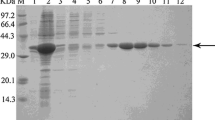
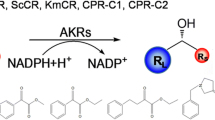
Abstract
Aldehyde ketone reductase (AKR) belongs to the redox enzyme superfamily. The aldo-keto reductase (AKR) protein superfamily contains over 190 members that fall into 16 families. Its biological catalysis in the preparation of chiral compounds has significant advantages. Therefore, application of aldehyde ketone reductase gains more benefit as industrial catalysts. Recently, a variety of microbial aldehyde ketone reductase has been used in the synthesizing the high value of chiral compounds. Here we reviewed structure and functional modification of aldehyde ketone reductase protein, gene mining of novel AKR enzyme for industrial catalysts and co-enzyme regeneration using the genome database.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Penning TM (2015) The aldo-keto reductases (AKRs): overview. Chem Biol Interact 234:236–246
Ren D, Villeneuve NF, Jiang T et al (2011) Brusatol enhances the efficacy of chemotherapy by inhibiting the Nrf2-mediated defense mechanism. Proc Natl Acad Sci U S A 108(4):1433–1438
Couture JF, Legrand P, Cantin L et al (2004) Loop relaxation, a mechanism that explains the reduced specificity of rabbit 20α-hydroxysteroid dehydrogenase, a member of the aldo-keto reductase superfamily. J Mol Biol 339:89–102
Sanli G, Blaber M (2001) Structural assembly of the active site in an aldo-keto reductase by NADPH cofactor. J Mol Biol 309(5):1209–1218
Widboom PF, Fielding EN, Liu Y et al (2007) Structural basis for cofactor-independent dioxygenation in vancomycin biosynthesis. Nature 447(7142):342–345
Ziegelmann-Fjeld KI, Musa MM, Phillips RS et al (2007) A Thermoanaerobacter ethanolicus secondary alcohol dehydrogenase mutant derivative highly active and stereoselective on phenylacetone and benzylacetone. Protein Eng Des Sel 20(2):47–55
Zhu D, Yang Y, Majkowicz S et al (2008) Inverting the enantioselectivity of a carbonyl reductase via substrate—enzyme docking-guided point mutation. Org Lett 10(4):525–528
Morikawa S, Nakai T, Yasohara Y et al (2005) Highly active mutants of carbonyl reductase s1 with inverted coenzyme specificity and production of optically active alcohols. Biosci Biotechnol Biochem 69(3):544–552
Zhang R, Xu Y, Sun Y et al (2009) Ser67Asp and His68Asp substitutions in Candida parapsilosis carbonyl reductase alter the coenzyme specificity and enantioselectivity of ketone reduction. Appl Environ Microbiol 75(7):2176–2183
Qin HM, Yamamura A, Miyakawa T et al (2013) Crystal structure of conjugated polyketone reductase (CPR-C1) from Candida parapsilosis IFO 0708 complexed with NADPH. Proteins 81:2059–2063
Fewer M, Martínez-Abarca F, Golyshin PN (2005) Mining genomes and metagenomes for novel catalysts. Curr Opin Biotechnol 16(6):588–593
Hidalgo A, Akond MA, Kita K et al (2001) Isolation and primary structural analysis of two conjugated polyketone reductases from Candida parapsilosis. Biosci Biotechnol Biochem 65(12):2785–2788
Kaluzna IA, Matsuda T, Sewell AK et al (2004) Systematic investigation of Saccharomyces cerevisiae enzymes catalyzing carbonyl reductions. J Am Chem Soc 126(40):12827–12832
Guo RY, Nie Y, Mu XQ et al (2014) Preparation of asymmetric transformation of (S)-N, N-dimethy1-3-hydroxyl-3-(2-thieny1)-1-propanamine(DHTP) by the novel AKR. Cheml Ind Eng Progr 33(9):2344–2349
Zhao H, Donk WA (2003) Regeneration of cofactors for use in biocatalysis. Curr Opin Biotechnol 4(6):583–589
Kataoka K, Kita M, Wada Y, Yasohara J et al (2003) Novel bioreduction system for the production of chiral alcohols. Appl Microbiol Biotechnol 62:437–445
Chin-Joe I, Straathof AJ, Pronk JT et al (2001) Influence of the ethanol and glucose supply rate on the rate and enantioselectivity of 3-oxo ester reduction by baker’s yeast. Biotechnol Bioeng 75:29–38
Makino Y, Ding JY, Negoro S et al (1989) Purification and characterization of a new glucose dehydrogenase from vegetative cells of Bacillus megaterimn. J Ferment Bioeng 67:374–379
Kizaki N, Yasohara Y, Hasegawa J et al (2001) Synthesis of optically pure ethyl(S)-4-chloro-3-hydroxybutanoate by Escherichia coli transformant cells coexpressing the carbonyl reductase and glucose dehydrogenase genes. Microbiol Biotechnol 55(5):590–595
Ni Y, Li CX, Wang LJ et al (2011) Highly stereoselective reduction of prochiral ketones by a bacterial reductase coupled with cofactor regeneration. Org Biomol Chem 9(15):5463–5468
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this paper
Cite this paper
Li, S. et al. (2018). Research Progress of Aldehyde Ketone Reductase for Asymmetric Catalysis of Chiral Compounds. In: Liu, H., Song, C., Ram, A. (eds) Advances in Applied Biotechnology. ICAB 2016. Lecture Notes in Electrical Engineering, vol 444. Springer, Singapore. https://doi.org/10.1007/978-981-10-4801-2_80
Download citation
DOI: https://doi.org/10.1007/978-981-10-4801-2_80
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-4800-5
Online ISBN: 978-981-10-4801-2
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)