Abstract
Benefiting from the complex system composed of various constituents, medicament portions, species, and places of origin, traditional Chinese medicine (TCM) possesses numerous customizable and adaptable efficacies in clinical practice guided by its theories. However, these unique features are also present challenges in areas such as quality control, screening active ingredients, studying cell and organ pharmacology, and characterizing the compatibility between different Chinese medicines. Drawing inspiration from the holistic concept, an integrated strategy and pattern more aligned with TCM research emerges, necessitating the integration of novel technology into TCM modernization. The microfluidic chip serves as a powerful platform for integrating technologies in chemistry, biology, and biophysics. Microfluidics has given rise to innovative patterns like lab-on-a-chip and organoids-on-a-chip, effectively challenging the conventional research paradigms of TCM. This review provides a systematic summary of the nature and advanced utilization of microfluidic chips in TCM, focusing on quality control, active ingredient screening/separation, pharmaceutical analysis, and pharmacological/toxicological assays. Drawing on these remarkable references, the challenges, opportunities, and future trends of microfluidic chips in TCM are also comprehensively discussed, providing valuable insights into the development of TCM.
Similar content being viewed by others
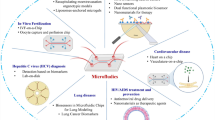
Introduction
With the remarkable success of traditional Chinese medicine (TCM) in combating the COVID-19 pandemic, an increasing number of individuals are turning to this ancient yet potent form of medicine for treatment, not only in Asia but across the globe [31]. TCMC aims to gradually discover and confirm the composition of prominent chemical substance groups within the overall chemical substance group of the compound. This is achieved by analyzing the interplay between chemical information flow and multi-parameter biological information flow across different levels of chemical substance groups. According to the perspective of TCMC, the compound of TCM, as an overall chemical substance group, can be seen as an organic combination of several related sub-chemical substance groups, including the global chemome, effective chemome, and effective constituent group. These sub-chemical substance groups manifest in two primary forms: medicinal materials and components (also known as effective components) that represent specific characteristics of the compound. Hence, in this sense, the compatibility of effective compositions and the combination of TCM are essentially aligned [32].
Key features reflecting synergy or antagonism were generated from the global chemome of TCM, the effective chemome, and the effective constituents group. To distinguish these ingredients, spectrum-effect relationships were used to link biological effects and pharmacodynamic data [66], Copyright, 2022, Elsevier; D Christmas tree model [67]; E PDMS chip fabrication [68], Copyright, 2022, Elsevier; F Six typical glass microstructure fabrication techniques [55]; G 3D Printed microfluidic chip [69]
Surface effects are crucial in microfluidics due to the high surface-to-volume ratios involved. Surface tension arises from the imbalanced interactions between molecules at the material's surface. Generally, strong polar liquids exhibit higher surface tension than nonpolar liquids, and surface tension can be reduced through chemical modifications such as fluorination. As the temperature of the fluid increases, surface tension decreases due to enhanced Brownian motion of solvent molecules. The mechanical imbalance on the liquid surface drives the formation of drops, with smaller diameter drops exhibiting higher surface tension. From this principle, droplet microfluidics has been developed as an analytical technique that manipulates mutually immiscible phases within a microchannel (Fig. 3A, B) [49,50,51].
Additionally, operating at the micro-scale leads to a significant reduction in the mass/volume of chemical and biological reagents, a phenomenon known as the scaling law. When a system decreases in size proportionally in all dimensions, the volume of the system will decrease cubically.
Construction of microfluidic chips
Closed or semi-closed structures, such as micro-tubes, microneedles, and microfluidic chips, serve as standard platforms for microfluidics researchers [52, 53]. Rigid materials ranging from glass to silica were previously exploited as the materials of chips for their optical properties, good insulating properties, high resistance to mechanical stress, high surface stability, and high solvent compatibility. The fabrication involved photolithography and wet etching protocols (Fig. 3F) [54, 55]. Computer software such as AutoCAD was employed to design the devices on different slides, and chemical etching [56, 57], laser etching [58], and fused operation [59] are viable methods for constructing designed patterns consisting of different channels, holes and chambers on glass/silica chips [60,61,62]. Surface modification offers the advantage of creating smoother bottom surfaces, reducing residues from bio-samples, and improving the optical properties of the chips [55, 63]. Hydroxylation is commonly achieved using a piranha solution, and plasma treatment and silanization reagents are used for surface hydrophobic modification. Compared with glass chips, silica chips offer advantages in applications related to refraction [64].
