Abstract
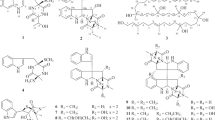
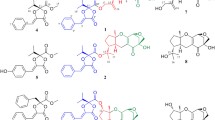
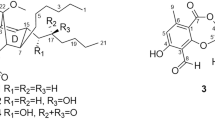
The dimeric epipolythiodiketopiperazine alkaloids are a family of fungal metabolites renowned for their structural and functional complexity [1]. Isolated across eight genera of fungi, over 40 members of this class of natural products have been discovered since the isolation of their inaugural members, (+)-chaetocin A (2) [2] and (+)-verticillin A [3], in 1970 (Fig. 9.1). As evidenced by the large number of constituents comprising this family, there are many avenues by which each member derives its identity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Notes
- 1.
We acknowledge financial support by NIH-NIGMS (GM089732).
References
(a) Waring P, Eichner RD, Mullbacher A (1988) Med Res Rev 8:499; (b) Waring P, Beaver J (1996) Gen Pharmacol 27:1311; (c) Gardiner DM, Waring P, Howlett BJ (2005) Microbiology 151:1021; (d) Patron NJ, Waller RF, Cozijnsen AJ, Straney DC, Gardiner DM, Nierman WC, Howlett BJ (2007) BMC Evol Biol 7:174; (e) Huang R, Zhou X, Xu T, Yang X, Liu Y (2010) Chem Biodivers 7:2809; (f) Iwasa E, Hamashima Y, Sodeoka M (2011) Isr J Chem 51:420
Hauser D, Weber HP, Sigg HP (1970) Helv Chim Acta 53:1061
Katagiri K, Sato K, Hayakawa S, Matsushima T, Minato H (1970) J Antibiot (Tokyo) 23:420
Son BW, Jensen PR, Kauffman CA, Fenical W (1999) Nat Prod Res 13:213
Fukuyama T, Nakatsuka S-I, Kishi Y (1981) Tetrahedron 37:2045
(a) Hino T, Nakagawa M (1989) In: Brossi A (ed) The alkaloids: chemistry and pharmacology, vol 34, Academic, New York, NY, pp 1–75; (b) Anthoni U, Christopherson C, Nielsen PH (1999) In: Pelletier SW (ed) Alkaloids: chemical and biological perspectives, vol 13, Pergamon, New York, NY pp 163–236
Movassaghi M, Schmidt MA (2007) Angew Chem Int Ed 46:3725
Movassaghi M, Schmidt MA, Ashenhurst JA (2008) Angew Chem Int Ed 47:1485
(a) Hendrickson JB, Rees R, Goschke R (1962) Proc Chem Soc:383; (b) Hino T, Yamada S (1963) Tetrahedron Lett 4:1757; (c) Scott AI, McCapra F, Hall ES (1964) J Am Chem Soc 86:302; (d) Nakagawa M, Sugumi H, Kodato S, Hino T (1981) Tetrahedron Lett 22:5323; (e) Fang C-L, Horne S, Taylor N, Rodrigo R (1994) J Am Chem Soc 116:9480; (f) Link JT, Overman LE (1996) J Am Chem Soc 118:8166; (g) Somei M, Osikiri N, Hasegawa M, Yamada F (1999) Heterocycles 51:1237; (h) Overman LE, Paone DV, Stearns BA (1999) J Am Chem Soc 121:7702; (i) Overman LE, Larrow JF, Stearns BA, Vance JM (2000) Angew Chem Int Ed 39:213; (j) Ishikawa H, Takayama H, Aimi N (2002) Tetrahedron Lett 43:5637; (k) Matsuda Y, Kitajima M, Takayama H (2005) Heterocycles 65:1031; (l) Verotta L, Orsini F, Sbacchi M, Scheidler MA, Amador TA, Elisabetsky E (2002) Bioorg Med Chem 10:2133
(a) Zhang Y-X, Chen Y, Guo X-N, Zhang X-W, Zhao W-M, Zhong L, Zhou J, ** Y, Lin L-P, Ding J (2005) Anti-Cancer Drug 16:515; (b) Chen Y, Zhang Y-X, Li M-H, Zhao W-M, Shi Y-H, Miao Z-H, Zhang X-W, Lin L-P, Ding (2005) J Biochem Biophys Res Commun 329:1334; (c) Chen Y, Miao Z-H, Zhao W-M, Ding J (2005) FEBS Lett 579:3683
Saito T, Suzuki Y, Koyama K, Natori S, Iitaka Y, Knoshita T (1998) Chem Pharm Bull 36:1942
