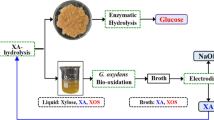
Abstract
Background
Xylo-oligomers are a kind of high value-added products in biomass fractionation. Although there are several chemical methods to obtain xylo-oligomers from biomass, the reports about the deep eutectic solvents (DESs)-mediated co-production of xylo-oligomers and fermentable sugars and the related kinetic mechanism are limited.
Results
In this work, glycolic acid-based DESs were used to obtain xylo-oligomers from corncob. The highest xylo-oligomers yield of 65.9% was achieved at 120 °C for 20 min, of which the functional xylo-oligosaccharides (XOSs, DP 2–5) accounted for up to 31.8%. Meanwhile, the enzymatic digestion of cellulose and xylan in residues reached 81.0% and 95.5%, respectively. Moreover, the addition of metal inorganic salts significantly accelerated the hydrolysis of xylan and even the degradation of xylo-oligomers in DES, thus resulting in higher selectivity of xylan removal. AlCl3 showed the strongest synergistic effect with DES on accelerating the processes, while FeCl2 is best one for xylo-oligomers accumulation, affording the highest xylo-oligomers yield of 66.1% for only 10 min. Furthermore, the kinetic study indicates that the ‘potential hydrolysis degree’ model could well describe the xylan hydrolysis processes and glycolic acid/lactic acid (3:1) is a promising solvent for xylo-oligomers production, in particular, it worked well with FeCl2 for the excellent accumulation of xylo-oligomers.
Conclusions
Glycolic acid-based deep eutectic solvents can be successfully applied in corncob fractionation with excellent xylo-oligomers and fermentable sugars yields on mild conditions, and the large amount of xylo-oligosaccharides accumulation could be achieved by specific process controlling. The strategies established here can be useful for develo** high-valued products from biomass.
Similar content being viewed by others
Background
As one of the main agricultural wastes, corncob is produced in large quantities. It is considered to be an important raw material for the production of high-valued biochemicals and energy fuels. Like the common lignocellulosic biomass, corncob is mainly composed of three major components, namely, cellulose, hemicellulose, and lignin [1, 2]. Hemicellulose of biomass is more sensitive and easily to be hydrolyzed; however, it is often neglected, because it is difficult to obtain high value-added products from it [3]. Hemicellulose is a miscellaneous polysaccharides containing branched polymers. It contains three types, and 65–85% of hemicellulose is made up of xylan backbones [4].The hydrolysis products of xylan include low xylan fragments (DP > 6), xylo-oligosaccharides (XOSs, DP < 6), and xylose, and the health benefits of XOSs have been reporte, including lowering blood cholesterol, increasing calcium absorption, antioxidant effects, maintaining gastrointestinal health, and reducing the risk of colon cancer. Also, they have toxic effects on human leukemia cells, and are benefits for patients suffering from type 2 diabetes [5]. Currently, there are different ways to obtain XOSs from biomass. For example, acquiring XOSs from poplar [6,7, Compound cellulase was purchased from Novozymes (China) and used as received. Corncob acquired from Guangdong, China was ground to a particle size of 65 mesh by a grinder. Then, the ground raw corncob was air-dried (water content was 6.0 wt%) before use. Choline chloride (ChCl, 98%) were purchased from Shanghai Macklin Biochemical Co., Ltd. (Macklin, Shanghai, China). Glycolic acid (98%), lactic acid (85%), guanidine hydrochloride (99%), ferrous chloride tetrahydrate (98%), aluminum chloride hexahydrate (99.99%), magnesium chloride hexahydrate (99.9%), glucose (99%), xylose (99%), and furfural (99%) were purchased from Aladdin Biochemical Technology Co., Ltd. (Aladdin, Shanghai, China). Xylobiose (98%) and Xylotriose (98%) were purchased from Shanghai Yuanye Bio-Technology Co., Ltd. (Yuanye, Shanghai, China). Xylotetraose (99%) and Xylopentaose (99%) were purchased from Shanghai ZZBIO Co., Ltd. (ZZBIO, Shanghai, China). Other chemicals were of the highest purity commercially available. Glycolic acid (G), choline chloride (C), lactic acid (L), guanidine hydrochloride (G), ferrous chloride tetrahydrate, aluminum chloride hexahydrate, and magnesium chloride hexahydrate were dried under vacuum at 80 °C for 5 h before use. The DES were prepared by mixing glycolic acid with choline chloride (GC), lactic acid (GL), and guanidine hydrochloride (GG) in a molar ratio of 1:1or 3:1. For the metal inorganic salts contained system, 5% (w/w) ferrous chloride tetrahydrate and aluminum chloride hexahydrate were added to GC (3:1), GL (3:1), and GG (3:1), respectively. Magnesium chloride hexahydrate could only dissolve in GL (3:1). The mixture was heated and stirred at a certain temperature in a closed flask until a homogenous colorless solution was formed. The prepared DES were then