Abstract
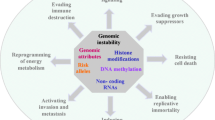
Intracellular and environmental agents are the genotoxic agents that cause toxicity in genome and are the main reason of genomic instability. Genome instability is the primary cause of cancer. In order to encounter the damage caused by endogenous or exogenous agents, there are cell cycle checkpoints that strictly regulate the replication of DNA. They also check and control proper distribution of chromosomes between two sister chromatids during cell division. DNA repair pathways; DNA damage response genes that trigger the repairing pathways, e.g., excision repair pathways (NER, BER, and DNA mismatch repair); and double-strand base repair pathway also correct damages and thus ensure integrity and survival of genome. Microsatellite, chromosomal, and nucleotide instabilities are the main transitions in cancer cells from normal that promote tumor metastasis caused by these toxic agents. Epigenome of individual (specific inherited patterns or changes that modify gene expression without affecting other sequences) plays an important role in maintaining the integrity of DNA, e.g., DNA methylation and histone modification, but the mutations or instability in these patterns can promote tumorigenesis. For diagnosis of large-scale and small-scale tumorigenesis, there are different techniques, e.g., FISH, AP-PCR, ISSR-PCR, and array CGH; they provide us the wide spectrum of therapeutic opportunities for characterizing the individual’s tumor.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Broustas CG, Lieberman HB (2014) DNA damage response genes and the development of cancer metastasis. Radiat Res 181(2):111–130
Chen H, Maxwell C, Connell M (2015) The generation, detection, and prevention of genomic instability during cancer progression and metastasis. In: Genomic instability and cancer metastasis. Springer, Cham, pp 15–38
Futreal PA, Coin L, Marshall M, Down T, Hubbard T, Wooster R, Rahman N, Stratton MR (2004) A census of human cancer genes. Nat Rev Cancer 4(3):177
Halling KC, Harper J, Moskaluk CA, Thibodeau SN, Petroni GR, Yustein AS, Tosi P, Minacci C, Roviello F, Piva P (1999) Origin of microsatellite instability in gastric cancer. Am J Pathol 155(1):205–211
Lengauer C, Kinzler KW, Vogelstein B (1998) Genetic instabilities in human cancers. Nature 396(6712):643
Almoguera C, Shibata D, Forrester K, Martin J, Arnheim N, Perucho M (1988) Most human carcinomas of the exocrine pancreas contain mutant c-K-ras genes. Cell 53(4):549–554
Lee HC, Yin PH, Lin JC, Wu CC, Chen CY, Wu CW, Chi CW, Tam TN, Wei YH (2005) Mitochondrial genome instability and mtDNA depletion in human cancers. Ann N Y Acad Sci 1042(1):109–122
Jefford CE, Irminger-Finger I (2006) Mechanisms of chromosome instability in cancers. Crit Rev Oncol Hematol 59(1):1–14
Gagos S, Irminger-Finger I (2005) Chromosome instability in neoplasia: chaotic roots to continuous growth. Int J Biochem Cell Biol 37(5):1014–1033
Wang SI, Puc J, Li J, Bruce JN, Cairns P, Sidransky D, Parsons R (1997) Somatic mutations of PTEN in glioblastoma multiforme. Cancer Res 57(19):4183–4186
Ferguson LR, Chen H, Collins AR, Connell M, Damia G, Dasgupta S, Malhotra M, Meeker AK, Amedei A, Amin A (2015) Genomic instability in human cancer: molecular insights and opportunities for therapeutic attack and prevention through diet and nutrition. Semin Cancer Biol 35:S5–S24
Aguilera A, Gómez-González B (2008) Genome instability: a mechanistic view of its causes and consequences. Nat Rev Genet 9(3):204
Houtgraaf JH, Versmissen J, van der Giessen WJ (2006) A concise review of DNA damage checkpoints and repair in mammalian cells. Cardiovasc Revasc Med 7(3):165–172
Sancar A, Lindsey-Boltz LA, Ünsal-Kaçmaz K, Linn S (2004) Molecular mechanisms of mammalian DNA repair and the DNA damage checkpoints. Annu Rev Biochem 73(1):39–85
Hanawalt PC, Spivak G (2008) Transcription-coupled DNA repair: two decades of progress and surprises. Nat Rev Mol Cell Biol 9(12):958
Thoms KM, Kuschal C, Emmert S (2007) Lessons learned from DNA repair defective syndromes. Exp Dermatol 16(6):532–544
Albertson TM, Ogawa M, Bugni JM, Hays LE, Chen Y, Wang Y, Treuting PM, Heddle JA, Goldsby RE, Preston BD (2009) DNA polymerase ε and δ proofreading suppress discrete mutator and cancer phenotypes in mice. Proc Natl Acad Sci 106(40):17101–17104
Ciccia A, Elledge SJ (2010) The DNA damage response: making it safe to play with knives. Mol Cell 40(2):179–204
Helleday T, Lo J, van Gent DC, Engelward BP (2007) DNA double-strand break repair: from mechanistic understanding to cancer treatment. DNA Repair 6(7):923–935
Wan T, Ma ES (2012) Molecular cytogenetics: an indispensable tool for cancer diagnosis. Chang Gung Med J 35(2):96–110
