Abstract
Background
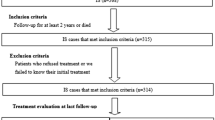
Ketogenic diet (KD) therapy is one of the main treatments for drug-resistant epilepsy. However, the KD therapy has been applied in only a small number of infantile spasm cases. In this large multicenter study, we investigated the efficacy of KD therapy in the treatment of infantile spasms.
Methods
In this retrospective, multicenter cohort study, clinical data from main epilepsy centers were analyzed. Patients were classified into different groups according to age, type of drug and whether glucocorticoid was used before initiation of KD.
Results
From October 2014 to March 2020, 481 patients (308 males and 173 females) with infantile spasms were treated with the KD therapy. The age of the patients ranged from 2 months to 20 years, with a mean age of 1 year and 10 months. The number of anti-seizure medications (ASMs) used before KD initiation ranged 0–6, with a median of 3. In different time from initiation(1, 3, 6, and 12 months), the rates of seizure freedom after KD were 6.9, 11.6, 16.0 and 16.8%, respectively (χ2 = 27.1772, P < 0.0001). There was a significant difference in the rate of seizure freedom between 3 months and 1 month (χ2 = 6.5498, P = 0.0105) groups, and 6 months and 3 months (χ2 = 3.8478, P = 0.0498) groups, but not between 12 months and 6 months (χ2 = 0.1212, P = 0.7278) groups. The rates of effectiveness were 44.7, 62.8, 49.1 and 32.0% (χ2 = 93.2674, P < 0.0001), respectively. The retention rates were 94.0, 82.5, 55.7 and 33.1% (χ2 = 483.7551, P < 0.0001), correspondingly. The rate of effectiveness and the retention rate of KD were significantly different among the 1, 3, 6 and 12 months. KD treatment was the first choice in 25 patients (5.2%), 55 patients (11.4%) started KD after the failure of the first ASM, 158 patients (32.8%) started KD after the failure of the second ASM, 157 patients (32.6%) started KD after the failure of the third drug, and 86 patients (17.9%) started KD after the failure of the fourth and more. The KD effect was not related to the number of ASMs used before KD startup (P > 0.05). Two hundred and eighteen patients (45.3%) failed to respond to corticotropin or glucocorticoid before initiation. There was no significant difference in the effectiveness rate at different time points between the group of KD therapy after glucocorticoid failure and the group after non-hormone failure (χ2 = 0.8613, P = 0.8348). The rate of adverse events of KD in 1, 3, 6, and 12 months after KD initiation were 22.3, 21.7, 16.8 and 6.9%, respectively. The adverse events mainly occurred during the first 3 months of KD, and the main adverse events were gastrointestinal disturbance and constipation.
Conclusions
The efficacy of the KD treatment for infantile spasms was not affected by age, medication, and glucocorticoid use before initiation. KD is one of the effective treatments for infantile spasms.
Trial registration
ChiCTR-IIR-16008342. Registered on 22 April, 2016 - Retrospectively registered, https://www.chictr.org.cn.