stored in a vacuum drying oven before use. Briefly, corncob samples were mixed with DES with a biomass loading of 5 wt%, and then the mixtures were stirred and kept at 120 °C for a specific time (10 min, 20 min, 40 min, 1 h, 2 h, 3 h, or 4 h). Once the treatment was completed, the residues were thoroughly washed with anhydrous ethanol and then water, and placed in a − 20 °C refrigerator for freeze-drying. After lyophilization, the samples were placed in a sealed bag and stored in a drying oven for the subsequent use. The cellulose, xylan, lignin, and ash contents of the corncob samples were determined according to the standard NREL analytical procedure, including acid hydrolysis, the subsequent HPLC, and gravimetric analysis [38], and these experiments were conducted in duplicate. The sugars were monitored using HPLC (Agilent 1260) equipped with a Bio-Rad Aminex HPX-87H column and a refractive index detector (Agilent 1260). The mobile phase was a 5 mM sulfuric acid aqueous solution, the flow rate was 0.5 mL/min, and the column and detector temperatures were 65 and 50 °C, respectively. The retention times for glucose and xylose were 11.4 and 12.2 min respectively. Enzymatic hydrolysis was implemented by mixing 20 mg of biomass and 8.3 U mL−1 cellulase in 7 mL of a citrate buffer (50 mmol L−1, pH 4.8) with stirring (120 rpm) at 50 °C. Aliquot samples (300 μL) were extraction at specified time spaces and boiled for 5 min to quench the enzymatic reaction. After being filtrated through a 0.22 μm membrane, the glucose and xylose concentrations were detected using the HPLC, as described above. All reactions were performed in duplicate, and the related data showed as mean values with standard derivations. The polysaccharide digestibility were calculated as follows: Polysaccharide digestibility (%) = (released sugar amount)/Theoretic sugar amount in the sample used for enzymatic hydrolysis × 100. FT-IR patterns of corncob and DES to treat solids were tested from an FT-IR (Nexus Thermo Nicolet, USA). The samples for test were combined with KBr (1/10 mass ratio), evenly grinding both and to press into flakes with 7 MPa and 30 s. 32 background and scans were taken from 400 to 4000 cm−1. To collect the total xylo-oligomers, deionized water of a certain volume was added after the reaction completed, and the mixture was centrifuged at 10,000 g for 5 min. The supernatant was collected. The xylo-oligomers measurement was conducted according to the previous report [18]. Briefly, the supernatant was obtained by centrifugation. Then, the supernatant was hydrolyzed with 4% H2SO4 at 121 °C for 1 h. The total xylo-oligomers yield was calculated based on the discrepancy between the content of xylose before and after hydrolysis. The equation was showed as follows: XOSs (DP 2–5) were analyzed based on the previous report [39]. The XOSs (DP 2–5) were monitored using HPLC (Waters 1525) equipped with an Agilent Hi-Plex Na column and a refractive index detector. The mobile phase was water, the flow rate was 0.3 mL/min, and the column and detector temperatures were 80 °C and 50 °C, respectively. The equation was expressed as follows: Furfural were analyzed based on our previous report [24]. The furfural was monitored using HPLC (Waters 2695) equipped with an Agilent C-18 column and a DAD detector at 280 nm. The mobile phase was acetonitrile/water (15/85, v/v), and the flow rate was 1 mL/min−1. The equation was expressed as follows: Kinetic models used in this study were referred as the previous report [16]. Briefly, DES of six kinds, GC (3:1), GL (3:1), GG (3:1), GL (3:1)/FeCl2·4H2O, GL (3:1)/AlCl3·6H2O, and GL (3:1) /MgCl2·6H2O were kept at 120℃ for 10 min, 20 min, 40 min, 1 h, 2 h, 3 h, and 4 h to treat corncob, and the content percentage of xylan in the residue and the concentration of xylo-oligomers in supernatant at different times were determined according to the above-mentioned methods. Origin 95 was used to fit the models. Xylan solubility and xylo-oligomers concentration were fitted according to the formula listed as follows: where Hd is the ‘potential degree of hydrolysis’ of xylan, and 0 ≤ Hd ≤ 1; C0 is the initial concentration of xylan in the pseudo-homogeneous system (mg/ml); Cxos is the concentration of the total xylo-oligomers in the treatment liquor (mg/mL); 1.136 is the conversion coefficient of xylan into xylose.Materials and methods
Materials
DES preparation
Treatment of corncob with DES
Compositional analysis of the corncob samples
Enzymatic hydrolysis of the corncob samples
Fourier transform infrared spectroscopy (FT-IR) analysis
Analysis of the total xylo-oligomers and XOSs (DP 2–5)
Mass balance of xylan
Kinetic study of xylan hydrolysis and xylo-oligomers accumulation
Availability of data and materials
All the data are available for publication and information used from online resources has been cited properly. All data generated or analyzed during this study are included in this published article.
