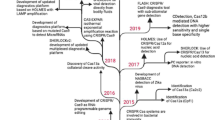
Abstract
Infectious diseases cause great economic loss and individual and even social anguish. Existing detection methods lack sensitivity and specificity, have a poor turnaround time, and are dependent on expensive equipment. In recent years, the clustered regularly interspaced short palindromic repeats (CRISPR)-CRISPR-associated protein (Cas) system has been widely used in the detection of pathogens that cause infectious diseases owing to its high specificity, sensitivity, and speed, and good accessibility. In this review, we discuss the discovery and development of the CRISPR-Cas system, summarize related analysis and interpretation methods, and discuss the existing applications of CRISPR-based detection of infectious pathogens using Cas proteins. We conclude the challenges and prospects of the CRISPR-Cas system in the detection of pathogens.
概要
感染性疾病对个人乃至社会均可造成极大的经济损失和精神痛苦。现有检测方法在灵敏度与特异性、检测时间、昂贵仪器设备依赖方面或多或少存在不足。成簇的规律间隔的短回文重复序列(CRISPR)-相关蛋白(Cas)系统,因其具备特异、灵敏、快速、便捷等优势,**年来被尝试应用于感染性疾病病原体的检测。本文首先回顾了CRISPR-Cas系统的发现发展进程,并列举可与之结合获得检测结果的检测结果阅读方式,之后从检测过程中所应用的不同Cas蛋白的角度简要描述了目前已有的基于CRISRP检测感染性疾病病原体的应用,最后还展望了CRISPR/Cas系统未来在检测方面的应用前景及挑战。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abudayyeh OO, Gootenberg JS, Konermann S, et al., 2016. C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science, 353(6299): aaf5573. https://doi.org/10.1126/science.aaf5573
Ackerman CM, Myhrvold C, Thakku SG, et al., 2020. Massively multiplexed nucleic acid detection with Cas13. Nature, 582(7811):277–282. https://doi.org/10.1038/s41586-020-2279-8
Ali Z, Aman R, Mahas A, et al., 2020. iSCAN: an RT-LAMP-coupled CRISPR-Cas12 module for rapid, sensitive detection of SARS-CoV-2. Virus Res, 288:198129. https://doi.org/10.1016/j.virusres.2020.198129
Anantharaman V, Makarova KS, Burroughs AM, et al., 2013. Comprehensive analysis of the HEPN superfamily: identification of novel roles in intra-genomic conflicts, defense, pathogenesis and RNA processing. Biol Direct, 8:15. https://doi.org/10.1186/1745-6150-8-15
Aquino-Jarquin G, 2019. CRISPR-Cas14 is now part of the artillery for gene editing and molecular diagnostic. Nanomedicine, 18:428–431. https://doi.org/10.1016/j.nano.2019.03.006
Arizti-Sanz J, Freije CA, Stanton AC, et al., 2020. Streamlined inactivation, amplification, and Cas13-based detection of SARS-CoV-2. Nat Commun, 11:5921. https://doi.org/10.1038/s41467-020-19097-x
Aumiller WM, Cakmak FP, Davis BW, et al., 2016. RNA-based coacervates as a model for membraneless organelles: formation, properties, and interfacial liposome assembly. Langmuir, 32(39):10042–10053. https://doi.org/10.1021/acs.langmuir.6b02499
Bhattacharyya RP, Thakku SG, Hung DT, 2018. Harnessing CRISPR effectors for infectious disease diagnostics. ACS Infect Dis, 4(9):1278–1282. https://doi.org/10.1021/acsinfecdis.8b00170
Broughton JP, Deng XD, Yu GX, et al., 2020. CRISPR-Cas12-based detection of SARS-CoV-2. Nat Biotechnol, 38(7): 870–874. https://doi.org/10.1038/s41587-020-0513-4
Cai QQ, Wang R, Qiao ZH, et al., 2021. Single-digit Salmonella detection with the naked eye using bio-barcode immunoassay coupled with recombinase polymerase amplification and a CRISPR-Cas12a system. Analyst, 146(17): 5271–5279. https://doi.org/10.1039/D1AN00717C
Chang YF, Deng Y, Li TY, et al., 2020. Visual detection of porcine reproductive and respiratory syndrome virus using CRISPR-Cas13a. Transbound Emerg Dis, 67(2):564–571. https://doi.org/10.1111/tbed.13368
