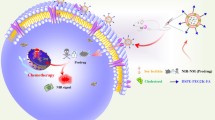
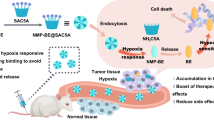
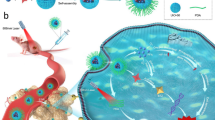
Abstract
Hypoxia is an important pathological phenomenon, and it can induce many tumor microenvironment changes, such as accumulations of intracellular lactic acid, decrease of tumor microenvironment pH value, and regulate a series of physiological and pathological processes such as adhesion, metastasis, and immune escape. Hypoxic tumor cells act as a key target for treating tumor. In this research, we designed and prepared PEG-nitroimidazole grafts, PEG-NI, and FA-PEG-NI. We first explored their physical and chemical properties to serve as a drug carrier. Then, the hypoxia-sensitive properties such as particle size changes and drug release were investigated. Finally, the tumor targeting ability was studied in vitro and in vivo, and anti-tumor capacity was determined. Both grafts showed excellent property as a nanodrug carrier and showed favorable drug encapsulation ability of sorafenib with the help of the hydrophobic chain of 6-(BOC-amino) hexyl bromide. The micelles responded to the hypoxic tumor environment with chemical and spatial structure changes leading to sensitive and fast drug release. With the modification of folic acid, FA-PEG-NI gained tumor targeting ability in vivo. FA-PEG-NI graft proved a potential targeting drug delivery system in the treatment of hypoxic hepatocellular carcinoma.
Graphical Abstract








Similar content being viewed by others
References
**g X, Yang F, Shao C, Wei K, **e M, Shen H, Shu Y. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer. 2019;157. https://doi.org/10.1186/s12943-019-1089-9.
Afshari R, Akhavan O, Hamblin MR, Varma RS. Review of oxygenation with nanobubbles: possible treatment for hypoxic COVID-19 patients. ACS Appl Nano Mater. 2021;11:11386–412. https://doi.org/10.1021/acsanm.1c01907.
Kumari R, Sunil D, Ningthoujam RS. Hypoxia-responsive nanoparticle based drug delivery systems in cancer therapy: an up-to-date review. J Control Release. 2020;319:135–56. https://doi.org/10.1016/j.jconrel.2019.12.041.
Rankin EB, Giaccia AJ. Hypoxic control of metastasis. Science. 2016;352:175–80. https://doi.org/10.1126/science.aaf4405.
Salem A, Asselin M-C, Reymen B, Jackson A, Lambin P, West CML, O’Connor JPB, Faivre-Finn C. Targeting hypoxia to improve non–small cell lung cancer outcome. J Natl Cancer Inst. 2018;110:14–30. https://doi.org/10.1093/jnci/djx160.
Peng HS, Liao MB, Zhang MY, **e Y, Xu L, Zhang YJ, Zheng XF, Wang HY, Chen YF. Synergistic inhibitory effect of hyperbaric oxygen combined with sorafenib on hepatoma cells. PLoS ONE. 2014;9:e100814. https://doi.org/10.1371/journal.pone.0100814.
Semenza GL. Hypoxia-inducible factors: mediators of cancer progression and targets for cancer therapy. Trends Pharmacol Sci. 2012;33:207–14. https://doi.org/10.1016/j.tips.2012.01.005.
Sun Y, Zhao D, Wang G, Wang Y, Cao L, Sun J, Jiang Q, He Z. Recent progress of hypoxia-modulated multifunctional nanomedicines to enhance photodynamic therapy: opportunities, challenges, and future development. Acta Pharm Sin B. 2020;10:1382–96. https://doi.org/10.1016/j.apsb.2020.01.004.
Wallace MC, Preen D, Jeffrey GP, Adams LA. The evolving epidemiology of hepatocellular carcinoma: a global perspective. Expert Rev Gastroent. 2015;9:765–79. https://doi.org/10.1586/17474124.2015.1028363.
Song. Standardizing management of hepatocellular carcinoma in China: devising evidence-based clinical practice guidelines. BioScience Trends. 2013;7:250–2. https://doi.org/10.5582/bst.2013.v7.5.250.
Zheng S, **e Q, Cheng J. Salvage liver transplant for hepatocellular carcinoma: rescues and benefits. Transl Gastroenterol Hepatol. 2018;3:65. https://doi.org/10.21037/tgh.2018.09.06.
Tong Y, Yang H, Xu X, Ruan J, Liang M, Wu J, Luo B. Effect of a hypoxic microenvironment after radiofrequency ablation on residual hepatocellular cell migration and invasion. Cancer Sci. 2017;108:753–62. https://doi.org/10.1111/cas.13191.
Villanueva A, Hernandez-Gea V, Llovet JM. Medical therapies for hepatocellular carcinoma: a critical view of the evidence. Nat Rev Gastroenterol Hepatol. 2013;10:34–42. https://doi.org/10.1038/nrgastro.2012.199.
Facciorusso A, Muscatiello N, Di Leo A, Barone M. Combination therapy with sorafenib and radiofrequency ablation for hepatocellular carcinoma: a glimmer of light after the storm trial? Am J Gastroenterol. 2015;110:770–1. https://doi.org/10.1038/ajg.2015.80.
