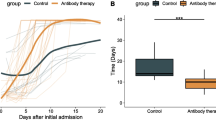
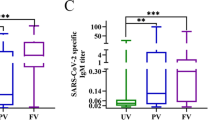
Abstract
Objective
This study aimed to observe the clinical and immune response characteristics of vaccinated persons infected with the delta variant of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in Yangzhou, China.
Methods
We extracted the medical data of 129 patients with delta-variant infection who were admitted to Northern Jiangsu People’s Hospital (Yangzhou, China) between August and September, 2021. The patients were grouped according to the number of vaccine doses received into an unvaccinated group: a one-dose group and a two-dose group. The vaccine used was SARS-CoV-2-inactivated vaccine developed by Sinovac. We retrospectively analyzed the patients’ epidemiological, clinical, laboratory, and imaging data.
Results
Almost all patients with delta-variant infection in Yangzhou were elderly, and patients with severe/critical illness were over 70 years of age. The rates of severe/critical illness (P=0.006), fever (P=0.025), and dyspnea (P=0.045) were lower in the two-dose group than in the unvaccinated group. Compared to the unvaccinated group, the two-dose group showed significantly higher lymphocyte counts and significantly lower levels of C-reactive protein (CRP), interleukin-6 (IL-6), and D-dimer during hospitalization and a significantly higher positive rate of immunoglobulin G (IgG) antibodies at admission (all P<0.05). The cumulative probabilities of hospital discharge and negative virus conversion were also higher in the two-dose group than in the unvaccinated group (P<0.05).
Conclusions
Two doses of the SARS-CoV-2-inactivated vaccine were highly effective at limiting symptomatic disease and reducing immune response, while a single dose did not seem to be effective.
Abstract
目的
观察扬州地区疫苗接种对德尔塔(Delta)变异型冠状病毒(SARS-CoV-2)感染者临床和免疫应答特征的影响。
方法
提取2021年8-9月苏北人民医院收治的129例Delta变异株感染患者的病例资料。根据接种疫苗的剂量将患者分为未接种组、一针组和两针组。所用疫苗为北京科兴公司研制的SARS-CoV-2灭活疫苗。我们回顾性分析了患者的流行病学、临床、实验室和影像学资料。
结果
扬州地区Delta变异感染患者多数为老年人,重症/危重患者年龄均在70岁以上。两针组重症/危重症(P=0.006)患者人数、发热(P=0.025)和呼吸困难(P=0.045)发生率均低于未接种组。与未接种组相比,两针组住院期间淋巴细胞计数显著升高,C反应蛋白(CRP)、白细胞介素-6(IL-6)和D-二聚体水**显著降低,入院时免疫球蛋白G(IgG)抗体阳性率显著升高(P<0.05)。两针组的累计出院概率和病毒转阴概率也高于未接种组(P<0.05)。
结论
接种两针SARS-CoV-2灭活疫苗在减轻新冠肺炎患者症状和降低免疫应答方面效果显著,接种一针疫苗效果不明显。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abu-Raddad LJ, Chemaitelly H, Butt AA, et al., 2021. Effectiveness of the BNT162b2 Covid-19 vaccine against the B.1.1.7 and B.1.351 variants. N Engl J Med, 385(2): 187–189. https://doi.org/10.1056/NEJMc2104974
Baden LR, el Sahly HM, Essink B, et al., 2021. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med, 384(5):403–416. https://doi.org/10.1056/NEJMoa2035389
Baj A, Novazzi F, Ferrante FD, et al., 2021. Spike protein evolution in the SARS-CoV-2 Delta variant of concern: a case series from Northern Lombardy. Emerg Microbes Infect, 10(1):2010–2015. https://doi.org/10.1080/22221751.2021.1994356
Beňová K, Hancková M, Koči K, et al., 2020. T cells and their function in the immune response to viruses. Acta Virol, 64(2):131–143. https://doi.org/10.4149/av_2020_203
Chen L, Liu HG, Liu W, et al., 2020. Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia. Chin J Tuberc Respir Dis, 43(3):203–208 (in Chinese). https://doi.org/10.3760/cma.j.issn.1001-0939.2020.03.013
Dhar MS, Marwal R, Vs R, et al., 2021. Genomic characterization and epidemiology of an emerging SARS-CoV-2 variant in Delhi, India. Science, 374(6570):eabj9932. https://doi.org/10.1126/science.abj9932
Gabarre P, Dumas G, Dupont T, et al., 2020. Acute kidney injury in critically ill patients with COVID-19. Intensive Care Med, 46(7):1339–1348. https://doi.org/10.1007/s00134-020-06153-9
Ghosh AK, Kaiser M, Molla MA, et al., 2021. Molecular and serological characterization of the SARS-CoV-2 delta variant in Bangladesh in 2021. Viruses, 13(11):2310. https://doi.org/10.3390/v13112310
Glowacka I, Bertram S, Muller MA, et al., 2011. Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response. J Virol, 85(9):4122–4134. https://doi.org/10.1128/JVI.02232-10
Gupta N, Kaur H, Yadav PD, et al., 2021. Clinical characterization and genomic analysis of samples from COVID-19 breakthrough infections during the second wave among the various states of India. Viruses, 13(9):1782. https://doi.org/10.3390/v13091782
Haileamlak A, 2021. After a year, SARS-CoV-2 is not well known. Ethiop J Health Sci, 31(2):212.