The solid materials were complex to fabricate sophisticated channels and chambers. Polymerics including polydimethylsiloxane (PDMS), polymethyl methacrylate, and cyclic olefin copolymer were employed to construct microfluidic chips due to their optical transparency, mouldability, and cost-effectiveness. Among them, PDMS is the most commonly used material. Cured PDMS exhibits biocompatibility and allows for the permeation of liquids and gases, making it suitable for in vitro tissue culture. Constructing PDMS chips through soft lithography involves two essential procedures [70, 71]. Firstly, a stamp (referred to as a master mold) with specific channels, chambers, or other complex structures is fabricated through a photography process. Photosensitive materials deposited on substrates can be classified as positive or negative photoresists. Following exposure, the pattern remains in the case of negative photoresist, while it dissolves in the case of positive photoresist. Secondly, a mixture of PDMS and curing agent is poured into the obtained stamp, and polymerization is induced by heat (Fig. 3E). Benefited from the functional group of PDMS, the peeling-off polymer was usually bonded in glass/silicon, and there were many hybrid chips, including glass-PDMS [72], silico-glass [73], sandwich (glass-PDMS-glass) [74]. The limitation of PDMS (e.g., weak chemical compatibility, absorption of molecules) presented opportunities for surface modification of PDMS or other materials on chips [75]. For example, with the coating layer formed by tetraethoxysilane and methyltriethoxysilane, the chemical resistance of the PDMS chip was improved significantly [76, 77]. Additionally, 3D printing has been increasingly exploited for fabricating microfluidic chips and was regarded as one of the most promising construction methods (Fig. 3G) [69].
The manipulation of multi-stream flows within microchannels is facilitated by strategically designed junctions, allowing for precise control following the introduction of liquid into the chip via inlet ports [66, 78]. The multi-stream laminar flow on a chip has the capacity to generate varying concentration of mixture, including linear and nonlinear gradients (e.g., bell-shaped) [79]. It finds widespread application in the preparation of drugs with diverse concentrations or compositions. T-shaped junction was employed to encapsulate molecules or cells within single droplets, thereby serving as a powerful tool for single-cell analysis (Fig. 3A–D). Moreover, the coaxial micro flow was accessible and the insoluble liquids fluid could be tailored to meet specific functional requirement, such as replicating the physiological characteristics of blood vessels in vitro [110, 111]. Furthermore, cell-on-a-chip technology has provided a convenient means to observe and validate intercellular interactions (e.g., cellular metastasis, cascade secretion of insulin) among different cells through flexible and physiologically mimicked co-culture patterns [61, 112]. With low sample consumption and high throughput capabilities, microfluidic chips are conducive to real-time assays of cellular physiopathology, thereby accelerating the discovery of significant cell responses to microenvironmental alterations [113]. Additionally, microfluidics could enhance drug toxicity evaluations on cells [114], indicating that mimicking cellular microenvironments using microfluidics will further improve drug evaluation systems.
Organ/organoids-on-a-chip
The development of biological affinity materials has made it possible to culture organs on chips [115]. Organ-on-a-chip, a result of integrating biology with microtechnology, has been used to simulate crucial aspects of human physiology [116]. Drawing upon our anatomical and physiological understanding of tissues and organs in animal bodies, in vitro organ culturing has been achieved by engineering different types of cells within specialized devices, including commercial instruments and laboratory-customized chips. The culture conditions are precisely controlled to maintain the physiological state of the organs. For instance, introducing a permeable endothelial barrier enables vascular flow, successfully linking multiple tissues and maintaining their structural and functional phenotypes for an extended period, up to a month [117]. The organ-on-a-chip system is capable of enabling communication between different tissues and maintaining stability over a long period of time. This provides an ideal platform for quantitative prediction of pharmacokinetics and pharmacodynamics during the pre-clinical stage [118]. Due to a shortage of monolayer cell cultures in drug screening and safety evaluation, there has been increased exploration of 3D culture techniques. Microfluidic chips simplify the multi-tissue culture process and offer more controllable properties, enabling a closer approximation of the physiological state (Fig. 4C–E). This advancement holds promise for capturing more comprehensive information regarding drug responses, including often overlooked toxicities [119].