Abbreviations
- DES:
-
Deep eutectic solvents
- GC:
-
Glycolic acid/choline chloride
- GL:
-
Glycolic acid/lactic acid
- GG:
-
Glycolic acid/guanidine hydrochloride
- GC (3:1)/FeCl2·4H2O:
-
Glycolic acid/choline chloride/ferrous chloride tetrahydrate
- GC (3:1)/AlCl3·6H2O:
-
Glycolic acid/lactic acid/aluminum chloride hexahydrate
- GG (3:1)/MgCl2·6H2O:
-
Glycolic acid/guanidine hydrochloride/magnesium chloride hexahydrate
References
Lynam JG, Kumar N, Wong MJ. Deep eutectic solvents’ ability to solubilize lignin, cellulose, and hemicellulose; thermal stability; and density. Bioresour Technol. 2017;238:684–9. https://doi.org/10.1016/j.biortech.2017.04.079.
Dias RM, Sosa F, Costa M. Dissolution of lignocellulosic biopolymers in ethanolamine-based protic ionic liquids. Polym Bull. 2020;77:3637–56. https://doi.org/10.1007/s00289-019-02929-2.
Cyba B, Yxyb C, Lhza B, Spoa B, Zjza B, Jia O. Comprehensive utilization of corncob for furfuryl alcohol production by chemo-enzymatic sequential catalysis in a biphasic system. Bioresour Technol. 2020;319:124156. https://doi.org/10.1016/j.biortech.2020.124156.
Ahring BK. A thermochemical pretreatment process to produce xylooligosaccharides (XOS), arabinooligosaccharides (AOS) and mannooligosaccharides (MOS) from lignocellulosic biomasses. Bioresour Technol. 2012;112:285–92. https://doi.org/10.1016/j.biortech.2012.01.162.
Luciana S, Constanza H, Romina C, Sebastián B, Benjamín A, Oriana S. Xylooligosaccharides from lignocellulosic biomass: a comprehensive review. Carbohydr Polym. 2020;251:117118. https://doi.org/10.1016/j.carbpol.2020.117118.
Wang J, Xu Y, Meng X, Pu Y, Zhang J. Production of xylo-oligosaccharides from poplar by acetic acid pretreatment and its impact on inhibitory effect of poplar lignin. Bioresour Technol. 2021;323:124593. https://doi.org/10.1016/j.biortech.2020.124593.
Hao X, Wen P, Wang J, Wang J, Zhang J. Production of xylooligosaccharides and monosaccharides from hydrogen peroxide-acetic acid-pretreated poplar by two-step enzymatic hydrolysis. Bioresour Technol. 2019;297:122349. https://doi.org/10.1016/j.biortech.2019.122349.
Hong LA, Wy A, **n L, Jza B, Yong X, Jzab C. Optimized production of xylooligosaccharides from poplar: a biorefinery strategy with sequential acetic acid/sodium acetate hydrolysis followed by xylanase hydrolysis. Bioresour Technol. 2022;347:126683. https://doi.org/10.1016/j.biortech.2022.126683.
Zhang H, Xu Y, Yu S. Co-production of functional xylooligosaccharides and fermentable sugars from corncob with effective acetic acid prehydrolysis. Bioresour Technol. 2017;234:343. https://doi.org/10.1016/j.biortech.2017.02.094.
Wen P, Zhang T, Wang J, Lian Z, Zhang J. Production of xylooligosaccharides and monosaccharides from poplar by a two-step acetic acid and peroxide/acetic acid pretreatment. IndCropProd. 2019;152:87. https://doi.org/10.1186/s13068-019-1423-x.
Flávia Sanchez Penalva Neto P, Roldána IUM, Galán JPM, Monti R, Oliveira SC, Masarina F. Model-based optimization of xylooligosaccharides production by hydrothermal pretreatment of eucalyptus by-product. IndCropProd. 2020;154:112707. https://doi.org/10.1016/j.indcrop.2020.112707.
