Abstract
Background
This study aims to reveal the clinical significance of stromal-infiltrating tumor-associated macrophages (TAMs) in muscle-invasive bladder cancer (MIBC).
Patients and Methods
This study included 288 patients from the TCGA database and 118 patients from Fudan University Shanghai Cancer Center with MIBC. The CIBERSORT model and immunohistochemistry were used to evaluate TAM infiltration. Cox regression analyses were employed to calculate their prognostic value.
Results
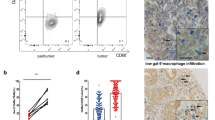
Among all 23 immune phenotypes analyzed in the TCGA cohort, pan-macrophage infiltration was significantly associated with poor prognosis (p = 0.001). Further analyses found that stromal TAM infiltration could be an independent prognostic predictor for recurrence-free survival (RFS; HR: 1.019, 95% CI: 1.006–1.033, p = 0.004). High stromal infiltration was related to unfavorable RFS. After stratification by adjuvant chemotherapy (ACT), patients without ACT could be differentiated by TAM infiltration (p = 0.036), while patients with ACT could not. Moreover, TAM infiltration was negatively associated with IFN-γ-related mRNA panel, which was shown to have strong predictive value for clinical response to programmed death-1 (PD-1) inhibition.
Conclusions
Stromal TAM infiltration could be an independent prognosticator for MIBC patients. This might have potential to guide precise treatments such as ACT and immune checkpoint blockade in MIBC.



Similar content being viewed by others
Data Availability Statement
All data relevant to the results presented herein are available from the corresponding author upon reasonable request.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34.
Ghandour R, Singla N, Lotan Y. Treatment options and outcomes in nonmetastatic muscle invasive bladder cancer. Trends Cancer. 2019;5(7):426–39.
Liu D, Abbosh P, Keliher D, et al. Mutational patterns in chemotherapy resistant muscle-invasive bladder cancer. Nat Commun. 2017;8(1):2193.
Vlachostergios PJ, Faltas BM. Treatment resistance in urothelial carcinoma: an evolutionary perspective. Nat Rev Clin Oncol. 2018;15(8):495–509.
Fridman WH, Pages F, Sautes-Fridman C, Galon J. The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer. 2012;12(4):298–306.
Li S, Xu F, Zhang J, et al. Tumor-associated macrophages remodeling EMT and predicting survival in colorectal carcinoma. Oncoimmunology. 2018;7(2):e1380765.
Ding T, Xu J, Wang F, et al. High tumor-infiltrating macrophage density predicts poor prognosis in patients with primary hepatocellular carcinoma after resection. Hum Pathol. 2009;40(3):381–9.
Sumitomo R, Hirai T, Fujita M, Murakami H, Otake Y, Huang CL. PD-L1 expression on tumor-infiltrating immune cells is highly associated with M2 TAM and aggressive malignant potential in patients with resected non-small cell lung cancer. Lung Cancer. 2019;136:136–44.
Turley SJ, Cremasco V, Astarita JL. Immunological hallmarks of stromal cells in the tumour microenvironment. Nat Rev Immunol. 2015;15(11):669–82.
Newman AM, Liu CL, Green MR, et al. Robust enumeration of cell subsets from tissue expression profiles. Nat Methods. 2015;12(5):453–7.
Fukumura D, Kloepper J, Amoozgar Z, Duda DG, Jain RK. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nat Revs Clin Oncol. 2018;15(5):325.
Ayers M, Lunceford J, Nebozhyn M, et al. IFN-gamma-related mRNA profile predicts clinical response to PD-1 blockade. J Clin Invest. 2017;127(8):2930–40.
Nielsen SR, Schmid MC. Macrophages as key drivers of cancer progression and metastasis. Mediat Inflamm. 2017;2017:9624760.
Chen Y, Song Y, Du W, Gong L, Chang H, Zou Z. Tumor-associated macrophages: an accomplice in solid tumor progression. J Biomed Sci. 2019;26(1):78.
Zhang J, Yan Y, Yang Y, et al. High infiltration of tumor-associated macrophages influences poor prognosis in human gastric cancer patients, associates with the phenomenon of EMT. Medicine. 2016;95(6):e2636.
Zhao Y, Ge X, Xu X, Yu S, Wang J, Sun L. Prognostic value and clinicopathological roles of phenotypes of tumour-associated macrophages in colorectal cancer. J Cancer Res Clin Oncol. 2019;145:3005–19.
Sjodahl G, Lovgren K, Lauss M, et al. Infiltration of CD3(+) and CD68(+) cells in bladder cancer is subtype specific and affects the outcome of patients with muscle-invasive tumors. Urol Oncol. 2014;32(6):791–7.