Different from organ-on-a-chip, which performed function and application based on the mastered knowledge about organ structure and derived from the array of multiple cells, organoids-on-a-chip depended on self-organization and differentiation of homogenous pluripotent stem cells within a microfluidic chip to simulate the structural and functional characteristics of target organs [120]. Organoids generated from human pluripotent stem cells hold great promising model in the application of biomedical [121]. Blood flow is one of the critical factors of tissue development, influencing the shape and function of organs, and has the potential to be accurately simulated (Fig. 4D). Organoids-on-a-chip, which mimic blood flow through microfluidics and are generated from stem cells, provide a more comprehensive platform for manufacturing and investigating organ and tissue responses to various stimuli, including endogenous and exogenous metabolites (e.g., glucose) [122].
By utilizing microfluidic chips, it has become possible to simulate complex body structures in vitro, including airways, intestines, and other cavity-like structures [126, 127]. Building upon the successes achieved in microfluidic chip technology and the culture of cells, organs, and organoids in vitro, the concept of human-on-a-chip has emerged, gaining momentum due to its potential applications in biomedicine (Fig. 4F).
Pharm-lab-on-a-chip
Microfluidic chips provide a versatile integration platform for the miniaturization of drug analysis and subsequent biological response assays. This is accomplished by utilizing appropriate assay technical media through macro-precise instruments or mini-sensors. The integration and automation of microfluidic chips have played a significant role in the emergence and adcancement of Pharm-on-a-chip technology [129]. In the intestine, there is drug absorption as well as efflux. Gastrointestinal cells derived from patients, incorporating personal genetic information and phenotypes, have emerged as powerful tools for drug selection using organoids-on-a-chip [130]. Intestinal cells also possess drug metabolism enzymes alongside liver cells. A novel culture pattern based on chips has showcased a more accurate representation of metabolism enzyme activity than Caco-2 monolayer cells [130], bringing it closer to real-world applications. Combining drugs is a common medical practice, but understanding its mechanism and potential harm to the body remains challenging. The flexible co-culture of gastrointestinal cells with other cell types, such as liver cells, presents an appealing approach for researchers. Using a culture solution that contains both target constituents and their metabolites simplifies the procedure, making it easier to obtain and analyze, and also improves throughput. This approach is conducive to simulating the process of drug absorption in the intestines [131].
The liver and kidney play crucial roles in drug metabolism as essential organs. It is challenging to evaluate the toxic effects of drug metabolites through conventional static well plate cultures, especially when combined with compound separation. However, using microchannels that connect different chambers, where cells/organoids/organs can coexist in vitro, has made it possible to assess potential pharmacological or toxic effects, even at trace levels of metabolites. For instance, Aflatoxin B1 exhibits no apparent toxicity on the kidney, but its metabolites harm the kidney in reverse. The hepatic-kidney chip demonstrated the indirect toxic effect of Aflatoxin B1, vividly illustrating the feasibility of a multi-organ chip in drug metabolism research [132]. By utilizing microfluidics, the transmission of trace drug metabolites becomes more efficient, eliminating time-consuming procedures such as separation and evaluation of drug metabolites. Additionally, the co-culture chip of bacteria and gastrointestinal cells, based on the intestine-chip, serves as another powerful platform for drug metabolism and screening of active metabolites.
When it comes to the distribution of medicines, it is necessary to identify the target organ of the drug. However, ethical concerns arise due to the extensive use of animal testing in drug distribution research, resulting in the sacrifice of numerous animals during pre-clinical studies. The 3D simulation of physical structures, ranging from monolayer vesicles and tissues to bio-barriers, provides a valuable approach for visualizing or predicting drug distribution in vivo. Among these structures, the blood–brain barrier (BBB) is paramount. Simulating the BBB is beneficial for understanding the effectiveness of drugs in treating brain diseases during the pre-clinical stage while saving time and resources. For instance, the BBB-glioma microfluidic chip, which consists of primary human brain microvascular endothelial cells, primary human astrocytes, primary human brain vascular pericytes, and glioma U251 cells, has been successfully employed to assess drug permeation [133].
To gain a deeper understanding of TCM combination theories about chemical compounds, multiple factors need to be taken into consideration. These factors include, but are not limited to, the following: (1) Codissolution and inhibition of compounds during the extraction process; (2) Interactions that occur during drug absorption; (3) The complex metabolic relationship of TCM constituents as substrates for drug-metabolizing enzymes; (4) Synergistic and antagonistic pharmacological effects. Among these factors, the last item is particularly important as it entails elucidating the intricate relationships between TCM and their targets (cells/tissues/organs), as well as the relationships between these targets and other entities.