Hall VJ, Foulkes S, Saei A, et al., 2021. COVID-19 vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine against infection (SIREN): a prospective, multicentre, cohort study. Lancet, 397(10286):1725–1735. https://doi.org/10.1016/S0140-6736(21)00790-X
Hamming I, Timens W, Bulthuis MLC, et al., 2004. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol, 203(2):631–637. https://doi.org/10.1002/path.1570
Hansrivijit P, Qian CC, Boonpheng B, et al., 2020. Incidence of acute kidney injury and its association with mortality in patients with COVID-19: a meta-analysis. J Investig Med, 68(7):1261–1270. https://doi.org/10.1136/jim-2020-001407
Hoffmann M, Kleine-Weber H, Schroeder S, et al., 2020. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell, 181(2):271–280.E8. https://doi.org/10.1016/j-cell.2020.02.052
Hu XF, Chen JF, Jiang XM, et al., 2020. CT imaging of two cases of one family cluster 2019 novel coronavirus (2019-nCoV) pneumonia: inconsistency between clinical symptoms amelioration and imaging sign progression. Quant Imaging Med Surg, 10(2):508–510. https://doi.org/10.21037/qims.2020.02.10
Huang CL, Wang YM, Li XW, et al., 2020. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet, 395(10223):497–506. https://doi.org/10.1016/S0140-6736(20)30183-5
Jalkanen P, Kolehmainen P, Häkkinen HK, et al., 2021. COVID-19 mRNA vaccine induced antibody responses against three SARS-CoV-2 variants. Nat Commun, 12:3991. https://doi.org/10.1038/s41467-021-24285-4
Kemp SA, Collier DA, Datir R, et al., 2020. Neutralising antibodies in Spike mediated SARS-CoV-2 adaptation. medRxiv, preprint. https://doi.org/10.1101/2020.12.05.20241927
Kleine-Weber H, Elzayat MT, Hoffmann M, et al., 2018. Functional analysis of potential cleavage sites in the MERS-coronavirus spike protein. Sci Rep, 8:16597. https://doi.org/10.1038/s41598-018-34859-w
Kong WF, Agarwal PP, 2020. Chest imaging appearance of COVID-19 infection. Radiol Cardiothorac Imaging, 2(1): e200028. https://doi.org/10.1148/ryct.2020200028
Lei JQ, Li JF, Li X, et al., 2020. CT imaging of the 2019 novel coronavirus (2019-nCoV) pneumonia. Radiology, 295(1): 18. https://doi.org/10.1148/radiol.2020200236
Li RF, Hou YL, Huang JC, et al., 2020. Lianhuaqingwen exerts anti-viral and anti-inflammatory activity against novel coronavirus (SARS-CoV-2). Pharmacol Res, 156: 104761. https://doi.org/10.1016/j.phrs.2020.104761
Li ZT, Yi YX, Luo XM, et al., 2020. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J Med Virol, 92(9):1518–1524. https://doi.org/10.1002/jmv.25727
Liang YW, Wang ML, Chien CS, et al., 2020. Highlight of immune pathogenic response and hematopathologic effect in SARS-CoV, MERS-CoV, and SARS-CoV-2 infection. Front Immunol, 11:1022. https://doi.org/10.3389/fimmu.2020.01022
Liu Y, Liu JY, **a HJ, et al., 2021. Neutralizing activity of BNT162b2-elicited serum. N Engl J Med, 384(15):1466–1468. https://doi.org/10.1056/NEJMc2102017
Long QX, Liu BZ, Deng HJ, et al., 2020. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat Med, 26(6):845–848. https://doi.org/10.1038/s41591-020-0897-1
Lu HH, Shi Y, Chen KL, et al., 2021. Impact of antitumor regimens on the outcomes of cancer patients with COVID-19: a pooled analysis. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 22(10):876–884. https://doi.org/10.1631/jzus.B2100151
Lu RJ, Zhao X, Li J, et al., 2020. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet, 395(10224): 565–574. https://doi.org/10.1016/S0140-6736(20)30251-8