Yan SA, Lf A, Peng WA, Cl A, Cha B, Zhe LA, Ss C, Qiang Y. Efficient production of xylooligosaccharides rich in xylobiose and xylotriose from poplar by hydrothermal pretreatment coupled with post-enzymatic hydrolysis. Bioresour Technol. 2021;342:125955. https://doi.org/10.1016/j.biortech.2021.125955.
Zhang X, Zhang W, Lei F, Yang S, Jiang J. Coproduction of xylooligosaccharides and fermentable sugars from sugarcane bagasse by seawater hydrothermal pretreatment. Bioresour Technol. 2020;309:123385. https://doi.org/10.1016/j.biortech.2020.123385.
Kumar B, Bhardwaj N, Verma P. Microwave assisted transition metal salt and orthophosphoric acid pretreatment systems: generation of bioethanol and xylo-oligosaccharides. RenewEnerg. 2020;158:574–84. https://doi.org/10.1016/j.renene.2020.05.006.
Zhang W, Lei F, Li P, Zhang X, Jiang J. Co-catalysis of magnesium chloride and ferrous chloride for xylo-oligosaccharides and glucose production from sugarcane bagasse. Bioresour Technol. 2019;291:121839. https://doi.org/10.1016/j.biortech.2019.121839.
Zhao X, Zhou Y, Liu D. Kinetic model for glycan hydrolysis and formation of monosaccharides during dilute acid hydrolysis of sugarcane bagasse. Bioresour Technol. 2012;105:160–8. https://doi.org/10.1016/j.biortech.2011.11.075.
Liu X, Wei W, Wu S. Synergism of organic acid and deep eutectic solvents pretreatment for the co-production of oligosaccharides and enhancing enzymatic saccharification. Bioresour Technol. 2019;290:121775. https://doi.org/10.1016/j.biortech.2019.121775.
Shen BZ, Hou SW, Yuan J, Yang CD, Yan S, Zhe L, Huang CX, Lai CH, Qiang Y. Synergistic effects of hydrothermal and deep eutectic solvent pretreatment on co-production of xylo -oligosaccharides and enzymatic hydrolysis of poplar. Bioresour Technol. 2021;341:125787. https://doi.org/10.1016/j.biortech.2021.125787.
Saeman JF. Kinetics of wood saccharification-hydrolysis of cellulose and decomposition of sugars in dilute acid at high temperature. Indengchem. 1945;37:43–52.
Chang X, Bai Y, Wu R, Liu D, Zhao X. Heterogeneity of lignocellulose must be considered for kinetic study: a case on formic acid fractionation of sugarcane bagasse with different pseudo-homogeneous kinetic models. RenewEnerg. 2020;162:2246–58. https://doi.org/10.1016/j.renene.2020.10.029.
Maloney MT, Chapman TW, Baker AJ. Dilute acid hydrolysis of paper birch: kinetics studies of xylan and acetyl-group hydrolysis. BiotechnolBioeng. 2010;27:355–61. https://doi.org/10.1002/bit.260270321.
Liang X, Zhu Y, Qi B, Li S, Wan Y. Structure-property-performance relationships of lactic acid-based deep eutectic solvents with different hydrogen bond acceptors for corn stover pretreatment. Bioresour Technol. 2021;336:125312. https://doi.org/10.1016/j.biortech.2021.125312.
Hou XD, Li AL, Lin KP, Wang YY, Cao SL. Insight into the structure-function relationships of deep eutectic solvents during rice straw pretreatment. Bioresour Technol. 2017;249:261. https://doi.org/10.1016/j.biortech.2017.10.019.
Li AL, Hou XD, Lin KP, Zhang X, Fu MH. Rice straw pretreatment using deep eutectic solvents with different constituents molar ratios: biomass fractionation, polysaccharides enzymatic digestion and solvent reuse. J Biosci Bioeng. 2018;126:346–54. https://doi.org/10.1016/j.jbiosc.2018.03.011.
Tang S, Dong Q, Fang Z, Miao ZD. Complete recovery of cellulose from rice straw pretreated with ethylene glycol and aluminum chloride for enzymatic hydrolysis. Bioresour Technol. 2019;284:98–104. https://doi.org/10.1016/j.biortech.2019.03.100.
Wang ZK, Shen XJ, Chen JJ, Jiang YQ. Lignocellulose fractionation into furfural and glucose by AlCl3-catalyzed DES/MIBK biphasic pretreatment. Int J Biol Macromol. 2018;117:721–6. https://doi.org/10.1016/j.ijbiomac.2018.05.232.