Microfluidic chips offer significant advantages in simulating physiological structures. By utilizing cell co-culture and external hardware-assisted molding methods, researchers have successfully constructed in vitro models of intestinal cell barrier, blood–brain barriers, and liver tissues [124, 135]. The phase separation of complex components and selective quantification of specific chemical compositions is facilitated by laminar flow [99, 102]. For instance, an induced phase separation extraction platform was established and applied in the analysis of TCM. This platform operated within a 5 × 2 cm chip featuring microchannels measuring 100 μm in width and 40 μm in depth. According to the results, the extraction efficiency of model compounds (chlorogenic acid, epigallocatechin gallate, rutin, quercetin, santonin, and alizarin) exceeded 90%. This method were also operated in the analysis of Scutellaria baicalensis extract. Aglycones (baicalein, wogonin) and glycosides (baicalin, wogonoside) were separated into organic phase and aqueous phase, respectively [136]. Notably, this approach significantly reduces sample and solvent consumption to microliter quantities, with an analytical period of less than 1 min. Furthermore, the miniaturization structure on chips help simplify the analytical system and decrease data variability, making them ideal for intra-batch measurements of precious medicinal herbs (e.g., Ginseng) [135]. In addition, this platform facilitated the comparison of the pharmacological effects of Schizonepeta tenuifolia obtained different region such as Anhui, Henan, and Yunan province in China. Performing all procedures on a micro-scale chip also enabled convenient tracing of metabolites of natural products and monitoring of the dynamic endogenous metabolism of biological tissues, owing to the powerful integration features of microfluidics [138].
Facing the challenges in clarifying active compounds of TCM consisting of plant, animal, and mineral, a proposed approach for quality evaluation based on actual effects was presented. Enzyme assays are a popular method for assessing drug effects due to their ability to measure target enzyme activation or inhibition, providing insight into drug efficacy. However, enzymes are fragile, and it isn't easy to recover proteins in vitro. A minor reaction system is needed to reduce protein loss and meet usage requirements. Microfluidic chips provide an ideal solution by offering a suitable chamber for protein and small molecule incubation from TCM. In the QC of QiShen YiQi Pills (Fig. 6A), 10 μL magnetic beads and 10 μL thrombin or ACE solution were added to form enzyme-magnetic bead complexes on a chip. The micro fluid was controlled by fluidic bridge and pneumatic valve. Subsequently, 10 μL of tested sample were pump in channel and incubated with enzyme. Following this incubation period, the reaction solution was recovered, and the absorbance was measured to assess quality. Simultaneously, the enzymes were retained in the chamber and remained available for further use. The versatile chip was utilized to explore potency variations across different batches of QiShen YiQi Pills. The results showed that this approach exhibited superior discrimination ability for aberrant samples compared to chromatographic fingerprinting techniques [108].
Demonstration of microfluidic chips in TCM. A Ligand fishing and quality control chip [108]. B Concentration gradient chip [144], Copyright, 2019, Elsevier; C Simulating of tumor microenvironment to evaluation of antimetastatic effects of TCM [147], Copyright, 2014, American Chemical Society; D Efficacy evaluation of TCM by biomimetic BBB chip [133], Copyright, 2023, Elsevier. E Endogenous and exogenous metabolites of cells were identified and monitored in real-time by the Chip-MS system [157], Copyright, 2022, American Chemical Society; F Cells co-culture chip for efficacy assay of TCM metabolites [106]
Managing the risks associated with TCM substances is a significant field of QC. Various analytical methods, including ICP-MS, HPLC-FLD, and GC, are employed to assay heavy metals, aflatoxins, and pesticide residues, respectively. However, the determination of risk substances is typically carried out using complex instruments, which can limit the timeliness of TCM quality evaluation. Benefited from emerged surface modification, it was feasible to immobilize antibodies on a chip, leading to the development of a novel immunoassay chip that was utilized in the analysis of aflatoxins in TCMs that are rich in lipids (e.g., Semen Platycladi, Pericarpium Citri Reticulataeas, Hellebore, and Semen Coicis) [139].