Similar content being viewed by others
Background
Infantile spasms (West syndrome) is a clinically common epilepsy syndrome of multiple causes, with typical manifestations of spasms, hypsarrhthmia (typical finding on interictal electroencephalogram [EEG]), and psychomotor development delay [1]. Although corticotropin (ACTH) and vigabatrin are used as the first-line treatment for infantile spasms, the epileptic spasms of some patients could not be effectively controlled. In addition, although ACTH is recommended as the first-line treatment in many countries, it has a high incidence of side effects and infantile spasms may relapse in nearly 33–50% of patients, which limits its generalized use [2,3,4]. Ketogenic diet (KD), a dietary model with high fat, low carbohydrate, and appropriate protein and other nutrients, can not only control seizures, but also improve EEG, development and cognition, thus having become an effective non-drug treatment for drug-resistant infantile spasms [5,6,7]. KD has developed rapidly since its introduction in China in 2004 [14, 16, 22,23,24]. Based on this report involving 17 epilepsy centers, although there was no overall difference in the efficiency and retention rate among centers (P > 0.05), in reality, the concept of KD management, the degree of family cooperation and the freedom of patients’ choice of medical treatment can all affect the retention rate. The retention rate further decreased from 3 months of KD, which may be related to the fact that the effect of KD was usually evaluated at 3–6 months. It is often more difficult to adhere to KD when the patient’s family believe that it is ineffective or the effect does not meet the expectation. In the withdrawal analysis, 20.0% of withdrawal were due to poor seizure improvement, which was similar to the previously reported results [6]; 17.1% of patients stopped KD due to family loss of follow-up and poor compliance / poor parental cooperation, which may be related to the difficulty in understanding KD by the elderly of some families or the difficulty of young parents in KD implementation. At the same time, 4.1% of patients received add-on of new ASMs or hospital transferring treatment, which not only reflected the flexibility of medical care, but also indicated that the effect of short-term KD might not meet the expectations of their families. Therefore, how to improve the effectiveness of KD individually, do a good job in the whole process of management, make evaluation of KD quantifiable, and simplify the operation are key issues that should receive more attention and be solved at present, because the KD retention rate is an important prognostic factor for maintenance of the curative effect [6].
In our study, adverse events occurred in some patients, but most of them were transient and tolerable, which were improved by conservative treatment or dietary adjustment. The incidence of hypoproteinemia, kidney stones, high triglycerides or cholesterol was low, and throughout the KD period, the incidence of adverse events was also low (< 2.0%). Good diet management and a reasonable scheme can reduce the occurrence of adverse events and increase the safety. Seizure control effect, expectation and dietary compliance play a key role in the continuation of KD. During KD, 8 patients died from different causes. Although the deaths were not directly caused by KD, careful observation is still needed during diet therapy to reduce complications and ensure a timely and appropriate treatment at its occurrence.
As a multicenter retrospective multicenter cohort study, the data might not be completely accurate. The long-term retention rate and the effectiveness rate, although similar to those reported in a prospective controlled study, were low in this study. In the future, multi-center large-sample clinical trials should be designed to study the related factors of retention rate and effectiveness, and further improve them.
Conclusions
In this study, we analyzed the effect and safety of KD treatment in 481 patients with infantile spasms from multicenter registered trial between 2014 to 2020. Most of the patients had drug-resistant epilepsy. At 1, 3, 6 and 12 months of KD, the seizure freedom rates were 6.9%, 11.6%, 16.0% and 16.8% respectively, and the retention rates were 94.0%, 82.5%, 55.7% and 33.1%, correspondingly. In summary, KD is effective in the treatment of infantile spasms with mild and acceptable side effects, and thus is suitable for clinical application. The anti-seizure effect and cognition improvement of KD therapy need to be further studied.
Availability of data and materials
Data available upon request to JX L.
Change history
07 March 2022
A Correction to this paper has been published: https://doi.org/10.1186/s42494-022-00081-5
Abbreviations
- KD:
-
Ketogenic diet
- ASMs:
-
Anti-seizure medications
- ACTH:
-
Corticotropin
- EEG:
-
Electroencephalogram
References
Hrachovy RA, Frost JD. Infantile epileptic encephalopathy with hypsarrhythmia (infantile spasms/west syndrome). J Clin Neurophysiol. 2003;20(6):408–25. https://doi.org/10.1097/00004691-200311000-00004.
Kossoff EH, Hedderick EF, Turner Z, Freeman JM. A case-control evaluation of the ketogenic diet versus ACTH for new-onset infantile spasms. Epilepsia. 2010;49(9):1504–9. https://doi.org/10.1111/j.1528-1167.2008.01606.x.
Lin HC, Young C, Wang PJ, Lee WT, Shen YZ. ACTH therapy for Taiwanese children with west syndrome -- efficacy and impact on long-term prognosis. Brain Dev. 2006;28(3):196–201. https://doi.org/10.1016/j.braindev.2005.07.002.