Chen JS, Ma EB, Harrington LB, et al., 2018. CRISPR-Cas12a target binding unleashes indiscriminate single-stranded DNase activity. Science, 360(6387):436–439. https://doi.org/10.1126/science.aar6245
Dai YF, Liu CC, 2019. Recent advances on electrochemical biosensing strategies toward universal point-of-care systems. Angew Chem Int Ed Engl, 131(36):12483–12496. https://doi.org/10.1002/ange.201901879
Dai YF, Somoza RA, Wang L, et al., 2019. Exploring the trans-cleavage activity of CRISPR-Cas12a (cpf1) for the development of a universal electrochemical biosensor. Angew Chem Int Ed Engl, 131(48):17560–17566. https://doi.org/10.1002/ange.201910772
Ding X, Yin K, Li ZY, et al., 2020. Ultrasensitive and visual detection of SARS-CoV-2 using all-in-one dual CRISPR-Cas12a assay. Nat Commun, 11:4711. https://doi.org/10.1038/s41467-020-18575-6
East-Seletsky A, O’Connell MR, Knight SC, et al., 2016. Two distinct RNase activities of CRISPR-C2c2 enable guide-RNA processing and RNA detection. Nature, 538(7624): 270–273. https://doi.org/10.1038/nature19802
Fozouni P, Son S, de León Derby MD, et al., 2021. Amplification-free detection of SARS-CoV-2 with CRISPR-Cas13a and mobile phone microscopy. Cell, 184(2):323–333.e9. https://doi.org/10.1016/j.cell.2020.12.001
Ge XL, Meng T, Tan X, et al., 2021. Cas14a1-mediated nucleic acid detectifon platform for pathogens. Biosens Bioelectron, 189:113350. https://doi.org/10.1016/j.bios.2021.113350
Gootenberg JS, Abudayyeh OO, Lee JW, et al., 2017. Nucleic acid detection with CRISPR-Cas13a/C2c2. Science, 356(6336):438–442. https://doi.org/10.1126/science.aam9321
Gootenberg JS, Abudayyeh OO, Kellner MJ, et al., 2018. Multiplexed and portable nucleic acid detection platform with Cas13, Cas12a, and Csm6. Science, 360(6387):439–444. https://doi.org/10.1126/science.aaq0179
Guo L, Sun XH, Wang XE, et al., 2020. SARS-CoV-2 detection with CRISPR diagnostics. Cell Discov, 6:34. https://doi.org/10.1038/s41421-020-0174-y
Haft DH, Selengut J, Mongodin EF, et al., 2005. A guild of 45 CRISPR-associated (Cas) protein families and multiple CRISPR/Cas subtypes exist in prokaryotic genomes. PLoS Comput Biol, 1(6):e60. https://doi.org/10.1371/journal.pcbi.0010060
Hajian R, Balderston S, Tran T, et al., 2019. Detection of unamplified target genes via CRISPR-Cas9 immobilized on a graphene field-effect transistor. Nat Biomed Eng, 3(6):427–437. https://doi.org/10.1038/s41551-019-0371-x
Hamm JN, Erdmann S, Eloe-Fadrosh EA, et al., 2019. Unexpected host dependency of Antarctic Nanohaloarchaeota. Proc Natl Acad Sci USA, 116(29):14661–14670. https://doi.org/10.1073/pnas.1905179116
Harrington LB, Burstein D, Chen JS, et al., 2018. Programmed DNA destruction by miniature CRISPR-Cas14 enzymes. Science, 362(6416):839–842. https://doi.org/10.1126/science.aav4294
He Q, Yu DM, Bao MD, et al., 2020. High-throughput and all-solution phase African Swine Fever Virus (ASFV) detection using CRISPR-Cas12a and fluorescence based point-of-care system. Biosens Bioelectron, 154:112068. https://doi.org/10.1016/j.bios.2020.112068
Hou TY, Zeng WQ, Yang ML, et al., 2020. Development and evaluation of a rapid CRISPR-based diagnostic for COVID-19. PLoS Pathog, 16(8):e1008705. https://doi.org/10.1371/journal.ppat.1008705
Huang Y, Gu D, Xue H, et al., 2021. Rapid and accurate Campylobacter jejuni detection with CRISPR-Cas12b based on newly identified Campylobacter jejuni-specific and -conserved genomic signatures. Front Microbiol, 12: 649010. https://doi.org/10.3389/fmicb.2021.649010