Haga Y, Kanda T, Nakamura M, Nakamoto S, Sasaki R, Takahashi K, Wu S, Yokosuka O. Overexpression of c-Jun contributes to sorafenib resistance in human hepatoma cell lines. PLoS ONE. 2017;12:e0174153. https://doi.org/10.1371/journal.pone.0174153.
Liu Y, Zong Y, Yang Z, Luo M, Li G, Yingsa W, Cao Y, **ao M, Kong T, He J, Liu X, Lei J. ACS Sustain Chem Eng. 2019;7(3):3614–23. https://doi.org/10.1021/acssuschemeng.8b06586.
Koirala N, Das D, Fayazzadeh E, Sen S, McClain A, Puskas JE, Drazba JA, Mclennan G. Folic acid conjugated polymeric drug delivery vehicle for targeted cancer detection in hepatocellular carcinoma. J Biomed Mater Res. 2019;107A:2522–35. https://doi.org/10.1002/jbm.a.36758.
Yang D, Tian HY, Zang TN, Li M, Zhou Y, Zhang JF. Hypoxia imaging in cells and tumor tissues using a highly selective fluorescent nitroreductase probe. Sci Rep. 2017;7:9174. https://doi.org/10.1038/s41598-017-09525-2.
Yu F, Shang X, Zhu Y, Lou H, Liu Y, Meng T, Hong Y, Yuan H, Hu F. Self-preparation system using glucose oxidase-inspired nitroreductase amplification for cascade-responsive drug release and multidrug resistance reversion. Biomaterials. 2021;275:120927. https://doi.org/10.1016/j.biomaterials.2021.120927.
Holínková P, Pekař M. Fluorescence study of aggregation behaviour of cationic surfactant carbethopendecinium bromide and its comparison with cetyltrimethylammonium bromide. Tenside Surfactant Deterg. 2019;56:300–5. https://doi.org/10.3139/113.110633.
Liu T, Yang C-T, Dieguez L, Denman JA, Thierry B. Robust and flexible fabrication of chemical micropatterns for tumor spheroid preparation. ACS Appl Mater Interfaces. 2014;6:10162–71. https://doi.org/10.1021/am501390v.
Liu X, Wang C, Ma H, Yu F, Hu F, Yuan H. Water-responsive hybrid nanoparticles codelivering ICG and DOX effectively treat breast cancer via hyperthermia-aided DOX functionality and drug penetration. Adv Healthc Mater. 2019;8:1801486. https://doi.org/10.1002/adhm.201801486.
Kim E-H, Won J-H, Hwang I, Yu J-W. Cobalt chloride-induced hypoxia ameliorates NLRP3-mediated caspase-1 activation in mixed glial cultures. Immune Netw. 2013;13:141–7. https://doi.org/10.4110/in.2013.13.4.141.
Yoo S-Y, Yoo J-Y, Kim H-B, Baik T-K, Lee J-H, Woo R-S. Neuregulin-1 protects neuronal cells against damage due to CoCl2-induced hypoxia by suppressing hypoxia-inducible factor-1α and P53 in SH-SY5Y cells. Int Neurourol J. 2019;23:111–8. https://doi.org/10.5213/inj.1938190.095.
Li D, Liu S, Zhu J, Shen L, Zhang Qy, Zhu H. Folic acid modified TPGS as a novel nano-micelle for delivery of nitidine chloride to improve apoptosis induction in Huh7 human hepatocellular carcinoma. BMC Pharmacol Toxicol. 2021;22:1(2021). https://doi.org/10.1186/s40360-020-00461-y.
Maghsoudinia F, Tavakoli MB, Samani RK, Motaghi H, Hejazi SH, Mehrgardi MA. Bevacizumab and folic acid dual-targeted gadolinium-carbon dots for fluorescence/magnetic resonance imaging of hepatocellular carcinoma. J Drug Deliv Sci Technol. 2021;61:102288. https://doi.org/10.1016/j.jddst.2020.102288.
Li F, Wang F, Wu J. Sorafenib inhibits growth of hepatoma with hypoxia and hypoxia-driven angiogenesis in nude mice. Neoplasma. 2017;64:718–24. https://doi.org/10.4149/neo_2017_509.
Assaraf YG, Leamon CP, Reddy JA. The folate receptor as a rational therapeutic target for personalized cancer treatment. Drug Resist Updat. 2014;17:89–95. https://doi.org/10.1016/j.drup.2014.10.002.
Funding
Tha study was financially supported by Zhejiang Provincial Natural Science Foundation of China (LQ20H300004) and the National Natural Science Foundation of China (81973267).
Author information
Authors and Affiliations
Contributions
Tingting Meng: conceptualization, methodology, formal analysis, investigation, writing (original draft), and funding acquisition. Yinghong Li: methodology and validation. Ying Tian: conceptualization, validation, and writing (review & editing). Mingxing Ma: methodology writing (original draft). Xuwei Shang: methodology and validation. Hong Yuan: resources, supervision, and project administration. Fuqiang Hu: conceptualization, resources, writing (review & editing), supervision, project administration, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meng, T., Li, Y., Tian, Y. et al. A Hypoxia-Sensitive Drug Delivery System Constructed by Nitroimidazole and its Application in the Treatment of Hepatocellular Carcinoma. AAPS PharmSciTech 23, 167 (2022). https://doi.org/10.1208/s12249-022-02316-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-022-02316-7