW, Xu L et al (2013) First-in-human evaluation of the human monoclonal antibody vantictumab (OMP-18R5; anti-frizzled) targeting the WNT pathway in a phase I study for patients with advanced solid tumors. J Clin Oncol Abstr 31:2540
Spranger S, Gajewski TF (2018) Impact of oncogenic pathways on evasion of antitumour immune responses. Nat Rev Cancer 18(3):139–147. https://doi.org/10.1038/nrc.2017.117
Spranger S, Dai D, Horton B, Gajewski TF (2017) Tumor-residing Batf3 dendritic cells are required for effector T cell trafficking and adoptive T cell therapy. Cancer Cell 31(5):711–23.e4. https://doi.org/10.1016/j.ccell.2017.04.003
Steinhart Z, Angers S (2018) Wnt signaling in development and tissue homeostasis. Development 145(11). https://doi.org/10.1242/dev.146589
Stettner M, Krämer G, Strauss A, Kvitkina T, Ohle S, Kieseier BC et al (2012) Long-term antiepileptic treatment with histone deacetylase inhibitors may reduce the risk of prostate cancer. Eur J Cancer Prev 21(1):55–64. https://doi.org/10.1097/CEJ.0b013e32834a7e6f
Sugano T, Masuda M, Takeshita F, Motoi N, Hirozane T, Goto N et al (2021) Pharmacological blockage of transforming growth factor-β signalling by a Traf2- and Nck-interacting kinase inhibitor, NCB-0846. Br J Cancer 124(1):228–236. https://doi.org/10.1038/s41416-020-01162-3
Takada R, Satomi Y, Kurata T, Ueno N, Norioka S, Kondoh H et al (2006) Monounsaturated fatty acid modification of Wnt protein: its role in Wnt secretion. Dev Cell 11(6):791–801. https://doi.org/10.1016/j.devcel.2006.10.003
Takada K, Zhu D, Bird GH, Sukhdeo K, Zhao JJ, Mani M et al (2012) Targeted disruption of the BCL9/β-catenin complex inhibits oncogenic Wnt signaling. Sci Transl Med 4(148):148ra17. https://doi.org/10.1126/scitranslmed.3003808
Takahashi-Yanaga F, Kahn M (2010) Targeting Wnt signaling: can we safely eradicate cancer stem cells? Clin Cancer Res 16(12):3153–3162. https://doi.org/10.1158/1078-0432.CCR-09-2943
Tam BY, Chiu K, Chung H, Bossard C, Nguyen JD, Creger E et al (2020) The CLK inhibitor SM08502 induces anti-tumor activity and reduces Wnt pathway gene expression in gastrointestinal cancer models. Cancer Lett 473:186–197. https://doi.org/10.1016/j.canlet.2019.09.009
Teo JL, Ma H, Nguyen C, Lam C, Kahn M (2005) Specific inhibition of CBP/beta-catenin interaction rescues defects in neuronal differentiation caused by a presenilin-1 mutation. Proc Natl Acad Sci U S A 102(34):12171–12176. https://doi.org/10.1073/pnas.0504600102
Thomas PD, Kahn M (2016) Kat3 coactivators in somatic stem cells and cancer stem cells: biological roles, evolution, and pharmacologic manipulation. Cell Biol Toxicol 32(1):61–81. https://doi.org/10.1007/s10565-016-9318-0
Thorne CA, Hanson AJ, Schneider J, Tahinci E, Orton D, Cselenyi CS et al (2010) Small-molecule inhibition of Wnt signaling through activation of casein kinase 1α. Nat Chem Biol 6(11):829–836. https://doi.org/10.1038/nchembio.453
Thun MJ, Henley SJ, Patrono C (2002) Nonsteroidal anti-inflammatory drugs as anticancer agents: mechanistic, pharmacologic, and clinical issues. J Natl Cancer Inst 94(4):252–266. https://doi.org/10.1093/jnci/94.4.252
Tian W, Han X, Yan M, Xu Y, Duggineni S, Lin N et al (2012) Structure-based discovery of a novel inhibitor targeting the β-catenin/Tcf4 interaction. Biochemistry 51(2):724–731. https://doi.org/10.1021/bi201428h
Tokunaga Y, Osawa Y, Ohtsuki T, Hayashi Y, Yamaji K, Yamane D et al (2017) Selective inhibitor of Wnt/β-catenin/CBP signaling ameliorates hepatitis C virus-induced liver fibrosis in mouse model. Sci Rep 7(1):325. https://doi.org/10.1038/s41598-017-00282-w
Trosset JY, Dalvit C, Knapp S, Fasolini M, Veronesi M, Mantegani S et al (2006) Inhibition of protein-protein interactions: the discovery of druglike beta-catenin inhibitors by combining virtual and biophysical screening. Proteins 64(1):60–67. https://doi.org/10.1002/prot.20955
Voronkov A, Holsworth DD, Waaler J, Wilson SR, Ekblad B, Perdreau-Dahl H et al (2013) Structural basis and SAR for G007-LK, a lead stage 1,2,4-triazole based specific tankyrase 1/2 inhibitor. J Med Chem 56(7):3012–3023. https://doi.org/10.1021/jm4000566
Wang C, Yan J, Yin P, Gui L, Ji L, Ma B et al (2020) β-Catenin inhibition shapes tumor immunity and synergizes with immunotherapy in colorectal cancer. Onco Targets Ther 9(1):1809947. https://doi.org/10.1080/2162402X.2020.1809947
Wei W, Chua MS, Grepper S, So SK (2011) Soluble Frizzled-7 receptor inhibits Wnt signaling and sensitizes hepatocellular carcinoma cells towards doxorubicin. Mol Cancer 10:16. https://doi.org/10.1186/1476-4598-10-16
