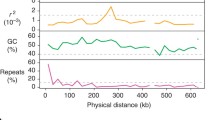
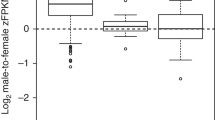
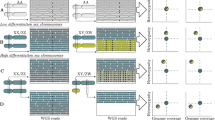
Abstract
Sex chromosomes and sex-linked genes usually show unusual features comparing to the rest of the genome and thus are of particular interests to evolutionary and developmental biologists. Here we describe recently developed bioinformatic methods for identifying sex-linked sequences, in a genome without priori linkage information. Some are developed during our course of studying avian genomes. These methods require sequence data, either assembled draft genome or raw sequences derived from the heterogametic sex (e.g., a female bird or a male mammal). Their application is not restricted to birds but can be used for any species with a sex chromosome pair that has diverged from each other for a substantial degree.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Charlesworth B, Coyne JA, Barton NH (1987) The relative rates of evolution of sex chromosomes and autosomes. Am Nat 130:113–146
Meisel RP, Malone JH, Clark AG (2012) Faster-X evolution of gene expression in Drosophila. PLoS Genet 8:e1003013. doi:10.1371/journal.pgen.1003013
Kharchenko PV, ** R, Park PJ (2011) Evidence for dosage compensation between the X chromosome and autosomes in mammals. Nat Genet 43:1167–1169. doi:10.1038/ng.991
Bellott DW, Hughes JF, Skaletsky H, Brown LG, Pyntikova T, Cho T-J, Koutseva N, Zaghlul S, Graves T, Rock S, Kremitzki C, Fulton RS, Dugan S, Ding Y, Morton D, Khan Z, Lewis L, Buhay C, Wang Q, Watt J, Holder M, Lee S, Nazareth L, Rozen S, Muzny DM, Warren WC, Gibbs RA, Wilson RK, Page DC (2014) Mammalian Y chromosomes retain widely expressed dosage-sensitive regulators. Nature 508:494–499. doi:10.1038/nature13206
Huynh KD, Lee JT (2003) Inheritance of a pre-inactivated paternal X chromosome in early mouse embryos. Nature 426:857–862. doi:10.1038/nature02222
Julien P, Brawand D, Soumillon M, Necsulea A, Liechti A, Schutz F, Daish T, Grutzner F, Kaessmann H (2012) Mechanisms and evolutionary patterns of mammalian and avian dosage compensation. PLoS Biol 10(5):e1001328. doi:10.1371/journal.pbio.1001328
Zhou Q, Zhang J, Bachtrog D, An N, Huang Q, Jarvis ED, Gilbert MTP, Zhang G (2014) Complex evolutionary trajectories of sex chromosomes across bird taxa. Science 346:1246338. doi:10.1126/science.1246338
Carvalho A, Clark A (2013) Efficient identification of Y chromosome sequences in the human and Drosophila genomes. Genome Res 23:1894–1907. doi:10.1101/gr.156034.113
Yandell M, Ence D (2012) A beginner’s guide to eukaryotic genome annotation. Nat Rev Genet 13:329–342. doi:10.1038/nrg3174
Parsch J, Ellegren H (2013) The evolutionary causes and consequences of sex-biased gene expression. Nat Rev Genet 14:83–87. doi:10.1038/nrg3376
Ellegren H, Hultin-Rosenberg L, Brunström B, Dencker L, Kultima K, Scholz B (2007) Faced with inequality: chicken do not have a general dosage compensation of sex-linked genes. BMC Biol 5:40. doi:10.1186/1741-7007-5-40
Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, Pimentel H, Salzberg SL, Rinn JL, Pachter L (2012) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 7:562–578. doi:10.1038/nprot.2012.016
Pertea M, Kim D, Pertea GM, Leek JT, Salzberg SL (2016) Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat Protoc 11:1650–1667. doi:10.1038/nprot.2016.095
Bray NL, Pimentel H, Melsted P, Pachter L (2016) Near-optimal probabilistic RNA-seq quantification. Nat Biotechnol 34:525–527. doi:10.1038/nbt.3519
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng Q, Chen Z, Mauceli E, Hacohen N, Gnirke A, Rhind N, di Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652. doi:10.1038/nbt.1883
**e Y, Wu G, Tang J, Luo R, Patterson J, Liu S, Huang W, He G, Gu S, Li S, Zhou X, Lam TW, Li Y, Xu X, Wong GKS, Wang J (2014) SOAPdenovo-Trans: de novo transcriptome assembly with short RNA-Seq reads. Bioinformatics 30:1660–1666. doi:10.1093/bioinformatics/btu077
Andrews S (2010) FastQC—a quality control tool for high throughput sequence data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc/. Accessed 25 Aug 2013
Chen N, Bellott DW, Page DC, Clark AG (2012) Identification of avian W-linked contigs by short-read sequencing. BMC Genomics 13:183. doi:10.1186/1471-2164-13-183
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnetjournal 17:10. doi:10.14806/ej.17.1.200
Li R, Yu C, Li Y, Lam TW, Yiu SM, Kristiansen K, Wang J (2009) SOAP2: an improved ultrafast tool for short read alignment. Bioinformatics 25:1966–1967. doi:10.1093/bioinformatics/btp336
Smit A, Hubley R, Green P (2016) RepeatMasker Open-4.0. http://www.repeatmasker.org/ (2013–2015)
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25:1754–1760. doi:10.1093/bioinformatics/btp324
She R, Chu JS, Uyar B, Wang J, Wang K, Chen N (2011) genBlastG: using BLAST searches to build homologous gene models. Bioinformatics 27(15):2141–2143. doi:10.1093/bioinformatics/btr342
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) The Sequence Alignment/Map format and SAMtools. Bioinformatics (Oxford, England) 25:2078–2079. doi:10.1093/bioinformatics/btp352
Wysoker A, Tibbetts K, Fennell T (2013) Picard-Tools 1.9.2. http://picard.sourceforge.net/. Accessed 28 May 2013
Quinlan AR, Hall IM (2010) BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26:841–842. doi:10.1093/bioinformatics/btq033
Marçais G, Kingsford C (2011) A fast, lock-free approach for efficient parallel counting of occurrences of k-mers. Bioinformatics 27:764–770. doi:10.1093/bioinformatics/btr011
Bellott DW, Skaletsky H, Pyntikova T, Mardis ER, Graves T, Kremitzki C, Brown LG, Rozen S, Warren WC, Wilson RK, Page DC (2010) Convergent evolution of chicken Z and human X chromosomes by expansion and gene acquisition. Nature 466:612–616. doi:10.1038/nature09172
Huang X, Chen X-G, Armbruster PA (2016) Comparative performance of transcriptome assembly methods for non-model organisms. BMC Genomics 17:523. doi:10.1186/s12864-016-2923-8
Li B, Fillmore N, Bai Y, Collins M, Thomson JA, Stewart R, Dewey C (2014) Evaluation of de novo transcriptome assemblies from RNA-Seq data. Genome Biol. doi:10.1101/006338
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Zhang, J., Li, J., Zhou, Q. (2017). Genomic and Transcriptomic Analyses of Avian Sex Chromosomes and Sex-Linked Genes. In: Sheng, G. (eds) Avian and Reptilian Developmental Biology. Methods in Molecular Biology, vol 1650. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7216-6_4
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7216-6_4
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7215-9
Online ISBN: 978-1-4939-7216-6
eBook Packages: Springer Protocols