(a) Greiner D, Bonaldi T, Eskeland R, Roemer E, Imhof A (2005) Nat Chem Biol 1:143; (b) Isham CR, Tibodeau JD, ** W, Timm MM, Bible KC (2007) Blood 109:2579; (c) Cherrier T, Suzanne S, Redel L, Calao M, Marban C, Samah B, Mukerjee R, Schwartz C, Gras G, Sawaya BE, Zeichner SL, Aunis D, Van Lint C, Rohr O (2009) Oncogene 28:3380; (d) Cook KM, Hilton ST, Mecinovic J, Motherwell WB, Figg WD, Schofield CJ (2009) J Bio Chem 284:26831; (e) Lakshmikuttyamma A, Scott SA, DeCoteau JF, Geyer CR (2010) Oncogene 29:576
Kwon-Chung KJ, Sugui JA (2009) Med Mycol 47:S97
(a) Munday R (1982) Chem Biol Interact 41:361; (b) Munday R (1987) J Appl Toxicol 7:17; (c) Chai CLL, Heath GA, Buleatt PB, O’Shea GA (1999) J Chem Soc Perk T 2:389; (d) Chai CLL, Waring P (2000) Redox Rep 5:257; (e) Scharf DH, Remme N, Heinekamp T, Hortschansky P, Brakhage AA, Hertweck C (2010) J Am Chem Soc 132:10136
Bernardo PH, Chai CLL, Deeble GJ, Liu X-M, Waring P (2001) Bioorg Med Chem Lett 11:483
Waring P, Mamchak A, Khan T, Sjaarda A, Sutton P (1995) Cell Death Differ 2:201
Waring P, Sjaarda A, Lin QH (1995) Biochem Pharmacol 49:1195
Hurne AM, Chai CLL, Waring P (2000) J Biol Chem 275:25202
Block KM, Wang H, Szabo LZ, Polaske NW, Henchey LK, Dubey R, Kushal S, Lazlo CF, Makhoul J, Song Z, Meuillet EJ, Olenyuk BZ (2009) J Am Chem Soc 131:18078.
(a) Jordan TW, Pedersen JS (1986) J Cell Sci 85:33; (b) Bernardo PH, Brasch N, Chai CLL, Waring P (2003) J Biol Chem 278:46549; (c) Orr JG, Leel V, Caermon GA, Marek CJ, Haughton EL, Erick LJ, Trim JE, Hawksworth GM, Halestrap AP, Wright MC (2004) Hepatology 40:232
Kirby GW, Robins DJ (1980) In: Steyn PS (ed) The biosynthesis of mycotoxins: a study in secondary metabolism, Academic, New York, NY, p 301
Sammes PG (1975) In: Herz W, Grisebach H, Kirby GW (ed) Progress in the chemistry of organic natural products, Springer, New York, NY, p 51
Taylor A (1971) In: Kadis S, Ciegler A (ed) Microbial toxins, Academic, New York, NY, p 337
(a) Fox EM, Howlett BJ (2008) Mycol Res 112:162; (b) Gardiner DM, Howlett BJ (2005) FEMS Microbiol Lett 248:241
Kim J, Ashenhurst JA, Movassaghi M (2009) Science 324:238
Kim J, Movassaghi M (2009) Chem Soc Rev 38:3035
Scharf DH, Remme N, Habel A, Chankhamjon P, Scherlach K, Heinekamp T, Hortschansky P, Brakhage AA, Hertweck C (2011) J Am Chem Soc 133:12322
Eccles GR (1888) Proc Am Pharm Assoc 84:382
Woodward RB, Yang NC, Katz TJ, Clark VM, Harley-Mason J, Ingleby RFJ, Sheppard N (1960) Proc Chem Soc:76
Robinson R, Teuber HJ (1954) Chem Ind:783
Crich D, Banerjee A (2007) Acc Chem Res 40:151
(a) Marsden SP, Depew KM, Danishefsky SJ (1994) J Am Chem Soc 116:11143; (b) Depew KM, Marsden SP, Zatorska D, Zatorski A, Bornmann WG, Danishefsky SJ (1999) J Am Chem Soc 121:11953; (c) Schkeryantz JM, Woo JCG, Siliphaivanh P, Depew KM, Danishefsky SJ (1999) J Am Chem Soc 121:11964
Bruncko M, Crich D, Samy R (1994) J Org Chem 59:5543
Trown PW (1968) Biochem Biophys Res Commun 33:402
Poisel H, Schmidt U (1971) Chem Ber 104:1714
(a) Kishi Y, Fukuyama T, Nakatsuka S (1973) J Am Chem Soc 95:6490; (b) Kishi Y, Fukuyama T, Nakatsuka S (1973) J Am Chem Soc 95:6492l; (c) Kishi Y, Nakatsuka S, Fukuyama T, Havel M (1973) J Am Chem Soc 95:6493; (d) Kishi Y, Fukuyama T (1976) J Am Chem Soc 98:6723; (e) Fukuyama T, Nakatsuka S, Kishi Y (1976) Tetrahedron Lett 38:3393