Yu LL, Ta YW, Ge H, Yang LY, Eng KN. Deep eutectic solvent and inorganic salt pretreatment of lignocellulosic biomass for improving xylose recovery. Bioresour Technol. 2018;249:818–25. https://doi.org/10.1016/j.biortech.2017.07.165.
Yan ZY, **an KZ, Peng FL, Fu HL, Jian XJ. Co-production of xylooligosaccharides and activated carbons from camellia oleifera shell treated by the catalysis and activation of zinc chloride. Bioresour Technol. 2020;306:123131. https://doi.org/10.1016/j.biortech.2020.123131.
Ai JS, Yi N, Chun XL, Zi H. Preparation of butyl chloride from butanol and hydrochloric acid using ionic liquids as catalyst. Chin J Chem Eng. 2008;16:151–4. https://doi.org/10.1016/S1004-9541(08)60054-3.
Chao Z, Qian JS, Zhong QM, Bin L, **ang J. Physical and chemical characterizations of corn stalk resulting from hydrogen peroxide presoaking prior to ammonia fiber expansion pretreatment. Ind Crop Prod. 2016;83:86–93. https://doi.org/10.1016/j.indcrop.2015.12.018.
Tainise V, Lourenon A, Fabricio A, Hansel BTA, Da Ramos CGSLP, Dmwle B, Magalhes B. Hardwood and softwood kraft lignins fractionation by simple sequential acid precipitation. Sep Purif Technol. 2015;154:82–8. https://doi.org/10.1016/j.seppur.2015.09.015.
Seesuriyachan P, Kawee-Ai A, Chaiyaso T. Green and chemical-free process of enzymatic xylooligosaccharide production from corncob: enhancement of the yields using a strategy of lignocellulosic destructuration by ultra-high pressure pretreatment. Bioresour Technol. 2017;241:537–44. https://doi.org/10.1016/j.biortech.2017.05.193.
Sun RC, Sun XF, Wen JL. Fractional and structural characterization of lignins isolated by alkali and alkaline peroxide from barley straw. J Agric Food Chem. 2001;49:5322. https://doi.org/10.1021/jf010645y.
**g Z, Wang X, Hu J, Qian L, Shen D, Rui X. Thermal degradation of softwood lignin and hardwood lignin by TG-FTIR and Py-GC/MS. Polym Degrad Stab. 2014;108:133–8. https://doi.org/10.1016/j.polymdegradstab.2014.06.006.
Seera SDK, Naik PK, Kundu D, Banerjee T, Gami P. Synthesis and characterization of xylan-gelatin cross-linked reusable hydrogel for the adsorption of methylene blue. Carbohydr Polym. 2021;256:177520. https://doi.org/10.1016/j.carbpol.2020.117520.
Kang KE, Park DH, Jeong GT. Effects of inorganic salts on pretreatment of Miscanthus straw. Bioresour Technol. 2013;132:160–5. https://doi.org/10.1016/j.biortech.2013.01.012.
Kamireddy SR, Li J, Tucker M, Degenstein J, Ji Y. Effects and mechanism of metal chlorides on pretreatment and enzymatic digestibility of corn stover. Ind Eng Chem Res. 2013;52:1775–82. https://doi.org/10.1021/ie3019609.
Sluiter A, Hames B, Ruiz R, Scarlata C, Templeton D, Crocker D. Determination of structural carbohydrates and lignin in biomass NREL/TP-510–42618. Lab Anal Proced Gold CO. 2008. https://doi.org/10.1021/jf1008023.
Lin QX, Li HL, Ren JL, Deng AJ, Li WY, Liu CF, Sun RC. Production of xylooligosaccharides by microwave-induced, organic acid-catalyzed hydrolysis of different xylan-type hemicelluloses: optimization by response surface methodology. Carbohydr Polym. 2017;157:214–25. https://doi.org/10.1016/j.carbpol.2016.09.091.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 22078068).
Author information
Authors and Affiliations
Contributions
HQD contributed majorly to perform the experiments, data analysis, and writing of the first draft of the manuscript. XHL and JTF helped to perform corncob pretreatment and xylo-oligomers determination experiments. PZF, JJG, and HLL helped to perform saccharification experiment. LHL and LHL helped to perform kinetic experiment. XHL and XDH analyzed the data and revised the manuscript. XDH and WYL supervised the project and revised the final manuscript. All authors read and approved the final manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Deng, HQ., Lin, XH., Fan, JT. et al. Glycolic acid-based deep eutectic solvents boosting co-production of xylo-oligomers and fermentable sugars from corncob and the related kinetic mechanism. Biotechnol Biofuels 16, 126 (2023). https://doi.org/10.1186/s13068-023-02369-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13068-023-02369-1