Moynahan ME, Jasin M (2010) Mitotic homologous recombination maintains genomic stability and suppresses tumorigenesis. Nat Rev Mol Cell Biol 11(3):196
Hall JM, Lee MK, Newman B, Morrow JE, Anderson LA, Huey B, King M-C (1990) Linkage of early-onset familial breast cancer to chromosome 17q21. Science 250(4988):1684–1689
Miller OJ, Schnedl W, Allen J, Erlanger BF (1974) 5-Methylcytosine localised in mammalian constitutive heterochromatin. Nature 251(5476):636
Sharma S, Kelly TK, Jones PA (2010) Epigenetics in cancer. Carcinogenesis 31(1):27–36
Wilson AS, Power BE, Molloy PL (2007) DNA hypomethylation and human diseases. Biochim Biophys Acta 1775(1):138–162
Ehrlich M (2002) DNA methylation in cancer: too much, but also too little. Oncogene 21(35):5400
Kouzarides T (2007) Chromatin modifications and their function. Cell 128(4):693–705
Fraga MF, Ballestar E, Villar-Garea A, Boix-Chornet M, Espada J, Schotta G, Bonaldi T, Haydon C, Ropero S, Petrie K (2005) Loss of acetylation at Lys16 and trimethylation at Lys20 of histone H4 is a common hallmark of human cancer. Nat Genet 37(4):391
Mavrich TN, Ioshikhes IP, Venters BJ, Jiang C, Tomsho LP, Qi J, Schuster SC, Albert I, Pugh BF (2008) A barrier nucleosome model for statistical positioning of nucleosomes throughout the yeast genome. Genome Res 18(7):1073–1083
Shen Z (2011) Genomic instability and cancer: an introduction. J Mol Cell Biol 3(1):1–3
Aguilera A, García-Muse T (2013) Causes of genome instability. Annu Rev Genet 47:1–32
Vogelstein B (1990) Cancer. A deadly inheritance [news; comment]. Nature 348:681–682
Kommajosyula N, Rhind N (2006) Cdc2 tyrosine phosphorylation is not required for the S-phase DNA damage checkpoint in fission yeast. Cell Cycle 5(21):2495–2500
Maresca TJ, Salmon E (2010) Welcome to a new kind of tension: translating kinetochore mechanics into a wait-anaphase signal. J Cell Sci 123(6):825–835
Lara-Gonzalez P, Westhorpe FG, Taylor SS (2012) The spindle assembly checkpoint. Curr Biol 22(22):R966–R980
de Medina-Redondo M, Meraldi P (2011) The spindle assembly checkpoint: clock or domino? In: Cell cycle in development. Springer, Berlin, pp 75–91
Lens SM, Voest EE, Medema RH (2010) Shared and separate functions of polo-like kinases and aurora kinases in cancer. Nat Rev Cancer 10(12):825
Anand S, Penrhyn-Lowe S, Venkitaraman AR (2003) AURORA-A amplification overrides the mitotic spindle assembly checkpoint, inducing resistance to Taxol. Cancer Cell 3(1):51–62
Foley EA, Kapoor TM (2013) Microtubule attachment and spindle assembly checkpoint signalling at the kinetochore. Nat Rev Mol Cell Biol 14(1):25
Jhappan C, Noonan FP, Merlino G (2003) Ultraviolet radiation and cutaneous malignant melanoma. Oncogene 22(20):3099
Hecht SS (1999) Tobacco smoke carcinogens and lung cancer. J Natl Cancer Inst 91(14):1194–1210
Orjuela M, Castaneda VP, Ridaura C, Lecona E, Leal C, Abramson DH, Orlow I, Gerald W, Cordon-Cardo C (2000) Presence of human papilloma virus in tumor tissue from children with retinoblastoma: an alternative mechanism for tumor development. Clin Cancer Res 6(10):4010–4016
Blount BC, Mack MM, Wehr CM, MacGregor JT, Hiatt RA, Wang G, Wickramasinghe SN, Everson RB, Ames BN (1997) Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: implications for cancer and neuronal damage. Proc Natl Acad Sci 94(7):3290–3295
Fenech M (2001) The role of folic acid and vitamin B12 in genomic stability of human cells. Mutat Res 475(1–2):57–67
Satia JA, Keku T, Galanko JA, Martin C, Doctolero RT, Tajima A, Sandler RS, Carethers JM (2005) Diet, lifestyle, and genomic instability in the North Carolina Colon Cancer Study. Cancer Epidemiol Biomarkers Prev 14(2):429–436
Mol BM, Massink MP, van der Hout AH, Dommering CJ, Zaman JM, Bosscha MI, Kors WA, Meijers-Heijboer HE, Kaspers GJ, Riele H (2014) High resolution SNP array profiling identifies variability in retinoblastoma genome stability. Genes Chromosom Cancer 53(1):1–14
Mettu RK, Wan Y-W, Habermann JK, Ried T, Guo NL (2010) A 12-gene genomic instability signature predicts clinical outcomes in multiple cancer types. Int J Biol Markers 25(4):219–228
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Gull, H., Masood, N. (2020). Genomic Instability and Cancer Metastasis. In: Masood, N., Shakil Malik, S. (eds) 'Essentials of Cancer Genomic, Computational Approaches and Precision Medicine. Springer, Singapore. https://doi.org/10.1007/978-981-15-1067-0_6
Download citation
DOI: https://doi.org/10.1007/978-981-15-1067-0_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-1066-3
Online ISBN: 978-981-15-1067-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)