Hwang H, Hwang BY, Bueno J, 2018. Biomarkers in infectious diseases. Dis Markers, 2018:8509127. https://doi.org/10.1155/2018/8509127
Hyman AA, Weber CA, Jülicher F, 2014. Liquid-liquid phase separation in biology. Ann Rev Cell Dev Biol, 30:39–58. https://doi.org/10.1146/annurev-cellbio-100913-013325
Ishino Y, Shinagawa H, Makino K, et al., 1987. Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J Bacteriol, 169(12): 5429–5433. https://doi.org/10.1128/jb.169.12.5429-5433.1987
Iwai K, Wwhrs M, Garber M, et al., 2022. Scalable and automated CRISPR-based strain engineering using droplet microfluidics. Microsyst Nanoeng, 8:31. https://doi.org/10.1038/s41378-022-00357-3
Jansen R, van Embden JDA, Gaastra W, et al., 2002. Identification of genes that are associated with DNA repeats in prokaryotes. Mol Microbiol, 43(6):1565–1575. https://doi.org/10.1046/j.1365-2958.2002.02839.x
Joung J, Ladha A, Saito M, et al., 2020. Detection of SARS-CoV-2 with SHERLOCK one-pot testing. N Engl J Med, 383(15): 1492–1494. https://doi.org/10.1056/NEJMc2026172
Karvelis T, Bigelyte G, Young JK, et al., 2020. PAM recognition by miniature CRISPR-Cas12f nucleases triggers programmable double-stranded DNA target cleavage. Nucleic Acids Res, 48(9):5016–5023. https://doi.org/10.1093/nar/gkaa208
Konermann S, Lotfy P, Brideau NJ, et al., 2018. Transcriptome engineering with RNA-targeting type VI-D CRISPR effectors. Cell, 173(3):665–676.e14. https://doi.org/10.1016/j.cell.2018.02.033
Koonin EV, Makarova KS, Zhang F, 2017. Diversity, classification and evolution of CRISPR-Cas systems. Curr Opin Microbiol, 37:67–78. https://doi.org/10.1016/j.mib.2017.05.008
Kostyusheva A, Brezgin S, Babin Y, et al., 2022. CRISPR-Cas systems for diagnosing infectious diseases. Methods, 203: 431–446. https://doi.org/10.1016/j.ymeth.2021.04.007
Leung RKK, Cheng QX, Wu ZL, et al., 2022. CRISPR-Cas12-based nucleic acids detection systems. Methods, 203: 276–281. https://doi.org/10.1016/j.ymeth.2021.02.018
Li F, Ye QH, Chen MT, et al., 2021a. Cas12aFDet: a CRISPR/Cas12a-based fluorescence platform for sensitive and specific detection of Listeria monocytogenes serotype 4c. Anal Chim Acta, 1151:338248. https://doi.org/10.1016/j.aca.2021.338248
Li F, Ye QH, Chen MT, et al., 2021b. An ultrasensitive CRISPR/Cas12a based electrochemical biosensor for Listeria monocytogenes detection. Biosens Bioelectron, 179:113073. https://doi.org/10.1016/j.bios.2021.113073
Li LX, Li SY, Wu N, et al., 2019. HOLMESv2: a CRISPR-Cas12b-assisted platform for nucleic acid detection and DNA methylation quantitation. ACS Synth Biol, 8(10): 2228–2237. https://doi.org/10.1021/acssynbio.9b00209
Li SY, Cheng QX, Wang JM, et al., 2018a. CRISPR-Cas12a-assisted nucleic acid detection. Cell Discov, 4:20. https://doi.org/10.1038/s41421-018-0028-z
Li SY, Cheng QX, Liu JK, et al., 2018b. CRISPR-Cas12a has both cis- and trans-cleavage activities on single-stranded DNA. Cell Res, 28(4):491–493. https://doi.org/10.1038/s41422-018-0022-x
Liu L, Xu ZH, Awayda K, et al., 2022. Gold nanoparticle-labeled CRISPR-Cas13a assay for the sensitive solid-state nanopore molecular counting. Adv Mater Technol, 7(3): 2101550. https://doi.org/10.1002/admt.202101550
Liu XY, Lin L, Tang LC, et al., 2021. Lb2Cas12a and its engineered variants mediate genome editing in human cells. FASEBJ, 35(4):e21270. https://doi.org/10.1096/fj.202001013RR
Liu YF, Xu HP, Liu C, et al., 2019. CRISPR-Cas13a nanomachine based simple technology for avian influenza a (H7N9) virus on-site detection. J Biomed Nanotechnol, 15(4):790–798. https://doi.org/10.1166/jbn.2019.2742