Screen of active ingredients
The effectiveness of TCM was validated by clinical application, and the predominated active constituents of TCM, including formula and single herbal medicine, needed to be clarified. Pharmacokinetics was a vital tool to describe the active compounds of TCM, and the in vivo distribution data of compounds were obtained from sacrificial animals. The biological samples were fragile, limited in volume, and non-renewable. The powerful analytical tech, especially mass spectrum, facilitated the determination and recognition of chemical constituents and their metabolites in vivo [140]. Many animals would be sacrificial under traditional sampling and determining strategies when researchers want to declare the combination mechanism of TCM by knock-in or knock-out any composition of medicinal formula.
Ligand fishing has been exploited in screening bioactive compounds, and the evaluation basis usually build on disease-associated biomacromolecules and cells [141, 142]. This promising strategy was hindered by the fragility of proteins and the high cost associated with the large volume of biological macromolecules. The consistency between in vivo and in vitro of proteins/cells has been concerned and discussed. With great biocompatibility and powerful integration potential, the micro-scale system, microfluidic chip, was found to be suitable for screening of active compounds (Table 1).
The design and application of microchannels reduced sample consumption and ensured the recovery of active macromolecule [108]. Three interconnected channels were employed in the competitive evaluation of drugs on double targets. According to the construe of chip, drug solution was directed to middle channels, while the solution containing target macromolecules was pumped to left or right channels. G-Quadruplex (HT24), dsDNA (DNA26) were used as efficacy and adverse reaction target, respectively. Based on the laminar flow, potential active components obtained from natural plant seed was screened one by one. As a result, sanguinarine and chelerythrine emerged as candidate drugs with stronger binding to HT24 than DNA26 [143]. The chip achieved the active compounds screening and toxicity evaluation simultaneously. Additionally, the Christmas tree structure integrated into the chip simplified the preparation of gradient concentration drugs. Drug candidates were mixed by laminar flow orderly and cells were treated with gradient concentration drug, thereby enhancing the throughput of active compound screening (Fig. 6B) [67, 144].
Different from homogenization evaluation, single-cell analysis contributes to precision medicine and targeted drug development [145]. In the case of micro dielectrophoresis devices, the mobility of bioparticles/particles closely correlates with the biophysical properties of the cells, facilitating the identification and trap** of special subpopulations from the whole vague population. After incubation of fluorescent zymosan bioparticles, RAW 264.7 cells were classified into different subpopulations within an EKMr range of 2.8 to 18.2 V/m2. Compared to the berberine treated group, RAW 264.7 cells with more engulfed fluorescent bioparticles tended to concentrate at an EKMr less than 8.2 V/m2. This result demonstrated that single cell phagocytic activity evaluation of TCM was accessible in microfluidic chips [146].
Compared to conventional single type cell culture or transwell cultures, multi-cell co-cultures or mimicking three-dimensional physiological structures can enhance cell-to-cell interactions, resulting in more comprehensive biological model structures and functions. Various methods have been developed to mimic the microenvironment of lesions, such as tumor spheroids, multi-cell co-culture, and organoids, to facilitate drug screening (Fig. 6B, C, F). For example, the human umbilical vein endothelial cells and cancer spheroids (40–100 μm in diameter) were placed into two adjacent channels on a chip. The collagen matrix allowed endothelial cell to form an intact endothelial monolayer and facilitated the diffusion of drug compound or cellular secretions. Twelve candidate constituents were added to the endothelial channel to resemble the drug diffusion across the capillary vessel in the circulatory system. After 36 h, the number of cell nuclei and spheroid dispersion were calculated. Nitidine and resveratrol significantly inhibited the dispersion of the spheroids, showing antimetastatic efficacy [147].
TCM formulas (e.g., kaixinsan, Yiqi Tongluo granule) have been clinically implemented as major or adjunctive compositions to treat brain disease [41]. The validation of permeability is as crucial as pharmacological assays, and microfluidics provide a conducive platform for mimicking this process. For instance, the BBB-U251 chip, a microfluidic chip that replicated the blood–brain barrier-glioma interface, was created by co-culturing human brain endothelial cells, pericytes, astrocytes, and U251 cells (Fig. 6D). This chip preserved the function of P-glycoprotein and exhibited permeability of various-sized FITC-dextran that was two to three orders of magnitude lower than that observed in Transwell systems [133]. Importantly, microfluidic chips not only simulate BBB in vitro but also provide a chamber for cell/organ culture. The co-existence of barrier tissues involved in drug metabolism and the ability to mimic lesions such as the tumor microenvironment significantly enhance the accuracy of preclinical evaluation for potential natural drug candidates [148].
