Abstract
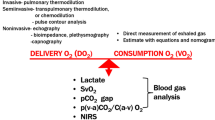
Dysoxia is inadequacy of tissue oxygenation, the condition when oxygen levels are so low that mitochondrial respiration can no longer be sustained [1]. It is assumed that tissue dysoxia and oxygen debt are major factors in the development and the propagation of multiple organ failure (MOF) in critically ill patients. Dysoxia is the result of an abnormal relationship between oxygen supply (DO2) and oxygen demand. In order to prevent its occurrence the maintenance of ‘adequate’ mean arterial pressure (MAP), cardiac output, and DO2 are essential goals of therapy. However, the adequacy of these goals is very difficult to define. Ultimately, a normal relationship between DO2 and oxygen demand should be determined at the mitochondrial level. The measurement of tissue bioenergetics would provide a needed gold standard [2]. Several strategies have been tried recently to avoid the development of oxygen debt in intensive care patients. These strategies involve improvement of systemic hemodynamics and oxygen-derived parameters and, more recently, have focused on regional parameters. This chapter presents these strategies and assesses their usefulness in current practice.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Connett RJ, Honig CR, Gayeski TEJ, Brooks GA (1990) Defining hypoxia: a system view of VO2, glycolysis, energetics and intracellular P02. J Appl Physiol 68: 833–842
Dantzker DR (1993) Adequacy of tissue oxygenation. Crit Care Med 21: S40 - S43
Robin ED (1980) Of men and mitochondria: co** with hypoxic dysoxia. Am Rev Respir Dis 122: 517–531
Vanderkooi JM, Erecinska M, Silver IA (1991) Oxygen in mammalian tissues: methods of measurement and affinities of various reactions. Am J Physiol 260: C1131 - C1150
Cain SM (1996) Metabolic alterations with hypoxia. Rean Urg 5: 174–177
Guery BPH, Mangalaboyi J, Menager P, Mordon S, Vallet B, Chopin C (1999) Redox status of cytochrome aa3: a non invasive indicator of dysoxia in regional hypoxic or ischemic hypoxia. Crit Care Med 27: 576–582
Cain SM (1965) Appearance of excess lactate in anesthetized dogs during anemic and hypoxic hypoxia. Am J Physiol 209: 604–610
West JB (1990) Gas transport to the periphery. In: West JB (ed) Respiratory physiology–the essentials. Williams and Wilkins, Baltimore, pp 69–85
Vincent JL (1998) The available clinical tools–oxygen-derived variables, lactate, and pHi. In: Sibbald WJ, Messmer K, Fink MP (eds) Tissue oxygenation in acute medicine. Springer, Berlin, pp 193–203
Schlichtig R, Pinsky MR (1991) Defining the hypoxic threshold. Crit Care Med 19: 147–149
Chapler CK, Cain SM (1986) The physiologic reserve in oxygen carrying capacity: studies in experimental hemodilution. Can J Physiol Pharmacol 64: 7–12
Cain SM (1977) Oxygen delivery and uptake in dogs during anemic and hypoxic hypoxia. J Appl Physiol 42: 228–234
Ronco JJ, Fenwick JC, Tweedale MG, et al (1993) Identification of the critical oxygen delivery for anaerobic metabolism in critically ill septic and non septic humans. JAMA 270: 1724–1730
Van Woerkens ECSM, Trouwborst A, Van Lanschot JJB (1992) Profound hemodilution: what is the critical level of hemodilution at which oxygen delivery-dependent oxygen consumption starts in an anesthetized human? Anesth Analg 75: 818–821
Wasserman K, Whipp BJ (1975) Exercise physiology in health and disease. Am Rev Respir Dis 112: 219–249
Vallet B, Lejus C, Ozier Y, Murat I, Lienhart A (1996) Peut-on définir le contenu et le transport artériel en oxygène adaptés aux besoins en cas d’anémie aiguë? Cah Anesthésiol 45: 117–122
Consensus Conference (1988) Perioperative red cell transfusion. JAMA 260: 2700–2703
Hébert P, Wells G, Blajchman MA, et al (1999) A multicenter, randomized, controlled clinical trial of transfusion requirements in critical care. N Engl J Med 340: 409–417
Shoemaker WC, Appel PL, Waxman K, Schwartz S, Chang P (1982) Clinical trial of survivors’ cardiorespiratory patterns as therapeutic goals in critically ill postoperative patients. Crit Care Med 10: 398–403
Vallet B, Chopin C, Curtis SE, et al (1993) Prognostic value of the dobutamine test in patients with sepsis syndrome and normal lactate values: a prospective, multicenter study. Crit Care Med 21: 1868–1875
Rhodes A, Malagnon I, Lamb FJ, et al (1996) Failure to increase oxygen consumption is a predictor of mortality in septic patients. Intensive Care Med 22: S274 (Abst)