Qiu S, Deng L, Liao X, et al. Tumor-associated macrophages promote bladder tumor growth through PI3K/AKT signal induced by collagen. Cancer Sci. 2019;110(7):2110–8.
Mitrofanova I, Zavyalova M, Telegina N, et al. Tumor-associated macrophages in human breast cancer parenchyma negatively correlate with lymphatic metastasis after neoadjuvant chemotherapy. Immunobiology. 2017;222(1):101–9.
Rakaee M, Busund LR, Jamaly S, et al. Prognostic value of macrophage phenotypes in resectable non-small cell lung cancer assessed by multiplex immunohistochemistry. Neoplasia. 2019;21(3):282–93.
Liu Z, Zhu Y, Xu L, et al. Tumor stroma-infiltrating mast cells predict prognosis and adjuvant chemotherapeutic benefits in patients with muscle invasive bladder cancer. Oncoimmunology. 2018;7(9):e1474317.
Waks AG, Stover DG, Guerriero JL, et al. The immune microenvironment in hormone receptor-positive breast cancer before and after preoperative chemotherapy. Clin Cancer Res. 2019;25(15):4644–55.
Leow JJ, Martin-Doyle W, Rajagopal PS, et al. Adjuvant chemotherapy for invasive bladder cancer: a 2013 updated systematic review and meta-analysis of randomized trials. Eur Urol. 2014;66(1):42–54.
Svatek RS, Shariat SF, Lasky RE, et al. The effectiveness of off-protocol adjuvant chemotherapy for patients with urothelial carcinoma of the urinary bladder. Clin Cancer Res. 2010;16(17):4461–7.
Galsky MD, Stensland KD, Moshier E, et al. Effectiveness of adjuvant chemotherapy for locally advanced bladder cancer. J Clin Oncol. 2016;34(8):825–32.
Bellmunt J, Powles T, Vogelzang NJ. A review on the evolution of PD-1/PD-L1 immunotherapy for bladder cancer: the future is now. Cancer Treat Rev. 2017;54:58–67.
Rouanne M, Roumiguie M, Houede N, et al. Development of immunotherapy in bladder cancer: present and future on targeting PD(L)1 and CTLA-4 pathways. World J Urol. 2018;36(11):1727–40.
Galsky MD, Wang H, Hahn NM, et al. Phase 2 trial of gemcitabine, cisplatin, plus ipilimumab in patients with metastatic urothelial cancer and impact of DNA damage response gene mutations on outcomes. Eur Urol. 2018;73(5):751–9.
Goodman AM, Kato S, Bazhenova L, et al. Tumor mutational burden as an independent predictor of response to immunotherapy in diverse cancers. Mol Cancer Ther. 2017;16(11):2598–608.
Acknowledgements
The authors thank Dr. Lingli Chen (Department of Pathology, Zhongshan Hospital, Fudan University, Shanghai, China) and Dr. Yunyi Kong (Department of Pathology, Fudan University Shanghai Cancer Center, Shanghai, China) for their excellent pathological technology help.
Funding
This study was funded by grants from National Natural Science Foundation of China (31770851, 81872082, 82002670, 82103408), Shanghai Municipal Natural Science Foundation (19ZR1431800), Shanghai Sailing Program (18YF1404500, 21YF1407000), Shanghai Municipal Commission of Health and Family Planning Program (201840168), and Fudan University Shanghai Cancer Center for Outstanding Youth Scholars Foundation (YJYQ201802). All these study sponsors had no role in the study design, or in the collection, analysis, and interpretation of data.
Author information
Authors and Affiliations
Contributions
SJ, HZ, ZL, and KJ: acquisition of data, analysis and interpretation of data, statistical analysis, and drafting of the manuscript; CL, SY, YY, RY, HZ, JZ, YC, and LX: technical and material support; JX, ZW, and YZ: study concept and design, analysis, and interpretation of data, drafting of the manuscript, obtained funding and study supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Disclosure
The authors declare no potential conflicts of interest.
Ethics Approval and Consent to Participate
The study followed the Declaration of Helsinki and was approved by the Clinical Research Ethics Committee of Fudan University Shanghai Cancer Center (No. 050432-4-1212B). Signed informed consent was acquired from each patient.
Consent for Publication
All authors provide their consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
**, S., Zeng, H., Liu, Z. et al. Stromal Tumor-Associated Macrophage Infiltration Predicts Poor Clinical Outcomes in Muscle-Invasive Bladder Cancer Patients. Ann Surg Oncol 29, 2495–2503 (2022). https://doi.org/10.1245/s10434-021-11155-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-11155-y