Abstract
Purpose
DNA repair genes are potential biomarkers for chemotherapy in muscle-invasive bladder cancer (MIBC). O6-methylguanine methyltransferase (MGMT) is involved in DNA repair and is found to affect the efficacy of platinum-based chemotherapy. However, the prognostic or predictive value of MGMT expression in chemotherapy for MIBC is unknown.
Materials and Methods
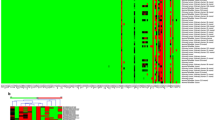
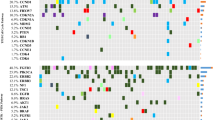
Immunohistochemical staining for MGMT was performed in paraffin-embedded tumor tissue of high-grade MIBC patients who underwent cystectomy in two independent cohorts [n = 74 for Fudan University Shanghai Cancer Center (FUSCC) cohort and n = 115 for Zhongshan Hospital (ZS) cohort]. MGMT messenger RNA (mRNA) analysis was conducted using patients’ clinical and fragments per kilobase of exon model per million mapped fragments mRNA data from The Cancer Genome Atlas (TCGA) database (n = 245).
Results
In our cohorts, high MGMT expression was significantly correlated with shorter overall survival (OS) in patients with platinum-based adjuvant chemotherapy [hazard ratio (HR) 2.386, p = 0.048; HR 2.920, p = 0.007; HR 2.324, p = 0.004, respectively, in FUSCC, ZS, and combination sets], but not in patients without chemotherapy. These findings were corroborated by the TCGA set (HR 1.952 and 0.697 for patients with and without chemotherapy, respectively). The chemotherapy–MGMT interaction for OS was significant in both the surgery set (p = 0.045) and TCGA set (p = 0.034).
Conclusions
We demonstrated that high MGMT expression is an independent poor prognostic factor in MIBC patients with platinum-based adjuvant chemotherapy, but not in patients without chemotherapy. MGMT expression may be a potential predictor for administration of adjuvant chemotherapy.



Similar content being viewed by others
References
Alfred Witjes J, Lebret T, Comperat EM, et al. Updated 2016 EAU guidelines on muscle-invasive and metastatic bladder cancer. Eur. Urol. 71(3):462–475 (2017).
Cognetti F, Ruggeri EM, Felici A, et al. Adjuvant chemotherapy with cisplatin and gemcitabine versus chemotherapy at relapse in patients with muscle-invasive bladder cancer submitted to radical cystectomy: an Italian, multicenter, randomized phase III trial. Ann. Oncol. 2012;23(3):695–700.
Sternberg CN, Skoneczna I, Kerst JM, et al. Immediate versus deferred chemotherapy after radical cystectomy in patients with pT3–pT4 or N+ M0 urothelial carcinoma of the bladder (EORTC 30994): an intergroup, open-label, randomised phase 3 trial. The Lancet Oncol. 2015;16(1):76–86.
Galsky MD, Stensland KD, Moshier E, et al. Effectiveness of adjuvant chemotherapy for locally advanced bladder cancer. J. Clin. Oncol. 2016;34(8):825–32.
Ruggeri EM, Fabbri MA, Nelli F. Should we use adjuvant chemotherapy for muscle-invasive bladder cancer after radical cystectomy? J. Clin. Oncol. 2016;34(26):3223.
Curtin NJ. DNA repair dysregulation from cancer driver to therapeutic target. Nat. Rev. Cancer. 2012;12(12):801–17.
Bellmunt J, Paz-Ares L, Cuello M, et al. Gene expression of ERCC1 as a novel prognostic marker in advanced bladder cancer patients receiving cisplatin-based chemotherapy. Ann. Oncol. 2007;18(3):522–8.
Font A, Taron M, Gago JL, et al. BRCA1 mRNA expression and outcome to neoadjuvant cisplatin-based chemotherapy in bladder cancer. Ann. Oncol. 2011;22(1):139–44.
Plimack ER, Dunbrack RL, Brennan TA, et al. Defects in DNA repair genes predict response to neoadjuvant cisplatin-based chemotherapy in muscle-invasive bladder cancer. Eur. Urol. 2015;68(6):959–67.
Gerson SL. MGMT: its role in cancer aetiology and cancer therapeutics. Nat. Rev. Cancer. 2004;4(4):296–307.