Lozano R, Naghavi M, Foreman K, et al., 2012. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet, 380(9859): 2095–2128. https://doi.org/10.1016/S0140-6736(12)61728-0
Ma L, Peng L, Yin LJ, et al., 2021. CRISPR-Cas12a-powered dual-mode biosensor for ultrasensitive and cross-validating detection of pathogenic bacteria. ACS Sens, 6(8):2920–2927. https://doi.org/10.1021/acssensors.1c00686
Ma L, Yin LJ, Li XY, et al., 2022. A smartphone-based visual biosensor for CRISPR-Cas powered SARS-CoV-2 diagnostics. Biosens Bioelectron, 195:113646. https://doi.org/10.1016/j.bios.2021.113646
Ma QN, Wang M, Zheng LB, et al., 2021. RAA-Cas12a-Tg: a nucleic acid detection system for Toxoplasma gondii based on CRISPR-Cas12a combined with recombinase-aided amplification (RAA). Microorganisms, 9(8):1644. https://doi.org/10.3390/microorganisms9081644
Maffert P, Reverchon S, Nasser W, et al., 2017. New nucleic acid testing devices to diagnose infectious diseases in resource-limited settings. Eur J Clin Microbiol Infect Dis, 36(10):1717–1731. https://doi.org/10.1007/s10096-017-3013-9
Makarova KS, Wolf YI, Alkhnbashi OS, et al., 2015. An updated evolutionary classification of CRISPR-Cas systems. Nat Rev Microbiol, 13(11):722–736. https://doi.org/10.1038/nrmicro3569
Matthijs G, Souche E, Alders M, et al., 2016. Guidelines for diagnostic next-generation sequencing. Eur J Hum Genet, 24(1):2–5. https://doi.org/10.1038/ejhg.2015.226
Mohanraju P, Makarova KS, Zetsche B, et al., 2016. Diverse evolutionary roots and mechanistic variations of the CRISPR-Cas systems. Science, 353(6299):aad5147. https://doi.org/10.1126/science.aad5147
Myhrvold C, Freije CA, Gootenberg JS, et al., 2018. Field-deployable viral diagnostics using CRISPR-Cas13. Science, 360(6387):444–448. https://doi.org/10.1126/science.aas8836
Nguyen LT, Smith BM, Jain PK, 2020. Enhancement of trans-cleavage activity of Cas12a with engineered crRNA enables amplified nucleic acid detection. Nat Commun, 11:4906. https://doi.org/10.1038/s41467-020-18615-1
O’Connell MR, 2019. Molecular mechanisms of RNA targeting by Cas13-containing type VI CRISPR-Cas systems. J Mol Biol, 431(1):66–87. https://doi.org/10.1016/j.jmb.2018.06.029
Pardee K, Green AA, Takahashi MK, et al., 2016. Rapid, low-cost detection of Zika virus using programmable biomolecular components. Cell, 165(5): 1255–1266. https://doi.org/10.1016/j.cell.2016.04.059
Pausch P, Al-Shayeb B, Bisom-Rapp E, et al., 2020. CRISPR-Cas® from huge phages is a hypercompact genome editor. Science, 369(6501):333–337. https://doi.org/10.1126/science.abb1400
Qin PW, Park M, Alfson KJ, et al., 2019. Rapid and fully microfluidic ebola virus detection with CRISPR-Cas13a. ACS Sens, 4(4):1048–1054. https://doi.org/10.1021/acssensors.9b00239
Sam IK, Chen YY, Ma J, et al., 2021. TB-QUICK: CRISPR-Cas12b-assisted rapid and sensitive detection of Mycobacterium tuberculosis. J Infect, 83(1):54–60. https://doi.org/10.1016/j.**f.2021.04.032
Sano T, Smith CL, Cantor CR, 1992. Immuno-PCR: very sensitive antigen detection by means of specific antibody-DNA conjugates. Science, 258(5079):120–122. https://doi.org/10.1126/science.1439758
Scheler O, Glynn B, Kurg A, 2014. Nucleic acid detection technologies and marker molecules in bacterial diagnostics. Expert Rev Mol Diagn, 14(4):489–500. https://doi.org/10.1586/14737159.2014.908710
Schnell C, 2019. Quantum imaging in biological samples. Nat Methods, 16(3):214. https://doi.org/10.1038/s41592-019-0346-6
