Summary
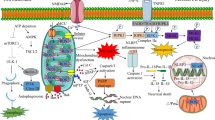
Ischemia leads to mitochondrial dysfunction. This is reversible if ischemia is of brief duration, but secondary mitochondrial damage is observed after longer periods of ischemia, particularly in selectively vulnerable neuronal populations. This damage is triggered during the period of ischemia, but secondary factors lead to its “maturation”. Probably, the most important of these factors is gradual accumulation of calcium in the mitochondria. The mechanisms involved encompass phospholipase A2-mediated breakdown of the lipid skeleton of membranes, and free-radical-mediated oxidation of its lipid and protein components. However, the immediate trigger may be the opening (“assembly”) of a mitochondrial permeability transition (MPT) pore, which is followed by calcium release, collapse of the electrochemical gradient for H+ and a burst of production of reactive oxygen species (ROS). The ensuing bioenergetic compromise and the mitochondrial production of ROS probably constitute important causes of cell death. Recent results suggest that mitochondrial dysfunction is a leading event in the cascade of events that cause apoptotic and necrotic cell death. Very likely, the trigger is the release of cytochrome c from depolarized or otherwise compromised mitochondria. By activating caspase-3, cytochrome c can initiate terminal events encompassing activation of proteases, endonucleases and poly (ADP-ribose) polymerase. Therefore, the type of enzyme activated may determine whether cell death is of the apoptotic or necrotic type.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Ankarcrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, Nicotera BB (1995) Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron 15: 961–973
Beal MF (1992) Does impairment of energy metabolism result in excitotoxic neuronal death in neurodegenerative illnesses? Ann Neurol 31: 119–130
Beal MF, Hyman BT, Koroshetz W (1993) Do defects in mitochondrial energy metabolism underlie the pathology of neurodegenerative diseases? Trends Neurosci 16: 125–131
Bernardi P (1996) The permeability transition pore. Control points of a cyclosporin A-sensitive mitochondrial channel involved in cell death. Biochim Biophys Acta 1275: 5–9
Bernardi P, Broekemeier KM, Pfeiffer DR (1994) Recent progress on regulation of the mitochondrial permeability transition pore; a cyclosporin-sensitive pore in the inner mitochondrial membrane. J Bioenerg Biomembr 26: 509–517
Bonfoco E, Krainc D, Ankarcrona M, Nicotera P, Lipton SA (1995) Apoptosis and necrosis: two distinct events induced, respectively, by mild and intense insults with N-methyl-D-aspartate or nitric oxide/superoxide in cortical cell cultures. Proc Natl Acad Sci U S A 92: 7162–7166
Canevari L, Kuroda S, Bates TE, Clark JB, Siesjö BK (1997) Activity of mitochondrial respiratory chain enzymes after transient focal ischemia in the rat. J Cereb Blood FIow Metab 17: 1166–1169
Crompton M, Costi A (1988) Kinetic evidence for a heart mitochondrial pore activated by Ca2+, inorganic phosphate and oxidative stress. A potential mechanism for mitochondrial dysfunction during cellular Ca2+ overload. Eur J Biochem 178: 489–501
Drake M, Friberg H, Boris Möller F, Sakata K, Wieloch T (1996) The immunosuppressant FK506 ameliorates ischaemic damage in the rat brain. Acta Physiol Scand 158: 155–159
Duchen MR, McGuinness D, Brown LA, Crompton M (1993) On the involvement of a cyclosporin A sensitive mitochondrial pore in myocardial reperfusion injury. Cardiovasc Res 27: 1790–1794
Endres M, Wang ZQ, Namura S, Waeber C, Moskowitz MA (1997) Ischemic brain injury is mediated by the activation of poly(ADP-ribose)polymerase. J Cereb Blood Flow Metab 17: 1143–1151