Ito M. Extremely low-dose ACTH therapy for west syndrome in Japan. Brain Dev. 2001;23(7):635–41. https://doi.org/10.1016/s0387-7604(01)00302-3.
Kossoff EH, Pyzik PL, Mcgrogan JR, Vining EPG, Freeman JM. Efficacy of the ketogenic diet for infantile spasms. Pediatrics. 2002;109(5):780–3. https://doi.org/10.1542/peds.109.5.780.
Eun SH, Kang HC, Dong WK, Kim HD. Ketogenic diet for treatment of infantile spasms. Brain Dev. 2006;28(9):566–71. https://doi.org/10.1016/j.braindev.2006.03.011.
Kossoff EH, Zupec-Kania BA, Auvin S, Ballaban-Gil KR, Bergqvist AGC, Blackford R, et al. Optimal clinical management of children receiving dietary therapies for epilepsy: Updated recommendations of the International Ketogenic Diet Study Group. Epilepsia Open. 2018;3(2). https://doi.org/10.1002/epi4.12225.
Dezhi C, **nguo L, Jianxiang L, Hu Y, Li B, Huang TS, et al. The ketogenic diet in the treatment of epileptic epilepsy. J Practical Pediatr. 2007;22(4):288–9.
Pires ME, Ilea A, Bourel E, Bellavoine V, Merdriu D, Berquin P, et al. Ketogenic diet for infantile spasms refractory to first-line treatments: an open prospective study. Epilepsy Res. 2013;105(1–2):189–94. https://doi.org/10.1016/j.eplepsyres.2012.11.009.
Cao D, Yan HU, Zhu Y, Zhao X, Li B, Chen L, et al. A randomized controlled study of corticotropin and ketogenic diet in the treatment of neonatal spasm. Chin J Clin Pediatr. 2011;18:1442–5.
Sharma S, Goel S, Kapoor D, Garg D, Panda I, Elwadhi A, et al. Evaluation of the Modified Atkins Diet for the Treatment of Epileptic Spasms Refractory to Hormonal Therapy: A Randomized Controlled Trial. J Child Neurol. 2021;36(8):686–91. https://doi.org/10.1177/08830738211004747.
Kim JA, Yoon JR, Lee EJ, Lee JS, Kim JT, Kim HD, et al. Efficacy of the classic ketogenic and the modified Atkins diets in refractory childhood epilepsy. Epilepsia. 2016;57(1):51–8. https://doi.org/10.1111/epi.13256.
Hong AM, Turner Z, Hamdy RF, Kossoff EH. Infantile spasms treated with the ketogenic diet:prospective single center experience in 104 consecutive infants. Epilepsia. 2010;51(8):1403–7. https://doi.org/10.1111/j.1528-1167.2010.02586.x.
Kayyali HR, Gustafson M, Myers T, Thompson L, Williams M, Abdelmoity A. Ketogenic diet efficacy in the treatment of intractable epileptic spasms. Pediatr Neurol. 2014;50(3):224–7. https://doi.org/10.1016/j.pediatrneurol.2013.11.021.
Lin SF, Qin J, Zhou SZ, Wang J, Liu ZS, Liao J. Ketogenic diet group of pediatric neurology society of Chiese medical association. Therapeutic effects of ketogenic diet on human health [J]. Prospective multicenter study of long-term ketogenic diet for refractory epilepsy in children. Chin J Pediatr. 2013;51(4):276–82. https://doi.org/10.3760/cma.j.issn.0578-1310.2013.04.008.
Li BM, Tong LL, Jia GJ, Lei GF, Yin P, et al. Short-term efficacy of ketogenic diet in the treatment of 31 children with refractory epileptic encephalopathy and its effect on electroencephalogram. Chin J Pediatr. 2013;51(005):362–6. https://doi.org/10.3760/cma.j.issn.0578-1310.2013.05.009.
