Abstract
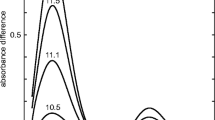
Recently, we have reported the use of near-infrared (NIR) Yb3+ vibronic sideband (VSB) spectroscopy to obtain structural information of metal-binding sites in proteins (1). In the NIR fluorescence spectrum of metalloproteins where Yb3+ is reconstituted into the metal binding site, weak vibronic sidebands that are shifted in energy with respect to the principal Yb3+ 2F5/2 -> 2F7/2 zero-phonon electronic transition (at ca. 970 nm), correspond to the vibrational frequencies of the Yb3+ ligands. This Yb3+ VSB spectrum provides a selective well-resolved “infrared-like” vibrational spectrum of the ligands which constitute the metal binding site.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
C.Roselli,A.Boussac and T.A. Mattioli (1994)Proc.Natl.Acad.Sci. 9112897
V L Pecoraro, W.R. Harris, C. J. Carrano and K. N. Raymond (1981) Biochemistry 20, 7033
A.D.Sherry,E.R.Birnbaum and D.W.Darnall,(1972)J.Biol.Chem. 247 3489
C. Berthomieu and A. Boussac(1995) submitted to Biospectriscopy
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1995 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Roselli, C., Boussac, A., Mattioli, T.A. (1995). Near Infrared Yb3+ Vibronic Sideband Spectroscopy of Proteins: Amino-Acid Side Chain Models. In: Merlin, J.C., Turrell, S., Huvenne, J.P. (eds) Spectroscopy of Biological Molecules. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-0371-8_36
Download citation
DOI: https://doi.org/10.1007/978-94-011-0371-8_36
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-4166-9
Online ISBN: 978-94-011-0371-8
eBook Packages: Springer Book Archive