Hegi ME, Diserens AC, Gorlia T, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N. Engl. J. Med. 2005;352(10):997–1003.
Arienti C, Tesei A, Verdecchia GM, et al. Peritoneal carcinomatosis from ovarian cancer: chemosensitivity test and tissue markers as predictors of response to chemotherapy. J. Transl. Med. 2011;9:94.
Liu Z, Liu Y, Xu L, et al. P2X7 receptor predicts postoperative cancer-specific survival of patients with clear-cell renal cell carcinoma. Cancer Sci. 2015;106(9):1224–31.
Tanaka S, Kobayashi I, Utsuki S, Oka H, Yasui Y, Fujii K. Down-regulation of O6-methylguanine-DNA methyltransferase gene expression in gliomas by platinum compounds. Oncol. Rep. 2005;14(5):1275–80.
Maki Y, Murakami J, Asaumi J, et al. Role of O6-methylguanine-DNA methyltransferase and effect of O6-benzylguanine on the anti-tumor activity of cis-diaminedichloroplatinum(II) in oral cancer cell lines. Oral Oncol. 2005;41(10):984–93.
Chen SH, Kuo CC, Li CF, et al. O(6)-methylguanine DNA methyltransferase repairs platinum-DNA adducts following cisplatin treatment and predicts prognoses of nasopharyngeal carcinoma. Int. J. Cancer. 2015;137(6):1291–305.
Arienti C, Tesei A, Verdecchia GM, et al. Role of conventional chemosensitivity test and tissue biomarker expression in predicting response to treatment of peritoneal carcinomatosis from colon cancer. Clin. Colorectal Cancer. 2013;12(2):122–7.
Braun MS, Richman SD, Quirke P, et al. Predictive biomarkers of chemotherapy efficacy in colorectal cancer: results from the UK MRC FOCUS trial. J. Clin. Oncol. 2008;26(16):2690–8.
Konduri SD, Ticku J, Bobustuc GC, et al. Blockade of MGMT expression by O6 benzyl guanine leads to inhibition of pancreatic cancer growth and induction of apoptosis. Clin. Cancer Res. 2009;15(19):6087–95.
Bobustuc GC, Patel A, Thompson M, et al. MGMT inhibition suppresses survivin expression in pancreatic cancer. Pancreas. 2015;44(4):626–35.
Ramakrishnan V, Kushwaha D, Koay DC, et al. Post-transcriptional regulation of O(6)-methylguanine-DNA methyltransferase MGMT in glioblastomas. Cancer Biomark. 2011;10(3–4):185–93.
Acknowledgment
The results shown here are in part based upon data generated by the TCGA Research Network (http://cancergenome.nih.gov/). The authors thank TCGA Research Network for original data, and Huaipeng Zhang (Facebook Inc., California) for his program to extract data from the TCGA website. This study was funded by grants from National Natural Science Foundation of China (81402082, 81472227 and 31770851), Shanghai Municipal Commission of Health and Family Planning Program (20144Y0223), Innovation Project of Shanghai Jiao Tong University School of Medicine (16XJ21003), Outstanding Young Talent Training Plan of Shanghai Municipal Commission of Health and Family Planning (no. XYQ2013102), Guide Project of Science and Technology Commission of Shanghai Municipality (no. 17411963100), and a grant from Shanghai Cancer Research Charity Center. All these study sponsors played no role in the study design or collection, analysis, and interpretation of data.
Author Contributions
Conception and design: JZ, YZ, YW, BD, JX. Development of methodology: JZ, YZ, YW, QF, HX, ZL, HF, YC. Acquisition of data: JZ, YZ, YW, HX, ZL. Analysis and interpretation of data: JZ, YZ, YW, BD, JX. Writing, review, and/or revision of the manuscript: JZ, YZ, YW, QF, BD, JX. Administrative, technical, or material support: QF, HX, ZL, HF, YC. Study supervision: BD, JX
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, J., Zhu, Y., Wang, Y. et al. Prognostic and Predictive Value of O6-methylguanine Methyltransferase for Chemotherapy in Patients with Muscle-Invasive Bladder Cancer. Ann Surg Oncol 25, 342–348 (2018). https://doi.org/10.1245/s10434-017-6145-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6145-3