Summary
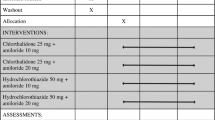
This 6-month multicentre, randomised, double-blind, parallel group clinical trial compared the tolerability and antihypertensive efficacy of a once-daily combination of fosinopril 20mg/hydrochlorothiazide 12.5mg (FOS/HCTZ) with a combination of amiloride 5mg/hydrochlorothiazide 50mg (AMI/HCTZ) in 217 patients with mild to moderate essential hypertension. Adverse events related to hypotension or to specifically targeted clinical laboratory values were observed infrequently with FOS/HCTZ compared with AMI/HCTZ: with FOS/HCTZ, only 4 of 104 patients (3.9%) experienced such events, compared with 16 of 113 (14.1%) in the AMI/HCTZ group (p < 0.001). While statistically significant differences were found between the two treatment groups for changes from baseline in serum potassium, cholesterol, triglyceride and glucose values, the metabolic profile was uniformly unfavourable towards the AMI/HCTZ group; for example, reductions in potassium and elevations in cholesterol, triglyceride and glucose were more pronounced with the AMI/HCTZ group than with the FOS/HCTZ group. Both antihypertensive regimens produced statistically significant reductions from baseline in seated diastolic blood pressure that were equivalent at most points of measurement during double-blind treatment. Therapeutic response rates were high (≥95%) and were similar for both regimens throughout the study. Because the relative risk for adverse events was markedly less over the long term with FOS/HCTZ than with AMI/HCTZ, the combination of fosinopril and hydrochlorothiazide may offer significant tolerability advantages over amiloride plus hydrochlorothiazide for such patients.
Similar content being viewed by others
References
Kaplan NM. Introduction. Drag Invest 1991; 3Suppl. 4: 1–2
Wagstaff AJ, Davis R, McTavish D. Fosinopril. A reappraisal of its pharmacology and therapeutic efficacy in essential hypertension. Drugs 1996; 51(5): 777–91
Pool JL, Cushman WC, Saini RK, et al. Use of the factorial design and quadratic response surface models to evaluate the fosinopril and hydrochlorothiazide combination therapy in hypertension. Am JHypertens 1997; 10: 117–23
Ward TD. The additive effect of fosinopril in patients taking chlorthalidone for the treatment of mild-to-moderate essential hypertension: a multicentre, placebo-controlled, dose-response study. Drag Invest 1991; 3Suppl. 4: 25–31
Fernández M, Madero R, Gonzalez D, et al. Combined versus single effect of fosinopril and hydrochlorothiazide in hypertensive patients. Hypertension 1994; 23 Suppl.: 207–10
Guthrie R, Reggi DR, Plesher MM, et al. Efficacy and safety of fosinopril/hydrochlorothiazide combinations on ambulatory blood pressure profiles in hypertension. Am J Hypertens 1996; 9: 306–11
Weber MA. Overview of fosinopril. Drug Invest 1991; 3Suppl. 4: 3–11
Duchin KL, Kripalani KJ, Marino MR, et al. Review of the pharmacokinetics of fosinopril in special populations. Drug Invest 1991; 3Suppl. 4: 18–24
Weiner IM, Mudge GH. Diuretics and other agents employed in the mobilization of edema fluid. In: Gilman AG, Goodman LS, Rall TW, et al., editors. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 7th ed. New York: MacMillan, 1985: 887–907
Berne C. Metabolic effects of ACE inhibitors. J Intern Med 1991; 229Suppl. 2: 119–25
Kronig B, Flygt G. Felodipine vs. Moduretic®: a double-blind parallel group multicentre study. Drugs 1987; 3: 162–9
Lacourciere Y, Poirier L, Cleroux J, et al. Comparative efficacy of hydrochlorothiazide and amiloride in elderly subjects with ambulatory hypertension. Hypertension 1995; 25(6): 1355
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saini, R., Romanini, M. & Mos, L. Tolerability and Efficacy of Fosinopril and Hydrochlorothiazide Compared with Amiloride and Hydrochlorothiazide in Patients with Mild to Moderate Hypertension. Clin. Drug Investig. 15, 91–99 (1998). https://doi.org/10.2165/00044011-199815020-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-199815020-00003