Abstract
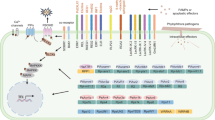
During the co-evolutionary arms race between plants and pathogens, plants evolved a sophisticated defense system to ward off their enemies. In this plant immune system , plant receptor proteins recognize non-self molecules of microbial origin, which leads to the activation of a basal level of disease resistance. The onset of these local plant immune reactions often triggers a systemic acquired resistance (SAR) in tissues distal from the site of infection. Beneficial microbes in the rhizosphere microbiome stimulate a phenotypically similar induced systemic resistance (ISR) that, like SAR, is effective against a broad spectrum of pathogens. There are differences and similarities in the SAR and ISR signaling pathways. The plant defense hormone salicylic acid is a major regulator of SAR, whereas jasmonic acid and ethylene play important roles in ISR. Priming of systemic tissue to express an accelerated defense response upon attack by a pathogen is a common phenomenon in both SAR and ISR. This chapter will outline the current concept of the plant immune system, with special emphasis on mechanisms of systemically induced disease resistance and priming for enhanced defense.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Alström S (1991) Induction of disease resistance in common bean susceptible to halo blight bacterial pathogen after seed bacterization with rhizosphere pseudomonads. J Gen Appl Microbiol 37:495–501
Bakker PAHM, Ran LX, Pieterse CMJ et al (2003) Understanding the involvement of rhizobacteria-mediated induction of systemic resistance in biocontrol of plant diseases. Can J Plant Pathol 25:5–9
Beckers GJM, Jaskiewicz M, Liu Y et al (2009) Mitogen-activated protein kinases 3 and 6 are required for full priming of stress responses in Arabidopsis thaliana. Plant Cell 21:944–953
Berendsen RL, Pieterse CMJ, Bakker PAHM (2012) The rhizosphere microbiome and plant health. Trends Plant Sci 17:478–486
Boller T, Felix G (2009) A renaissance of elicitors: perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu Rev Plant Biol 60:379–406
Champigny M, Shearer H, Mohammad A et al (2011) Localization of DIR1 at the tissue, cellular and subcellular levels during systemic acquired resistance in Arabidopsis using DIR1:GUS and DIR1:EGFP reporters. BMC Plant Biol 11:125
Conrath U (2011) Molecular aspects of defence priming. Trends Plant Sci 16:524–531
De Vleesschauwer D, Höfte M (2009) Rhizobacteria-induced systemic resistance. In: Van Loon LC (ed) Plant Innate Immunity, vol 51. Advances in botanical research. Academic Press Ltd-Elsevier Science Ltd, London, pp 223–281
Dempsey DA, Klessig DF (2012) SOS—too many signals for systemic acquired resistance? Trends Plant Sci 17:538–545
Dodds PN, Rathjen JP (2010) Plant immunity: towards an integrated view of plant-pathogen interactions. Nat Rev Genet 11:539–548
Frost CJ, Mescher MC, Carlson JE et al (2008) Plant defense priming against herbivores: getting ready for a different battle. Plant Physiol 146:818–824
Fu ZQ, Dong X (2013) Systemic acquired resistance: turning local infection into global defense. Annu Rev Plant Biol 64:839–863
Fu ZQ, Yan S, Saleh A et al (2012) NPR3 and NPR4 are receptors for the immune signal salicylic acid in plants. Nature 486:228–232
Hoffland E, Pieterse CMJ, Bik L et al (1995) Induced systemic resistance in radish is not associated with accumulation of pathogenesis-related proteins. Physiol Mol Plant Pathol 46:309–320
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66
Jaskiewicz M, Conrath U, Peterhansel C (2011) Chromatin modification acts as a memory for systemic acquired resistance in the plant stress response. EMBO Rep 12:50–55
Jones JDG, Dangl JL (2006) The plant immune system. Nature 444:323–329
Kachroo A, Robin GP (2013) Systemic signaling during plant defense. Curr Opin Plant Biol 16:527–533
Lee B, Farag MA, Park HB et al (2012) Induced resistance by a long-chain bacterial volatile: elicitation of plant systemic defense by a C13 volatile produced by Paenibacillus polymyxa. PLoS ONE 7:e48744