Folbergrová J, Zhao Q, Katsura K, Siesjö BK (1995) N-tert-butyl-α-phenylnitrone improves recovery of brain energy state in the rats following transient focal ischemia. Proc Nat Acad Sci, USA 92: 5057–5061
Griffiths EJ, Halestrap AP (1995) Mitochondrial non-specific pores remain closed during cardiac ischaemia, but open upon reperfusion. Biochem J 307: 93–98
Gunter TE, Pfeiffer DR (1990) Mechanisms by which mitochondria transport calcium. Am J Physiol 258: C755–C786
Gunter TE, Gunter KK, Sheu SS, Gavin CE (1994) Mitochondria) calcium transport: physiological and pathological relevance. Am J Physiol 267: C313–C339
Haletrap AP, Connern CP, Griffiths EJ, Kerr PM (1997) Cyclosporine A binding to mitochondrial cyclophilin inhibits the permeability transition pore and protects hearts from ischemia/reperfusion injury. Mol Cell Biochem 174: 167–172
Hara H, Friedlander RM, Gagliardini V, Ayata C, Fink K, Huang Z, Shimizu-Sasamata M, Yuan J, Moskowitz MA (1997) Inhibition of interleukin 1β converting enzyme family proteases reduces ischemic and excitotoxic neuronal damage. Proc Natl Acad Sci U S A 94: 2007–2012
Hirsch T, Marchetti P, Susin SA, Dallaporta B, Zamzami N, Marco I, Geuskens M, Kroemer G (1997) The apoptosis-necrosis paradox. Apoptogenic proteases activated after mitochondrial permeability transition determine the mode of cell death. Oncogene 15: 1573–1581
Hockenbery DM, Oltvai ZN, Yin XM, Milliman CL, Korsmeyer SJ (1993) Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 75: 241–251
Kane DJ, Sarafian TA, Anton R, Hahn H, Gralla EB, Valentine JS, Ord T, Bredesen DE (1993) Bcl-2 inhibition of neural death: decreased generation of reactive oxygen species. Science 262: 1274–1277
Kluck RM, Bossy Wetzel E, Green DR, Newmeyer DD (1997) The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275: 1132–1136
Kluck RM, Martin SJ, Hoffman BM, Zhou JS, Green DR, Newmeyer DD (1997) Cytochrome c activation of CPP32-like proteolysis plays a critical role in a Xenopus cell-free apoptosis system. EMBO J 16: 4639–4649
Kristian T, Siesjö BK (1998) Calcium in ischemic cell death. Stroke 29: 705–718
Kuroda S, Siesjö BK (1996) Postischemic administration of FK506 reduced infarct volume following transient brain ischemia. Neurosci Res Com 19: 83–90
Kuroda S, Siesjö BK (1997) Reperfusion damage following focal ischemia: pathophysiology and therapeutic windows. Clin Neurosci 4: 199–212
Kuroda S, Katsura K, Hillered L, Bates TE, Siesjö BK (1996) Delayed treatment with α-phenyl-N-tert-butyl nitrone (PBN) attenuates secondary mitochondrial dysfunction after transient focal cerebral ischemia in the rat. Neurobiol Dis 3: 149–157
Li PA, Uchino H, Elmér E, Siesjö BK (1997) Amelioration by cyclosporin A of brain damage following 5 or 10 min of ischemia in rats subjected to preischemic hyperglycemia. Brain Res 753: 133–140
Liu X, Kim CN, Yang J, Jemmerson R, Wang X (1996) Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell 86: 147–157
Markgraf CG, Velayo NL, Johnson MP, McCarty DR, Medhi S, Koehl JR, Chmielewski PA, Linnik MD (1998) Six-hour window of opportunity for calpain inhibition in focal cerebral ischemia in rats. Stroke 29: 152–158
Nakai A, Kuroda S, Kristián T, Siesjö BK (1997) The immunosuppressant drug FK506 ameliorates secondary mitochondrial dysfunction following transient focal cerebral ischemia in the rat. Neurobiol Dis 4: 288–300
Nicolli A, Basso E, Petronilli V, Wenger RM, Bernardi P (1996) Interactions of cyclophilin with the mitochondrial inner membrane and regulation of the permeability transition pore, a cyclosporin A-sensitive channel. J Biol Chem 271: 2185–2192
Ouyang YB, Kuroda S, Kristián T, Siesjö BK (1997) Release of mitochondrial aspartate aminotransferase (mAST) following transient focal cerebral ischemia suggests the opening of a mitochondrial permeability transition pore. Neurosci Res Commun 20: 167–173