Zhou YF, Gong XY, Zhou SZ, Wang Y, Chai YM, Yu LF,et al. Clinical characteristics and short-term efficacy of ketogenic diet in children with late-onset epileptic spasms. Chinese Journal of Evidence-based Pediatrics, 2015, 10(4):287–291.doi:3969/j.issn.1673–5501.2015.04.010.
Zhang J, Chen G, Wang J, Jiang Y, Yang Z, Xu K, et al. Efficacy of the ketogenic diet on ACTH- or corticosteroid-resistant infantile spasm: a multicentre prospective control study. Epileptic Disorders. 2021;23(2):337–45. https://doi.org/10.1684/epd.2021.1256.
Zhu XB, Chen O, Zhang DH, ** RF, Li FH, Wang YB, et al. A prospective study on the treatment of infantile spasms with first-line topiramate followed by low-dose ACTH. Epilepsy Res. 2011;93(2–3):149–54. https://doi.org/10.1016/j.2010.12.002.
Sharma S, Sankhyan N, Gulati S, Agarwala A. Use of the modified Atkins diet in infantile spasms refractory to first-line treatment. Seizure. 2012;21(1):45–8. https://doi.org/10.1016/j.seizure.2011.08.009.
Caraballo RH, Cersosimo RO, Sakr D, Cresta A, Escobal N, Fejerman N. Ketogenic diet in patients with Dravet syndrome. Epilepsia. 2010;46(9):1539–44. https://doi.org/10.1111/j.1528-1167.2005.05705.x.
Kang HC, Yong JK, Dong WK, Kim HD. Efficacy and safety of the Ketogenic diet for intractable childhood epilepsy: Korean multicentric experience. Epilepsia. 2005;46. https://doi.org/10.1111/j.0013-9580.2005.48504.x.
Wu GF, Hu JS, Wu SH, Zhu HM, Mao B, Sun D, et al. Effect and retention rate of ketogenic diet on infantile spasms. Chin J Pract Pediatr. 2017;032(10):780–3. https://doi.org/10.3760/cma.j.issn.2095-428X.2017.10.016.
Zha J, Cao FF, Yi ZS, Li XY, Chen H, Sun XL, et al. Clinical observation of ketogenic diet in the treatment of 112 children with drug-resistant epilepsy. Jiangxi Med J. 2020;55(10):1499–502. https://doi.org/10.3969/j.issn.1006-2238.2020.10.050.
Acknowledgements
We thank Professor Shichuo Li and Mr. Lirong Duan, Ms. Hui Zhang, China Association against Epilepsy for supporting and guiding the study. We appreciate and thank parents and children who consented for the ketogenic diet therapy.
Funding
This study was supported by the Sanming Project of Medicine in Shenzhen (SZSM201812005); Shenzhen Key Medical Discipline Construction Fund (No.SZXK033), and Shenzhen Fund for Guangdong Provincial High Level Clinical Key Specialties (No.SZGSP012).
Author information
Authors and Affiliations
Consortia
Contributions
JXL, JQ, BQY and YZY designed the study, and revised the manuscript. Other authors conducted the study. MY and JXL drafted the manuscript. The author(s) read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Medical Ethics Committee of Shenzhen Children’s Hospital, with a Clinical trial registration No. of ChiCTR-IIR-16008342. Informed consent was obtained from all patients in this study.
Consent for publication
Not applicable.
Competing interests
Author JQ is the editorial board member for Acta Epileptologica. Author JQ was not involved in the journal’s review of, or decisions related to this manuscript. JX L is a consultant of Shenzhen Zeneca Biotechnology Co., LTD. Shenzhen, China.
Additional information
The original online version of this article was revised: Declarations have been updated.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ye, Y., Sun, D., Li, H. et al. A multicenter retrospective cohort study of ketogenic diet therapy in 481 children with infantile spasms. Acta Epileptologica 4, 11 (2022). https://doi.org/10.1186/s42494-021-00077-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s42494-021-00077-7