Shen JJ, Zhou XM, Shan YY, et al., 2020. Sensitive detection of a bacterial pathogen using allosteric probe-initiated catalysis and CRISPR-Cas13a amplification reaction. Nat Commun, 11:267. https://doi.org/10.1038/s41467-019-14135-9
Shinoda H, Taguchi Y, Nakagawa R, et al., 2021. Amplification-free RNA detection with CRISPR-Cas13. Commun Biol, 4:476. https://doi.org/10.1038/s42003-021-02001-8
Shmakov S, Abudayyeh OO, Makarova KS, et al., 2015. Discovery and functional characterization of diverse class 2 CRISPR-Cas systems. Mol Cell, 60(3):385–397. https://doi.org/10.1016/j.molcel.2015.10.008
Smargon AA, Cox DBT, Pyzocha NK, et al., 2017. Cas13b is a type VI-B CRISPR-associated RNA-guided rnase differentially regulated by accessory proteins Csx27 and Csx28. Mol Cell, 65(4):618–630.e7. https://doi.org/10.1016/j.molcel.2016.12.023
Sun YY, Yu L, Liu CX, et al., 2021. One-tube SARS-CoV-2 detection platform based on RT-RPA and CRISPR/Cas12a. J Transl Med, 19:74. https://doi.org/10.1186/s12967-021-02741-5
Taylor SC, Laperriere G, Germain H, 2017. Droplet Digital PCR versus qPCR for gene expression analysis with low abundant targets: from variable nonsense to publication quality data. Sci Rep, 7:2409. https://doi.org/10.1038/s41598-017-02217-x
Teng F, Guo L, Cui TT, et al., 2019. CDetection: CRISPR-Cas12b-based DNA detection with sub-attomolar sensitivity and single-base specificity. Genome Biol, 20:132. https://doi.org/10.1186/s13059-019-1742-z
Terns MP, Terns RM, 2011. CRISPR-based adaptive immune systems. Curr Opin Microbiol, 14(3):321–327. https://doi.org/10.1016/j.mib.2011.03.005
Tsou JH, Leng QX, Jiang F, 2019. A CRISPR test for detection of circulating nuclei acids. Transl Oncol, 12(12):1566–1573. https://doi.org/10.1016/j.tranon.2019.08.011
Wang B, Wang R, Wang DQ, et al., 2019. Cas12aVDet: a CRISPR/Cas12a-based platform for rapid and visual nucleic acid detection. Anal Chem, 91(19):12156–12161. https://doi.org/10.1021/acs.analchem.9b01526
Wang R, Qian CY, Pang YN, et al., 2021. opvCRISPR: one-pot visual RT-LAMP-CRISPR platform for SARS-cov-2 detection. Biosens Bioelectron, 172:112766. https://doi.org/10.1016/j.bios.2020.112766
Wang S, Li H, Kou Z, et al., 2021. Highly sensitive and specific detection of hepatitis B virus DNA and drug resistance mutations utilizing the PCR-based CRISPR-Cas13a system. Clin Microbiol Infect, 27(3):443–450. https://doi.org/10.1016/j.cmi.2020.04.018
Wang XJ, Ji PP, Fan HY, et al., 2020a. CRISPR/Cas12a technology combined with immunochromatographic strips for portable detection of African swine fever virus. Commun Biol, 3:62. https://doi.org/10.1038/s42003-020-0796-5
Wang XJ, Shang XY, Huang XX, 2020b. Next-generation pathogen diagnosis with CRISPR/Cas-based detection methods. Emerg Microbes Infect, 9(1):1682–1691. https://doi.org/10.1080/22221751.2020.1793689
Wang XJ, Zhong MT, Liu Y, et al., 2020c. Rapid and sensitive detection of COVID-19 using CRISPR/Cas12a-based detection with naked eye readout, CRISPR/Cas12a-NER. Sci Bull, 65(17):1436–1439. https://doi.org/10.1016/j.scib.2020.04.041
Wei YD, Tao ZZ, Wan L, et al., 2022. Aptamer-based Cas14a1 biosensor for amplification-free live pathogenic detection. Biosens Bioelectron, 211:114282. https://doi.org/10.1016/j.bios.2022.114282
Wu JH, Mukama O, Wu W, et al., 2020. A CRISPR/Cas12a based universal lateral flow biosensor for the sensitive and specific detection of African swine-fever viruses in whole blood. Biosensors, 10(12):203. https://doi.org/10.3390/bios10120203