Pike RB, Zhao X, Newcomb JK, Wang KK, Postmantur RM, Hayes RL (1998) Temporal relationships between de novo protein synthesis, calpain and caspase 3-like protease activation, and DNA fragmentation during apoptosis in septo-hippocampal cultures. J Neurosci Res (in press)
Reed JC (1997) Cytochrome c: can’t live with it-can’t live without it. Cell 91: 559–562
Richter C, Gogvadze V, Laffranchi R, Schlapbach R, Schweizer M, Suter M, Walter P, Yaffee M (1995) Oxidants in mitochondria: from physiology to diseases. Biochim Biophys Acta 1271: 67–74
Siesjö BK (1992) Pathophysiology and treatment of focal cerebral ischemia. Part I: Pathophysiology. J Neurosurg 77: 169–184
Siesjö BK, Kristián T, Katsura K (1995) The role of calcium in delayed postischemic brain damage. In: Moskowitz M, Caplan LR (eds) Cerebrovascular diseases. Butterworth-Heinemann, Boston, pp 353–370
Sims NR (1991) Selective impairment of respiration in mitochondria isolated from brain subregions following transient forebrain ischemia in the rat. J Neurochem 56: 1836–1844
Sims NR, Pulsinelli WA (1987) Altered mitochondrial respiration in selectively vulnerable brain subregions following transient forebrain ischemia in the rat. J Neurochem 49: 1367–1374
Steiner JP, Connolly MA, Valentine HL, Hamilton GS, Dawson TM, Hester L, Snyder SH (1997) Neurotrophic actions of nonimmunosuppressive analogues of immunosuppressive drugs FK506, rapamycin and cyclosporin A. Nat Med 3: 421–428
Takahashi A, Goldschmidt Clermont PJ, Alnemri ES, Fernandes Alnemri T, Yoshizawa Kumagaya K, Nakajima K, Sasada M, Poirier GG, Earnshaw WC (1997) Inhibition of ICE-related proteases (caspases) and nuclear apoptosis by phenylarsine oxide. Exp Cell Res 231: 123–131
Uchino H, Elmér E, Uchino K, Lindvall O, Siesjö BK (1995) Cyclosporin A dramatically ameliorates CA1 hippocampal damage following transient forebrain ischaemia in the rat. Acta Physiol Scand 155: 469–471
Yang J, Liu X, Bhalla K, Kim CN, Ibrado AM, Cai J, Peng TI, Jones DP, Wang X (1997) Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275: 1129–1132
Zamzami N, Marchetti P, Castedo M, Decaudin D, Macho A, Hirsch T, Susin SA, Petit PX, Mignotte B, Kroemer G (1995) Sequential reduction of mitochondrial transmembrane potential and generation of reactive oxygen species in early programmed cell death. J Exp Med 182: 367–377
Zamzami N, Marchetti P, Castedo M, Hirsch T, Susin SA, Masse B, Kroemer G (1996) Inhibitors of permeability transition interfere with the disruption of the mitochondrial transmembrane potential during apoptosis. FEBS Lett 384: 53–57
Zamzami N, Hirsch T, Dallaporta B, Petit PX, Kroemer G (1997) Mitochondrial implication in accidental and programmed cell death: apoptosis and necrosis. J Bioenerg Biomembr 29: 185–193
Zhao Q, Pahlmark K, Smith ML, Siesjö BK (1994) Delayed treatment with the spin trap α-phenyl-N-tert-butyl nitrone (PBN) reduces infarct size following transient middle cerebral artery occlusion in rats. Acta Physiol Scand 152: 349–350
Zoratti M, Szabò I (1995) The mitochondrial permeability transition. Biochim Biophys Acta 1241: 139–176
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1999 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Siesjö, B.K., Ouyang, Y., Kristián, T., Elmér, E., Li, PA., Uchino, H. (1999). Role of Mitochondria in Immediate and Delayed Reperfusion Damage. In: Ito, U., Fieschi, C., Orzi, F., Kuroiwa, T., Klatzo, I. (eds) Maturation Phenomenon in Cerebral Ischemia III. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-58602-6_26
Download citation
DOI: https://doi.org/10.1007/978-3-642-58602-6_26
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-65023-2
Online ISBN: 978-3-642-58602-6
eBook Packages: Springer Book Archive