Malik JA, Mulla AH, Farooqi T, et al., 2021. Targets and strategies for vaccine development against SARS-CoV-2. Biomed Pharmacother, 137:111254. https://doi.org/10.1016/j.biopha.2021.111254
Mudatsir M, Fajar JK, Wulandari L, et al., 2020. Predictors of COVID-19 severity: a systematic review and meta-analysis. F1000Res, 9:1107. https://doi.org/10.12688/f1000research.26186.2
National Health Commission of the People’s Republic of China, 2020. Technical guidance for laboratory testing of 2019-nCoV infection (Third Edition). Biosaf Health, 2(1):3–5. https://doi.org/10.1016/j.bsheal.2020.02.001
National Health Commission of the People’s Republic of China, National Administration of Tranditional Chinese Medicine, 2022. Diagnosis and treatment protocol for COVID-19 (trial version 9). http://www.nhc.gov.cn/yzygj/s7653p/202203/b74ade1ba4494583805a3d2e40093d88.shtml (in Chinese).
Novelli G, Colona VL, Pandolfi PP, 2021. A focus on the spread of the delta variant of SARS-CoV-2 in India. Indian J Med Res, 153(5–6):537–541. https://doi.org/10.4103/ijmr.ijmr_1353_21
Pan F, Ye TH, Sun P, et al., 2020. Time course of lung changes at chest CT during recovery from coronavirus disease 2019 (COVID-19). Radiology, 295(3):715–721. https://doi.org/10.1148/radiol.2020200370
Parums DV, 2021. Editorial: Revised World Health Organization (WHO) terminology for variants of concern and variants of interest of SARS-CoV-2. Med Sci Monit, 27:e933622. https://doi.org/10.12659/MSM.933622
Shi HS, Han XY, Jiang NC, et al., 2020. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis, 20(4): 425–434. https://doi.org/10.1016/S1473-3099(20)30086-4
Shi Y, Wang G, Cai XP, et al., 2020. An overview of COVID-19. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(5): 343–360. https://doi.org/10.1631/jzus.B2000083
Shinde V, Bhikha S, Hoosain Z, et al., 2021. Efficacy of NVX-CoV2373 COVID-19 vaccine against the B.1.351 variant. N Engl J Med, 384(20):1899–1909. https://doi.org/10.1056/NEJMoa2103055
Skowronski DM, de Serres G, 2021. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med, 384(16):1576–1577. https://doi.org/10.1056/NEJMc2036242
Sultana J, Mazzaglia G, Luxi N, et al., 2020. Potential effects of vaccinations on the prevention of COVID-19: rationale, clinical evidence, risks, and public health considerations. Expert Rev Vaccines, 19(10):919–936. https://doi.org/10.1080/14760584.2020.1825951
Tang N, Bai H, Chen X, et al., 2020. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost, 18(5):1094–1099. https://doi.org/10.1111/jth.14817
Voysey M, Clemens SAC, Madhi SA, et al., 2021. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet, 397(10269):99–111. https://doi.org/10.1016/S0140-6736(20)32661-1
Wan ZK, Zhou ZF, Liu Y, et al., 2020. Regulatory T cells and T helper 17 cells in viral infection. Scand J Immunol, 91(5):e12873. https://doi.org/10.1111/sji.12873
Wang J, Jiang MM, Chen X, et al., 2020. Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts. J Leukoc Biol, 108(1):17–41. https://doi.org/10.1002/JLB.3COVR0520-272R
Wang ZF, Su F, Lin XJ, et al., 2011. Serum D-dimer changes and prognostic implication in 2009 novel influenza A (H1N1). Thromb Res, 127(3):198–201. https://doi.org/10.1016/j.thromres.2010.11.032
Wu XJ, Wu P, Shen YF, et al., 2018. CD8+ resident memory T cells and viral infection. Front Immunol, 9:2093. https://doi.org/10.3389/fimmu.2018.02093
Wu ZY, McGoogan JM, 2020. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA, 323(13):1239–1242. https://doi.org/10.1001/jama.2020.2648