Hayes MA, Timmins AC, Yau EHS, et al (1997) Oxygen transport patterns in patients with sepsis syndrome or septic shock: influence of treatment and relationship to outcome. Crit Care Med 25: 926–936
Vallet B, Chopin C and the dobutamine in sepsis study group (2000) The supranormal oxygen delivery controversy. Crit Care Med (in press)
Hayes MA, Timmins AC, Yau EHS, et al (1994) Elevation of systemic oxygen delivery in the treatment of critically ill patients. N Engl J Med 330: 1717–1722
Gutierrez G, Clark C, Brown SD, et al (1994) Effect of dobutamine on oxygen consumption and gastric mucosal pH in septic patients. Am J Respir Crit Care Med 150: 324–329
Gutierrez G, Palizas F, Doglio G, et al (1992) Gastric intramucosal pH as a therapeutic index of tissue oxygenation in critically ill patients. Lancet 339: 195–199
Bowles SA, Schlichtig R, Kramer DJ, Klions HA (1992) Arteriovenous pH and partial pressure of carbon dioxide detect critical oxygen delivery during progressive hemorrhage in dogs. J Crit Care 7: 95–105
Van der Linden P, Rausin I, Deltell A, et al (1995) Detection of tissue hypoxia by arteriovenous gradient for PCO2 and pH in anesthetized dogs during progressive hemorrhage. Anesth Analg 80: 269–275
Zhang H, Vincent JL (1993) Arteriovenous difference in PCO2 and pH are good indicators of critical hypoperfusion. Am Rev Respir Dis 148: 867–871
Teboul JL, Michard F, Richard C (1996) Critical analysis of venoarterial CO, gradient as a marker of tissue hypoxia. In: Vincent JL (ed) Yearbook of intensive care and emergency medicine. Springer-Verlag, Berlin, pp 296–307
Vallet B, Teboul JL, Cain SM, Curtis SE (2000) Veno-arterial CO2 difference during regional ischemic or hypoxic hypoxia. J Appl Physiol (in press)
Salzman AL, Wang H, Wollert PS, et al (1994) Endotoxin-induced ileal mucosal hyperpermeability in pigs: role of tissue acidosis. Am J Physiol 266: G633 - G646
Landow L, Andersen LW (1994) Splanchnic ischaemia and its role in multiple organ failure. Acta Anaesthesiol Scand 38: 626–639
Schlichtig R, Bowles SA (1994) Distinguishing between aerobic and anaerobic appearance of dissolved CO2 in intestine during low flow. J Appl Physiol 76: 2443–2451
Vallet B, Durinck L, Chagnon JL, Nevière R (1996) Effects of hypoxic hypoxia on veno-and gut mucosal-arterial PCO2 difference in pigs. Anesthesiology, 1996, 85: A607
Vallet B, Lund N, Curtis SE, Kelly D, Cain SM (1994) Gut and muscle tissue P02 in endotoxemic dogs during shock and resuscitation. J Appl Physiol 76: 793–800
Lote CJ, Harper L, Savage COS (1996) Mechanisms of acute renal failure. Br J Anaesth 77: 82–89
Vallet B, Nevière R, Chagnon JL (1996) Gastrointestinal mucosal ischemia. In: Rombeau JL, Takala J (eds) Gut dysfunction in critical illness. Springer-Verlag, Berlin, pp 233–245
Temmesfeld-Wollbrück B, Szalay A, Mayer K, et al (1998) Abnormalities of gastric mucosal oxygenation in septic shock. Am J Respir Crit Care Med 157: 1586–1592
Dawson AM, Trenchard D, Guz A (1965) Small bowel tonometry: assessment of small gut muco-sal oxygen tension in dog and man. Nature 206: 943–944
Kivisaari J, Niinikoski J (1973) Use of silastic sampling tube and capillary sampling technique in the measurement of tissue PO, and PCO2. Am J Surg 125: 623–627
Fiddian-Green RG, Pittenger G, Whitehouse WM (1982) Back-diffusion of CO2 and its influence on the intramural pH in gastric mucosa. J Surg Res 33: 39–48
Mythen M, Faehnrich J (1996) Monitoring gut perfusion. In: Rombeau JL, Takala J (eds) Gut dysfunction in critical illness. Springer-Verlag, Berlin, pp 233–245
Lebuffe G, Deco ene C, Pol A, Prat A, Vallet B (1999) Regional capnometry with air-automated tonometry detects circulatory failure earlier than conventional hemodynamics after cardiac surgery. Anesth Analg 89: 1084–1090
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2000 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Vallet, B., Tavernier, B., Lund, N. (2000). Assessment of Tissue Oxygenation in the Critically III. In: Vincent, JL. (eds) Yearbook of Intensive Care and Emergency Medicine 2000. Yearbook of Intensive Care and Emergency Medicine, vol 2000. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-13455-9_59
Download citation
DOI: https://doi.org/10.1007/978-3-662-13455-9_59
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-66830-5
Online ISBN: 978-3-662-13455-9
eBook Packages: Springer Book Archive