Abstract
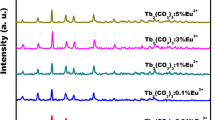
Eu3+/Tb3+co-doped YBO3 three-dimensional (3D) microstructures have been hydrothermally prepared by adjusting solvent and the molar ratio of Y3+ to B (Y/B) at 180 °C. The whole process was carried out under alkaline conditions without the use of any surfactant or catalyst. Characterizations of the samples are carried out using X-ray powder diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and high-resolution TEM (HRTEM). The photoluminescence (PL) colors of YBO3 sample co-doped with Eu3+ and Tb3+ under ultraviolet excitation can be tuned from red, through yellow and green-yellow, to green by changing the relative do** concentrations of the two activator ions. These phosphors with multicolor emissions in the visible region be potentially used as labels for light-display systems, optoelectronic devices and biological molecules.
Similar content being viewed by others
References
M. Ren, J. H. Lin, Y. Dong, L. Q. Yang, M. Z. Su, Chem. Mater, (1999), 11, 1576–1580.
G. H. Pan, H. W. Song, X. Bai, Z. X. Liu, H. Q. Yu, W. H. Di, S. Li, L. Fan, X. Ren, S. Lu, Chem. Mater,(2006), 18, 4526–4532.
Y.H. Wang, T. Endo, L. He, C.F. Wu, J. Cryst. Growth. (2004), 268, 568.
T. Kim, S. Kang, Mater. Res. Bull. (2005), 40, 1945.
L.L. Wang, Y.H. Wang, J. Lumin. (2007), 122-123, 921.
R. Sato, S. Takeshita, T. Isobe, T. Sawayama and S. Niikura, ECS J. Solid State Sci. Technol. (2012) 1, 6, R163–R168.
M.S. Shur, A. Zukauskas, Proc. IEEE (2005), 93, 1691–1703.
Y. Sato, N. Takahashi, S. Sato, Jpn. J. Appl. Phys. Part 2, (1996), 35, L838–L839.
G. Blasse, B. C. Grabmaier, Luminescent Materials, Springer- Verlag: Berlin Heideberg, 1994.
R. C. Evans, L. D. Carlos, Peter. Douglas and J. Rocha, J. Mater. Chem., 2008, 18, 1100.
P. Ghosh, A. Kar and A. Patra, Nanoscale, 2010, 2, 1196.
S. Mukherjee, V. Sudarsan, R.K. Vatsa, S. V. Godbole, R.M. Kadam, U.M. Bhatta, A.K. Tyagi, Nanotechnology. (2008), 19, 325704.
G. Jia, H.P. You, K. Liu, Y. Zheng, N. Guo, J. Jia, and H. Zhang, Chem. Eur. J. (2010), 16, 2930–2937
A.A. Setlur, Electrochemical and Solid – State Letters, (2012), 15, (6) J25–J27.
X.W. Zhang, A. Marathe, S. Sohal, M. Holtz, M. Davis, L.J. Hope-Weeks, J. Chaudhuri, J. Mater. Chem.. (2012), 22, 6485.
G.K. Das, T. Tan, T. Y. J. Phys. Chem. C (2008), 112, 11211.
S. Mukherjee, V. Sudarsan, R.K. Vatsa, S.V. Godbole, R.M. Kadam, U.M. Bhatta, A.K. Tyagi Nanotechnology (2008), 19, 325704.
G. Blasse and A. Bril, J. Chem. Phys., (1967), 47, 1920.
Z. Wei, L. Sun, C. Liao, J. Yin, X. Jiang, and C. Yan, J. Phys. Chem. B. (2002), 106, 10610.
G. Blasse, Philips Res. Rep. (1969), 24, 131.
N. Guo, Y. Huang, H. You, M. Yang, Y. Song, K. Liu and, Y. Zheng Inorg. Chem, (2010), 49, 10907.