Abstract
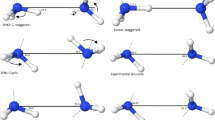
Behavior of shared proton in symmetric dimers of ammonia and lower amine homologs were studied by several theoretical methods. Corresponding optimized structures by density functional theory show an intuitive hypsochromic shift as the degree of methylation is enhanced. Inclusion of nuclear quantum effect, however, changes the whole picture. It was found out that the fundamental vibrational transition corresponding to the shared proton’s stretching motion, νsp is counter intuitive. Based from these calculations, there is a bathochromic shift from ammonia to trimethylamine. These ramifications do clearly indicate that proton is a quantum object. Furthermore, spectroscopic features for the stretching modes of the shared proton and H-bond donor-acceptor atoms were proposed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Cleland WW, Kreevoy MM (1994) Science 264:1887
Voet D, Voet J (2004) Biochemistry, 3rd edn. Wiley, New York
Baker EN (2006) Int Tables Crystallogr F:546–552
Graham JD, Buytendyk AM, Wang D, Bowen KH, Collins KD (2013) Biochemistry 53:344
DeCoursey TE (2003) Physiol Rev 83:475
Garczarek F, Gerwert K (2006) Nature 439:109
Lee J, Kim CH, Joo T (2013) J Phys Chem A 117:1400–1405
Pimentel GC, McClellan AL (1960) The hydrogen bond. Freeman and Company, San Francisco
Hilbert GE, Wulf OR, Hendricks SB, Liddel U (1936) J Am Chem Soc 58:548–555
Ellis JW (1939) J Am Chem Soc 51:1384
Schwarz HA (1980) J Chem Phys 72:284–287
Price JM, Crofton MW, Lee YT (1989) J Chem Phys 91:2749–2751
Price JM, Crofton MW, Lee YT (1989) J Chem Phys 95:2182–2195
Roscioli JR, McCunn LR, Johnson MA (2007) Science 316:249–254
Scheiner S, Harding LB (1981) J Am Chem Soc 103:2169–2173
Asada T, Haraguchi H, Kitaura K (2001) J Phys Chem A 105:7423–7428
Yang Y, Kühn O (2011) Chem Phys Lett 505:1–4
Asmis KR, Yang Y, Santambrogio G, Brümmer M, Roscioli JR, McCunn LR, Johnson MA, Kühn O (2007) Angew Chem Int Ed 46:8691–9694
Yang Y (2008) PhD Dissertation, Freie Universität Berlin
Ishibashi H et al (2008) Chem Phys Chem 9:383
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Jr.,Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian, Inc., Wallingford. Gaussian 09, Revision A.02
Tuckerman ME, Marx D, Klein ML, Parrinello (1996) J Chem Phys 104:5579
Marx D, Tuckerman ME, Hutter J, Parrinello M (1999) Nature 397:601
Grotendorst J, Marx D, Muramatsu A (2002) Path integration via molecular dynamics. In: Von Neumann J (ed) Quantum simulations of complex many-body systems: from theory to algorithms, vol 10. Institute for Computing, Julich, p 269
Simons J, Nichols J (1997) Quantum mechanics in chemistry. Oxford University Press, New York
Ratner MA, Schatz GC (2000) Introduction to quantum mechanics in chemistry. Prentice Hall, Englewood Cliffs
Acknowledgments
This work was financially supported by the Academia Sinica and the National Science Council (NSC101-2113-M-001-023-MY3) of Taiwan. Computational resources are supported in part by the National Center for High Performance Computing. We wish to thank contributions from Linda Shen and Tzu-Chien Wang at the early stage of this project. Fruitful discussions with Prof. Yonggang Yang and Prof. Masanori Tachikawa are also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this paper
Cite this paper
Tan, J.A., Li, JW., Kuo, JL. (2015). Proton Quantum Confinement on Symmetric Dimers of Ammonia and Lower Amine Homologs. In: Nascimento, M., Maruani, J., Brändas, E., Delgado-Barrio, G. (eds) Frontiers in Quantum Methods and Applications in Chemistry and Physics. Progress in Theoretical Chemistry and Physics, vol 29. Springer, Cham. https://doi.org/10.1007/978-3-319-14397-2_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-14397-2_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-14396-5
Online ISBN: 978-3-319-14397-2
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)