Wu YT, Liu SX, Wang F, et al., 2019. Room temperature detection of plasma Epstein-Barr virus DNA with CRISPR-Cas13. Clin Chem, 65(4):591–592. https://doi.org/10.1373/clinchem.2018.299347
Xu W, ** T, Dai YF, et al., 2020. Surpassing the detection limit and accuracy of the electrochemical DNA sensor through the application of CRISPR Cas systems. Biosens Bioelectron, 155:112100. https://doi.org/10.1016/j.bios.2020.112100
Yan FC, Wang W, Zhang JQ, 2019. CRISPR-Cas12 and Cas13: the lesser known siblings of CRISPR-Cas9. Cell Biol Toxicol, 35(6):489–492. https://doi.org/10.1007/s10565-019-09489-1
Yan WX, Chong SR, Zhang HB, et al., 2018. Cas13d is a compact RNA-targeting type VI CRISPR effector positively modulated by a WYL-domain-containing accessory protein. Mol Cell, 70(2):327–339.e5. https://doi.org/10.1016/j.molcel.2018.02.028
Yan WX, Hunnewell P, Alfonse LE, et al., 2019. Functionally diverse type V CRISPR-Cas systems. Science, 363(6422): 88–91. https://doi.org/10.1126/science.aav7271
You Y, Zhang PP, Wu GS, et al., 2021. Highly specific and sensitive detection of Yersinia pestis by portable Cas12a-UPTLFA platform. Front Microbiol, 12:700016. https://doi.org/10.3389/fmicb.2021.700016
Yu ACH, Vatcher G, Yue X, et al., 2012. Nucleic acid-based diagnostics for infectious diseases in public health affairs. Front Med, 6(2):173–186. https://doi.org/10.1007/s11684-012-0195-5
Zetsche B, Gootenberg JS, Abudayyeh OO, et al., 2015. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell, 163(3):759–771. https://doi.org/10.1016/j.cell.2015.09.038
Zhang F, Abudayyeh OO, Gootenberg JS, 2020. A protocol for detection of COVID-19 using CRISPR diagnostics. https://broad.io/sherlockprotocol
Zhang YQ, Chen MY, Liu CR, et al., 2021. Sensitive and rapid on-site detection of SARS-CoV-2 using a gold nanoparticle-based high-throughput platform coupled with CRISPR/Cas12-assisted RT-LAMP. Sens Actuators B Chem, 345: 130411. https://doi.org/10.1016/j.snb.2021.130411
Zhao YX, Chen F, Li Q, et al., 2015. Isothermal amplification of nucleic acids. Chem Rev, 115(22):12491–12545. https://doi.org/10.1021/acs.chemrev.5b00428
Zhou J, Yin LJ, Dong YN, et al., 2020. CRISPR-Cas13a based bacterial detection platform: sensing pathogen Staphylococcus aureus in food samples. Anal Chim Acta, 1127:225–233. https://doi.org/10.1016/j.aca.2020.06.041
Acknowledgments
This work was supported by the Natural Science Basic Research Plan in Shaanxi Province of China (No. 2020JQ-148), the Guangdong Basic and Applied Basic Research Foundation (No. 2021A1515110793), and the Shenzhen Pathogen High-Throughput Sequencing Technical Engineering Laboratory Upgrading Project (No. 2019-986), China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Hongdan GAO prepared the manuscript of the article, including all figures and tables. Zifang SHANG completed the literature collection, the article writing, and the drawing of all the figures and tables. Siew Yin CHAN provided the proofreading of the article. Dongli MA provided guidance and suggestions for the article writing. All authors have read and approved the final version.
Compliance with ethics guidelines
Hongdan GAO, Zifang SHANG, Siew Yin CHAN, and Dongli MA declare that there are no conflicts of interest.
This review does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Gao, H., Shang, Z., Chan, S.Y. et al. Recent advances in the use of the CRISPR-Cas system for the detection of infectious pathogens. J. Zhejiang Univ. Sci. B 23, 881–898 (2022). https://doi.org/10.1631/jzus.B2200068
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200068