Lugtenberg B, Kamilova F (2009) Plant-growth-promoting rhizobacteria. Annu Rev Microbiol 63:541–556
Luna E, Bruce TJA, Roberts MR et al (2012) Next-generation systemic acquired resistance. Plant Physiol 158:844–853
Luna E, Van Hulten M, Zhang Y et al (2014) Plant perception of β-aminobutyric acid is mediated by an aspartyl-tRNA synthetase. Nat Chem Biol 10:450–456
Maldonado AM, Doerner P, Dixon RA et al (2002) A putative lipid transfer protein involved in systemic resistance signalling in Arabidopsis. Nature 419:399–403
Mendes R, Kruijt M, De Bruijn I et al (2011) Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 332:1097–1100
Mishina TE, Zeier J (2006) The Arabidopsis flavin-dependent monooxygenase FMO1 is an essential component of biologically induced systemic acquired resistance. Plant Physiol 141:1666–1675
Pastor V, Luna E, Mauch-Mani B et al (2013) Primed plants do not forget. Environ Exp Bot 94:46–56
Pel MJC, Pieterse CMJ (2013) Microbial recognition and evasion of host immunity. J Exp Bot 64:1237–1248
Pieterse CMJ, Van Wees SCM, Hoffland E et al (1996) Systemic resistance in Arabidopsis induced by biocontrol bacteria is independent of salicylic acid accumulation and pathogenesis-related gene expression. Plant Cell 8:1225–1237
Pieterse CMJ, Van Wees SCM, Van Pelt JA et al (1998) A novel signaling pathway controlling induced systemic resistance in Arabidopsis. Plant Cell 10:1571–1580
Pieterse CMJ, Van der Does D, Zamioudis C et al (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Biol 28:489–521
Pieterse CMJ, Zamioudis C, Berendsen RL et al (2014) Induced systemic resistance by beneficial microbes. Annu Rev Phytopathol 52:347–375
Pozo MJ, Van der Ent S, Van Loon LC et al (2008) Transcription factor MYC2 is involved in priming for enhanced defense during rhizobacteria-induced systemic resistance in Arabidopsis thaliana. New Phytol 180:511–523
Rasmann S, De Vos M, Casteel CL et al (2012) Herbivory in the previous generation primes plants for enhanced insect resistance. Plant Physiol 158:854–863
Robert-Seilaniantz A, Grant M, Jones JDG (2011) Hormone crosstalk in plant disease and defense: more than just jasmonate-salicylate antagonism. Annu Rev Phytopathol 49:317–343
Ross AF (1961) Systemic acquired resistance induced by localized virus infections in plants. Virology 14:340–358
Ryu C-M, Farag MA, Hu CH et al (2004) Bacterial volatiles induce systemic resistance in Arabidopsis. Plant Physiol 134:1017–1026
Segarra G, Van der Ent S, Trillas I et al (2009) MYB72, a node of convergence in induced systemic resistance triggered by a fungal and a bacterial beneficial microbe. Plant Biol 11:90–96
Shah J, Zeier J (2013) Long-distance communication and signal amplification in systemic acquired resistance. Front Plant Sci 4:30
Slaughter A, Daniel X, Flors V et al (2012) Descendants of primed Arabidopsis plants exhibit resistance to biotic stress. Plant Physiol 158:835–843
Spoel SH, Koornneef A, Claessens SMC et al (2003) NPR1 modulates cross-talk between salicylate- and jasmonate-dependent defense pathways through a novel function in the cytosol. Plant Cell 15:760–770
Spoel SH, Mou ZL, Tada Y et al (2009) Proteasome-mediated turnover of the transcription coactivator NPR1 plays dual roles in regulating plant immunity. Cell 137:860–872
Stein E, Molitor A, Kogel KH et al (2008) Systemic resistance in Arabidopsis conferred by the mycorrhizal fungus Piriformospora indica requires jasmonic acid signaling and the cytoplasmic function of NPR1. Plant Cell Physiol 49:1747–1751
Van de Mortel JE, De Vos RCH, Dekkers E et al (2012) Metabolic and transcriptomic changes induced in Arabidopsis by the rhizobacterium Pseudomonas fluorescens SS101. Plant Physiol 160:2173–2188
Van der Ent S, Verhagen BWM, Van Doorn R et al (2008) MYB72 is required in early signaling steps of rhizobacteria-induced systemic resistance in Arabidopsis. Plant Physiol 146:1293–1304