Wend P, Fang L, Zhu Q, Schipper JH, Loddenkemper C, Kosel F et al (2013) Wnt/β-catenin signalling induces MLL to create epigenetic changes in salivary gland tumours. EMBO J 32(14):1977–1989. https://doi.org/10.1038/emboj.2013.127
**ao W, Jiang W, Shen J, Yin G, Fan Y, Wu D et al (2015) Retinoic acid ameliorates pancreatic fibrosis and inhibits the activation of pancreatic stellate cells in mice with experimental chronic pancreatitis via suppressing the Wnt/β-catenin signaling pathway. PLoS One 10(11):e0141462. https://doi.org/10.1371/journal.pone.0141462
Yamauchi T, Oike Y, Kamon J, Waki H, Komeda K, Tsuchida A et al (2002) Increased insulin sensitivity despite lipodystrophy in Crebbp heterozygous mice. Nat Genet 30(2):221–226. https://doi.org/10.1038/ng829
Yazici Y, McAlindon TE, Fleischmann R, Gibofsky A, Lane NE, Kivitz AJ et al (2017) A novel Wnt pathway inhibitor, SM04690, for the treatment of moderate to severe osteoarthritis of the knee: results of a 24-week, randomized, controlled, phase 1 study. Osteoarthr Cartil 25(10):1598–1606. https://doi.org/10.1016/j.joca.2017.07.006
Yazici Y, McAlindon TE, Gibofsky A, Lane NE, Clauw D, Jones M et al (2020) Lorecivivint, a novel intraarticular CDC-like kinase 2 and dual-specificity tyrosine phosphorylation-regulated kinase 1A inhibitor and Wnt pathway modulator for the treatment of knee osteoarthritis: a phase II randomized trial. Arthritis Rheumatol 72(10):1694–1706. https://doi.org/10.1002/art.41315
Yazici Y, McAlindon TE, Gibofsky A, Lane NE, Lattermann C, Skrepnik N et al (2021) A phase 2b randomized trial of lorecivivint, a novel intra-articular CLK2/DYRK1A inhibitor and Wnt pathway modulator for knee osteoarthritis. Osteoarthr Cartil 29(5):654–666. https://doi.org/10.1016/j.joca.2021.02.004
Zhang W, Lu W, Ananthan S, Suto MJ, Li Y (2017) Discovery of novel frizzled-7 inhibitors by targeting the receptor’s transmembrane domain. Oncotarget 8(53):91459–91470. https://doi.org/10.18632/oncotarget.20665
Zhang H, Bi Y, Wei Y, Liu J, Kuerban K, Ye L (2021) Blocking Wnt/β-catenin signal amplifies anti-PD-1 therapeutic efficacy by inhibiting tumor growth, migration, and promoting immune infiltration in glioblastomas. Mol Cancer Ther. https://doi.org/10.1158/1535-7163.MCT-20-0825
Zhao Y, Masiello D, McMillian M, Nguyen C, Wu Y, Melendez E et al (2016) CBP/catenin antagonist safely eliminates drug-resistant leukemia-initiating cells. Oncogene 35(28):3705–3717. https://doi.org/10.1038/onc.2015.438
Zhao Y, Ren J, Hillier J, Lu W, Jones EY (2020) Antiepileptic drug carbamazepine binds to a novel pocket on the Wnt receptor Frizzled-8. J Med Chem 63(6):3252–3260. https://doi.org/10.1021/acs.jmedchem.9b02020
Zhong Z, Sepramaniam S, Chew XH, Wood K, Lee MA, Madan B et al (2019) PORCN inhibition synergizes with PI3K/mTOR inhibition in Wnt-addicted cancers. Oncogene 38(40):6662–6677. https://doi.org/10.1038/s41388-019-0908-1
Zhou L, Li Y, Hao S, Zhou D, Tan RJ, Nie J et al (2015) Multiple genes of the renin-angiotensin system are novel targets of Wnt/β-catenin signaling. J Am Soc Nephrol 26(1):107–120. https://doi.org/10.1681/ASN.2014010085
Zinzalla V, Drobits-Handl B, Savchenko A, Rinnenthal J, Bauer MJ, Sanderson M et al (2019) BI 905677: a first-in-class LRP5/6 antagonist targeting Wnt-driven proliferation and immune escape. Cancer Res 79(13 Suppl):Abstract nr DDT01-01
Acknowledgements
The authors thank David P. Lin for assistance with illustrations/graphics. Keane K.Y. Lai has been supported by US National Institutes of Health (NIH) grant K08AA025112. Michael Kahn has been supported by City of Hope Comprehensive Cancer Center Support Grant NIH P30CA033572, NIH grants R01CA166161, R21NS074392, R21AI105057, and R01HL112638, and the Rotary Coins for Alzheimer’s Research Trust (CART).
Conflicts of Interest
Michael Kahn has an equity position in 3 + 2 Pharma.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Lai, K.K.Y., Kahn, M. (2021). Pharmacologically Targeting the WNT/β-Catenin Signaling Cascade: Avoiding the Sword of Damocles. In: Schulte, G., Kozielewicz, P. (eds) Pharmacology of the WNT Signaling System. Handbook of Experimental Pharmacology, vol 269. Springer, Cham. https://doi.org/10.1007/164_2021_523
Download citation
DOI: https://doi.org/10.1007/164_2021_523
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-85498-0
Online ISBN: 978-3-030-85499-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)