(a) Strunz GM, Kakushima M (1974) Experientia 30:719; (b) Srinivasan A, Kolar AJ, Olsen RK (1981) J Heterocycl Chem 18:1545; (c) Shimazaki N, Shima I, Hemmi K, Tsurumi Y, Hashimoto M (1987) Chem Pharm Bull 35:3527; (d) Wu Z, Williams LJ, Danishefsky SJ (2000) Angew Chem Int Ed 39:3866
Hino T, Sato T (1971) Tetrahedron Lett 12:3127
Williams RM, Rastetter WH (1980) J Org Chem 45:2625
Coffen DL, Katonak DA, Nelson NR, Sancilio FD (1977) J Org Chem 42:948
Ohler E, Poisel H, Tatruch F, Schmidt U (1972) Chem Ber 105:635
Ohler E, Tataruch F, Schmidt U (1973) Chem Ber 106:396
Yoshimura J, Nakamura H, Matsunari K, Sugiyama Y (1974) Chem Lett:559
Ottenheijm HCJ, Kerkhoff GPC, Bijen JWHA (1975) J Chem Soc Chem Comm:768
Overman LE, Sato T (2007) Org Lett 9:5267
(a) Springer JP, Buchi G, Kobbe B, Demain AL, Clardy J (1977) Tetrahedron Lett 18:2403; (b) Barrow CJ, Cai P, Snyder JK, Sedlock DM, Sun HH, Cooper R (1993) J Org Chem 58:6016
Seebach D, Boes M, Naef R, Schweizer WB (1983) J Am Chem Soc 105:5390
(a) Aresta M, Rossi M, Sacco A (1969) Inorg Chim Acta 3:227; (b) Yamada Y, Momose D-I (1981) Chem Lett:1277; (c) Bagal SK, Adlington RM, Baldwin JE, Marquez R (2004) J OrgChem 69:9100
Rauk A, Yu D, Taylor J, Shustov GV, Block DA, Armstrong DA (1999) Biochemistry 38:9089
Sala T, Sargent MV (1978) J Chem Soc Chem Commun:253
Firouzabadi H, Vessal B, Naderi M (1982) Tetrahedron Lett 23:1847
Gardner KA, Mayer JM (1995) Science 269:1849
Strassner T, Houk KN (2000) J Am Chem Soc 122:7821
Ruble JC, Fu GC (1998) J Am Chem Soc 120:11532
Woodward RB, Brutcher FV (1958) J Am Chem Soc 80:209
For prior epitrithiodiketopiperazine syntheses, see: (a) Brewer D, Rahman R, Safe S, Taylor A (1968) Chem Commun 24:1571; (b) Murdock KC, Angier RB (1970) Chem Comm:55; (c) Safe S, Taylor A (1970) J Chem Soc C 3:432; (d) Ref. [39]; (e) Ohler E, Tatruch F, Schmidt U (1972) Chem Ber 105:3658; (f) Ottenheijm HCJ, Herscheid JDM, Kerkhoff GPC, Spande TF (1976) J Org Chem 41:3433; (g) Curtis PJ, Greatbanks D, Hesp B, Cameron AF, Freer AA (1977) J Chem Soc Perk T I 2:180; (h) Coffen DL, Katonak DA, Nelson NR, Sancilio FD (1977) J Org Chem 42:948; (i) Kirby GW, Rao GV, Robins DJ, Stark WM (1986) Tetrahedron Lett 27:5539; For prior epitetrathiodiketopiperazine syntheses, see: (j) Refs. [39, 61b, c, 61g–i]; (k) Poisel H, Schmidt U (1971) Angew Chem Int Ed 10:130; (l) Yoshimura J, Sugiyama Y, Matsunari K, Nakamura H (1974) Bull Chem Soc Jpn 47:1215; (m) Strunz GM, Kakushima M, Stillwell MA (1975) Can J Chem 53:295; (n) Kirby GW, Robins DJ, Stark M (1983) J Chem Soc Chem Commun:812; (o) Waring P, Eichner RD, Tiwari-Palni U, Mullbacher A (1987) Aust J Chem 40:991
Kim J, Movassaghi M (2010) J Am Chem Soc 132:14376
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Kim, J., Movassaghi, M. (2012). (+)-11,11′-Dideoxyverticillin A. In: Li, J., Corey, E. (eds) Total Synthesis of Natural Products. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-34065-9_9
Download citation
DOI: https://doi.org/10.1007/978-3-642-34065-9_9
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-34064-2
Online ISBN: 978-3-642-34065-9
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)