Elucidation of TCM’s compatibility
Studying the molecular-level combination mechanism is crucial for modernizing TCM, but it also poses challenges to further advancements. Many advanced theories have been proposed and applied in this field [4, 39, 44]. However, the biological evaluation and validation of complex compounds from TCM remain challenging projects. For example, the synergistic effect between different substance group of total ginsenosides and total salvianolic acid were observed a decade ago, and it is a massive consumption of labor and materials to gain an in-depth understanding of the compatibility between two groups of compounds [149,150,6F) [106].
Toxicological efficacy could be identified and assessed using microfluidic chips. For instance, a three-phase laminar flow chip simulated the flow of micro-flow containing medical molecules and biomacromolecule. The efficacy and toxicity of four alkaloids that came from Macleaya cordata were assayed simultaneously by studying their binding affinity and mode [143]. Based on cell microfluidic chip-mass spectrometry system, HT22 cells were employed to investigate the neurotoxicity mechanism of aconitine. The dynamic monitoring platform demonstrated changes in metabolic profiling of amino acids and compounds related to energy metabolism. Following treatment with aconitine, HT22 cells were continuously observed for 24 h. Glutamic acid and aspartic acid were accumulated to induce neurotoxicity, and this process was followed by the energy metabolism disorder including the accumulation of lactic acid and reduction of glucose [159]. Furthermore, employing multiple channels and gases can achieved the manipulation of micro flow. This approach can generate multiple compartment cell particles and applied in the toxic evaluation of TCM. Adjacent parts allowed the interaction of different cells, offering a novel method to mimic microenvironment. For example, 3D co-culture particles (heart-on-a-particle) including HL-1 and HUVEC cells were utilized as heart models to investigate the toxic mechanism of aconitine on the heart [84].
Application framework of microfluidic chips for TCM
To outline a comprehensive scheme for the utilization of microfluidic chips in TCM studies, an application framework of microfluidic chips for TCM is proposed and illustrated in Fig. 7. The design and fabrication of chips must align with the scientific objectives, and the scheme highlights key considerations during this process. In chemical compounds detection of TCM, including extraction, separation and quantitative assay, the design of microchannels is paramount. For example, the shape and number of channels on a chip influence the laminar flow dynamics, and the performance of compounds separation chips was also limited to detailed structure of junctions. The multiple channels and its downstream functional devices allows for high throughput assays on a single chip, and customized channel configurations can be implemented. Notably, the integration of HPLC–UV, HPLC–MS or other advanced detection technologies significantly enhances the sensitivity and accuracy of miniaturized total analysis systems for TCM.
In the biological evaluation of TCM, the fragility of enzymes (diseases target proteins) should be considered. The material composition of chamber influences the activity and feasibility of proteins and cultured cells. PDMS is a typical biocompatible materials used in chip manufacturing. The specific chamber design depends on the requirement of the study; for instance, circle culture chamber meet the needs of single layer cell cultures. For multi-cell co-culture chips, porous membrane or multiple layer structure are necessary in chamber construction. The flow within channels facilitates cell metabolism and material exchange between the input flow and chamber solution, thereby facilitating the screening of active ingredients in TCM.
Continuous fluid flow enables real-time sampling, and chip tandem MS system has been reported and applied in the field of drug metabolism and metabolomics. Multiple layers of channels and chambers can be form on one chip. For example, the upper channel can be used to pump flow containing TCM constituents, while intestinal epithelial cells and vascular epithelial cells can be cultured in the middle layers to partially mimic drug absorption and metabolism processes in vivo. The flow in the bottom channel can be infused with TCM metabolites, which can then be used to treat target cells.
Microfluidic chip is a powerful manufacture tool. Single cell droplets can be formed using cross junction or specific trap chambers, allowing for the acquisition of biological information distinct from homogenized analysis. Multi-stream laminar flow is beneficial for cell/organ particles and is one method for constructing organs in vitro. Microenvironment simulation is an excellent feature of microfluidic chips, and the closer the imitation, the more detailed information about efficacy or toxicity of TCM can be obtained. Hence, if necessary, complex systems containing organoids and the co-culture of different organoids are also accessible.
In summary, the manipulation of microflow is achieved through customized micro devices. This continuous fluid serves as both an input and output mechanism on the chip. Biomacromolecules, cells, or organoids can be accommodated within biocompatible chambers. The active region serves as the information exchange center on the chip. The integration of different functional regions on chips enables the execution of various analysis protocols for modernizing TCM.