**ong Y, Liu Y, Cao L, et al., 2020. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID-19 patients. Emerg Microbes Infect, 9(1):761–770. https://doi.org/10.1080/22221751.2020.1747363
Xu X, Sun J, Nie S, et al., 2020. Seroprevalence of immunoglobulin M and G antibodies against SARS-CoV-2 in China. Nat Med, 26(8):1193–1195. https://doi.org/10.1038/s41591-020-0949-6
Xu XT, Chen P, Wang JF, et al., 2020. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci China Life Sci, 63(3):457–460. https://doi.org/10.1007/s11427-020-1637-5
Yan SM, Wu G, 2021. Is lymphopenia different between SARS and COVID-19 patients? FASEB J, 35(2):e21245. https://doi.org/10.1096/fj.202002512
Zhang GM, Zhang J, Wang BW, et al., 2020. Analysis of clinical characteristics and laboratory findings of 95 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a retrospective analysis. Respir Res, 21:74. https://doi.org/10.1186/s12931-020-01338-8
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos. 82171207 and 82172190), the Jiangsu Association for Science and Technology Young Scientific and Technological Talents Support Project (No. 2021-008), the Jiangsu Province “333” High-level Talents Training Project (No. 2022-3-6-146), and the Yangzhou Science and Technology Plan Project (Nos. YZ2021088 and YZ2021148). The authors thank Prof. Yang YANG (Clinical Medical College, Yangzhou University, Yangzhou, China) for her guidance on statistics.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Author contributions
Cun** WANG, Yong LI, Yuchen PAN, and Luo**g ZHOU contributed to data collection, analysis, interpretation, and writing of the manuscript. ** ZHANG, Yan WEI, and Fang GUO contributed to figure preparation and manuscript editing. Yusheng SHU and Ju GAO contributed to the study concept and design, clinical and pathological analyses, study supervision, and critical revision of the manuscript. All authors have read and approved the final manuscript, and therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Cun** WANG, Yong LI, Yuchen PAN, Luo**g ZHOU, ** ZHANG, Yan WEI, Fang GUO, Yusheng SHU, and Ju GAO declare that they have no conflict of interest.
This study was approved by the Research Ethics Committees of Northern Jiangsu People’s Hospital (No. 2021ky284), and was performed in compliance with World Medical Association guidelines and the Helsinki Declaration of 1975, as revised in 2008 (5). The need for written informed consent was waived because this was a chart review, and the study protocol was approved under rapid review due to the urgent need to collect and report data. The data were collected and analyzed by three authors, who ensured the accuracy and integrity of the data and ensured that the data-collection process complied with the protocol.
Supplementary information
Figs. S1–S5
Supplementary Information
11585_2022_647_MOESM1_ESM.pdf
Clinical and immune response characteristics among vaccinated persons infected with SARS-CoV-2 delta variant: a retrospective study
Rights and permissions
About this article
Cite this article
Wang, C., Li, Y., Pan, Y. et al. Clinical and immune response characteristics among vaccinated persons infected with SARS-CoV-2 delta variant: a retrospective study. J. Zhejiang Univ. Sci. B 23, 899–914 (2022). https://doi.org/10.1631/jzus.B2200054
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200054
Key words
- Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
- Delta variant
- Vaccine
- Hospitalization
- Immune response