Van der Ent S, Van Hulten MHA, Pozo MJ et al (2009) Priming of plant innate immunity by rhizobacteria and ß-aminobutyric acid: differences and similarities in regulation. New Phytol 183:419–431
Van Hulten M, Pelser M, Van Loon LC et al (2006) Costs and benefits of priming for defense in Arabidopsis. Proc Natl Acad Sci U S A 103:5602–5607
Van Loon LC, Bakker PAHM (2005) Induced systemic resistance as a mechanism of disease suppression by rhizobacteria. In: Siddiqui ZA (ed) PGPR: biocontrol and biofertilization. Springer, Dordrecht, pp 39–66
Van Loon LC, Rep M, Pieterse CMJ (2006) Significance of inducible defense-related proteins in infected plants. Annu Rev Phytopathol 44:135–162
Van Oosten VR, Bodenhausen N, Reymond P et al (2008) Differential effectiveness of microbially induced resistance against herbivorous insects in Arabidopsis. Mol Plant-Microbe Interact 21:919–930
Van Peer R, Niemann GJ, Schippers B (1991) Induced resistance and phytoalexin accumulation in biological control of fusarium wilt of carnation by Pseudomonas sp. strain WCS417r. Phytopathology 81:728–734
Van Wees SCM, Luijendijk M, Smoorenburg I et al (1999) Rhizobacteria-mediated induced systemic resistance (ISR) in Arabidopsis is not associated with a direct effect on expression of known defense-related genes but stimulates the expression of the jasmonate-inducible gene Atvsp upon challenge. Plant Mol Biol 41:537–549
Van Wees SCM, De Swart EAM, Van Pelt JA et al (2000) Enhancement of induced disease resistance by simultaneous activation of salicylate- and jasmonate-dependent defense pathways in Arabidopsis thaliana. Proc Natl Acad Sci U S A 97:8711–8716
Van Wees SCM, Van der Ent S, Pieterse CMJ (2008) Plant immune responses triggered by beneficial microbes. Curr Opin Plant Biol 11:443–448
Verhagen BWM, Glazebrook J, Zhu T et al (2004) The transcriptome of rhizobacteria-induced systemic resistance in Arabidopsis. Mol Plant-Microbe Interact 17:895–908
Vernooij B, Friedrich L, Morse A et al (1994) Salicylic acid is not the translocated signal responsible for inducing systemic acquired resistance but is required in signal transduction. Plant Cell 6:959–965
Vlot AC, Dempsey DA, Klessig DF (2009) Salicylic acid, a multifaceted hormone to combat disease. Annu Rev Phytopathol 47:177–206
Vos IA, Pieterse CMJ, Van Wees SCM (2013) Costs and benefits of hormone-regulated plant defences. Plant Pathol 62:43–55
Walters DR, Paterson L, Walsh DJ et al (2008) Priming for plant defense in barley provides benefits only under high disease pressure. Physiol Mol Plant Pathol 73:95–100
Walters DR, Ratsep J, Havis ND (2013) Controlling crop diseases using induced resistance: challenges for the future. J Exp Bot 64:1263–1280
Wei G, Kloepper JW, Tuzun S (1991) Induction of systemic resistance of cucumber to Colletrotichum orbiculare by select strains of plant-growth promoting rhizobacteria. Phytopathology 81:1508–1512
Wu Y, Zhang D, Chu JY et al (2012) The Arabidopsis NPR1 protein is a receptor for the plant defense hormone salicylic acid. Cell Rep 1:639–647
Zamioudis C, Pieterse CMJ (2012) Modulation of host immunity by beneficial microbes. Mol Plant-Microbe Interact 25:139–150
Zamioudis C, Hanson J, Pieterse CMJ (2014a) β-Glucosidase BGLU42 is a MYB72-dependent key regulator of rhizobacteria-induced systemic resistance and modulates iron uptake responses in Arabidopsis roots. New Phytol 204:368–379
Zamioudis C, Korteland J, Van Pelt JA et al (2014b) Root bacteria stimulate iron uptake in plants via a novel photosynthesis-dependent iron sensing system. Submitted
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Pieterse, C., Van Wees, S. (2015). Induced Disease Resistance. In: Lugtenberg, B. (eds) Principles of Plant-Microbe Interactions. Springer, Cham. https://doi.org/10.1007/978-3-319-08575-3_14
Download citation
DOI: https://doi.org/10.1007/978-3-319-08575-3_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-08574-6
Online ISBN: 978-3-319-08575-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)