Abstract
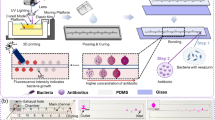
According to the World Health Organization (WHO) forecasts, AntiMicrobial Resistance (AMR) will represent the leading cause of death worldwide in the next decades. To prevent this phenomenon, rapid antimicrobial susceptibility testing is needed to guide the choice of the proper antibiotic. In this context, we propose a chip-scale system, mainly based on a microfluidic channel combined with a pattern of engineered electrodes, to efficiently test an antibiotic on a bacteria sample. The use of dielectrophoretic (DEP) forces enable the sorting of live/dead bacteria, such as Escherichia Coli, with an efficiency larger than 99% for rapid monitoring of the antimicrobial susceptibility at the single-bacterium level.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Brunetti, G., Conteduca, D., Armenise, M.N., Ciminelli, C.: Novel micro-nano optoelectronic biosensor for label-free real-time biofilm monitoring. Biosensors 11(10), 361 (2021)
Stewart, P.S., Costerton, J.W.: Antibiotic resistance of bacteria in biofilm. Lancet 358, 135–138 (2001)
Andersson, D.I., Hughes, D.: Antibiotic resistance and its cost: is it possible to reverse resistance? Nat. Rev. Microbiol. 8, 260–271 (2010)
Chang, H.H., Cohe, T., Grad, Y.H., Hanage, W.P., O’Brien, T.F., Lipsitch, M.: Origin and proligeration of multiple-drug resistance in bacterial pathogens. Microbiol. Mol. Biol. Rev. 79, 101–116 (2015)
“World Health Statistics 2020, World Health Organization: Geneva, Switzerland, (2020)
“Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations, London, UK (2014)
Arabski, M., et al.: The use of lysozyme modified with fluorescein for the detection of Gram-positive bacteria. Microbiol. Res. 170, 242–247 (2015)
Dickson, R.P., et al.: Enrichment of the lung microbiome with gut bacteria in sepsis and the acute respiratory distress syndrome. Nat. Microbiol. 1, 16113 (2016)
Delcour, A.H.: Outer membrane permeability and antibiotic resistance. Biochim. Biophys. Acta 1794, 808–816 (2009)
David, F., Hebeisen, M., Schade, G., Franco-Lara, E., Di Berardino, M.: Viability and membrane potential analysis of bacillus megaterium cells by impedance flow cytometry. Biotechnol. Bioeng. 109, 483–492 (2012)
Conteduca, D., Brunetti, G., Dell’Olio, F., Armenise, M.N., Krauss, T. F., Ciminelli, C.: Monitoring of individual bacteria using electro-photonic traps. Biomed. Optics Express 10, 3463–3471 (2019)
Petrovszki, D., et al.: An integrated electro-optical biosensor system for rapid, low-cost detection of bacteria. Microelectron. Eng. 239–240, 111523 (2021)
Kim, D., Sonker, M., Ros, A.: Dielectrophoresis: from molecular to micrometer-scale analytes. Anal. Chem. 91, 277−295 (2019)
Rahman, N.A., Ibrahim, F., Yafouz, B.: Dielectrophoresis for biomedical sciences applications: a review. Sensors 17, 449 (2017)
Pethig, R.: Review article—dielectrophoresis: status of the theory, technology, and applications. Biomicrofluidics 4, 022811 (2010)
Bai, W., Zhao, K.S., Asami, K.: Dielectric properties of E. coli cell as simulated by the three-shell spheroidal model. Biophys. Chem. 122, 136–142 (2006)
Subramanian, S., Tolstaya, E.I., Winkler, T., Bentley, W.E., Ghodssi, R.: An integrated microsystem for real-time detection and threshold-activated treatment of bacterial biofilms. ACS Appl. Mater. Interfaces 9, 31362–31371 (2017)
Chung, C.-C., Cheng, F., Chen, H.-M., Kan, H.-C., Yang, W.-H., Chang, H.-C.: Screening of antibiotic susceptibility to β-lactam-induced elongation of gram-negative bacteria based on dielectrophoresis. Anal. Chem. 84, 3347–3354 (2012)
Del Moral-Zamora, B., et al.: Combined dielectrophoretic and impedance system for on-chip controlled bacteria concentration: application to Escherichia coli. Electrophoresis 36, 1405–1413 (2015)
Garcia, P.A., et al.: Intracranial nonthermal irreversible electroporation: in vivo analysis. J. Membr. Biol. 236, 127–136 (2010)
Jen, C.P., Chen, T.W.: Selective trap** of live and dead mammalian cells using insulator-based dielectrophoresis within open-top microstructures. Biomed. Microdevices 11, 597–607 (2009)
Castellarnau, M., Errachid, A., Madrid, C., Juarez, A., Samitier, J.: Dielectrophoresis as a tool to characterize and differentiate isogenic mutants of Escherichia coli. Biophys. J. 91, 3937–3945 (2006)
Lewis, C.L., Craig, C.C., Senecal, A.G.: Mass and density measurements of live and dead gram-negative and gram-positive bacterial populations. Appl. Environ. Microbiol. 80, 3622–3631 (2014)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
di Toma, A., Brunetti, G., Sasanelli, N., Armenise, M.N., Ciminelli, C. (2023). Sorting of Live/dead Escherichia Coli by Means of Dielectrophoresis for Rapid Antimicrobial Susceptibility Testing. In: Berta, R., De Gloria, A. (eds) Applications in Electronics Pervading Industry, Environment and Society. ApplePies 2022. Lecture Notes in Electrical Engineering, vol 1036. Springer, Cham. https://doi.org/10.1007/978-3-031-30333-3_25
Download citation
DOI: https://doi.org/10.1007/978-3-031-30333-3_25
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-30332-6
Online ISBN: 978-3-031-30333-3
eBook Packages: EngineeringEngineering (R0)