Abstract
Background
Living a happy and meaningful life is an eternal topic in positive psychology, which is crucial for individuals’ physical and mental health as well as social functioning. Well-being can be subdivided into pleasure attainment related hedonic well-being or emotional well-being, and self-actualization related eudaimonic well-being or psychological well-being plus social well-being. Previous studies have mostly focused on human brain morphological and functional mechanisms underlying different dimensions of well-being, but no study explored brain network mechanisms of well-being, especially in terms of topological properties of human brain morphological similarity network.
Methods
Therefore, in the study, we collected 65 datasets including magnetic resonance imaging (MRI) and well-being data, and constructed human brain morphological network based on morphological distribution similarity of cortical thickness to explore the correlations between topological properties including network efficiency and centrality and different dimensions of well-being.
Results
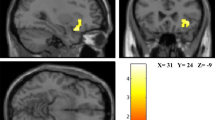
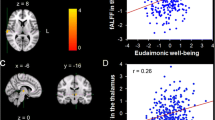
We found emotional well-being was negatively correlated with betweenness centrality in the visual network but positively correlated with eigenvector centrality in the precentral sulcus, while the total score of well-being was positively correlated with local efficiency in the posterior cingulate cortex of cortical thickness network.
Conclusions
Our findings demonstrated that different dimensions of well-being corresponded to different cortical hierarchies: hedonic well-being was involved in more preliminary cognitive processing stages including perceptual and attentional information processing, while hedonic and eudaimonic well-being might share common morphological similarity network mechanisms in the subsequent advanced cognitive processing stages.
Similar content being viewed by others
Background
Living a happy and meaningful life is a major topic in positive psychology, which have a great impact on individuals’ physical and mental health and help people flourish in their lives, in their communities, and in the world [1,2,3]. Researchers have proved that healthy people with higher levels of well-being tend to have better emotional states, better interpersonal relationships, and stronger senses of belonging to a group [2, 4, 5], thus they were less likely to suffer from mental illnesses [6]. Compared with other models of well-being mostly focusing on emotional (or subjective) aspect of well-being, Keyes [7, 8] developed the mental health continuum model composed of three well-being components: emotional (subjective) well-being, psychological well-being, and social well-being. Specifically, emotional well-being reflect the hedonic aspect of well-being that encompassed pleasure attainment, positive affective states, and high levels of life satisfaction [9]. Psychological well-being and social well-being together are considered as eudaimonic well-being, which refer to the actualization of individuals’ potential or true value and evaluation of one’s circumstance and functioning in society [10, 11]. Previous studies have shown that these three dimensions of well-being were moderately correlated with each other, and they were interrelated but distinct constructs [12,13,14].
Recently, neuroimaging studies have used different experimental approaches to enrich our understanding of both anatomical and functional substrates of different dimensions of well-being and showed a variety of association results [15]. For instance, a result from an electroencephalography study showed that the greater left than right superior frontal activation was associated with the higher levels of both hedonic and eudaimonic well-being [16]. MRI studies revealed many correlations between different dimensions of well-being and human brain structural metrics [e.g., the regional gray matter volume (rGMV) or regional gray matter density (rGMD)]. In more detail, social well-being was correlated with both rGMV in the left dorsolateral prefrontal cortex [17] and rGMD in the left orbitofrontal cortex [18], which were both involved in emotional regulation [19,20,21] and social-cognitive processes [22, 23]. Besides, several other studies reported the associations between emotional well-being and rGMV in the precuneus [24, 25], the rostral anterior cingulate [25, 26] and the left dorsolateral prefrontal cortex [26, 27]; as well as the correlation between psychological well-being and rGMV in the insula [27, 28]. Meanwhile, several resting-state fMRI studies also reported the links between (1) emotional well-being and human brain functional measurements [e.g., regional homogeneity (ReHo) and amplitude of low-frequency fluctuations (ALFF)] in the prefrontal cortex [28,29,http://volbrain.upv.es) [113]; (2) image intensity inhomogeneity correction; (3) tissue segmentation of cerebrospinal fluid (CSF), white matter (WM) and deep gray matter (GM); (4) generation of the GM-WM (white surface) and GM-CSF interface (pial surface); (5) spatial registration via matching of the cortical folding patterns across participants by recon-all in FreeSurfer and Gaussian spatial smoothing (FWHM = 6mm, Full Width at Half Maxima); (6) Finally, the 3D (dimensional) structure images were projected onto the fsaverage5 standard cortical surface with 10,242 vertices per hemisphere.
Quality control procedure
Quality control is very significant for solid data analysis. The CCS provided quality control procedures for both functional and structural images. For structural MRI in this study, the quality control procedure (QCP) was as follows: (1) brain extraction or skull strip**; (2) image tissue segmentation; (3) reconstruction of pial and white surface; and (4) head motion. We performed the visual inspection on all the original structural images and excluded participants with obvious structural brain abnormalities and significant motor artifacts during the scan. The CCS provides screenshots of the brain tissue segmentation as well as screenshots of pial and white surface reconstruction. We visually checked the screenshots, and participants with bad brain tissue segmentation and surface reconstruction were excluded from the subsequent analysis. All the participants passed the quality control. The final sample included 65 participants and their descriptive information and inter-variable correlations were shown in Table 1.
Morphological similarity network construction
In the study, we used a macro-scale brain network parcellation, which subdivided the entire cortical surface into 51 spatially connected parcels which were derived from a clustering approach on MRI images of 1000 subjects to identify networks of functionally coupled regions across the cerebral cortex [114], to construct human brain morphological network based on their distributions, and then we calculated mean cortical thickness of each parcel. We excluded the parcels whose vertex number was less than 50, and finally got 32 parcels reserved for final group analysis: expanding across all the Yeo-7 networks: visual network, somatomotor network, dorsal attention network, ventral attention network, limbic network, frontoparietal (control) network, and default mode network (see Table 2).
As in our previous study [47], we estimated distribution similarity of cortical thickness for each pair of parcels to construct human brain morphological similarity network. Firstly, for each pair of parcels, we segmented both of their cortical thickness into 30 bins. Secondly, we calculated the vertex frequency for each bin of the two parcels, and then we got the frequency distribution histogram for each parcel. Finally, we computed the Pearson’s correlation to estimate the similarity of cortical thickness distribution, and then we obtained a 32 × 32 morphological correlation matrix for each participant. There were both positive and negative connections between different brain regions which respectively demonstrated co-varying and anti-correlated distribution curves, and the negative connections only occupied a tiny proportion of the entire connection matrix. Therefore, in the study, we considered the absolute values of connections to computing network topological measurements considering the little effects of negative connections on the whole brain network topology. Then, we used orthogonal minimal spanning trees (OMST) analysis, which was a threshold-free method to derive the strongest connections of a network and reserve important information about brain network organization [115], to get an undirected weighted graph, and then the topological measurements could be computed based on the binary (unweighted) correlation matrix.
Topological measurements
We computed network efficiency including global efficiency (Eglob), nodal efficiency (Enodal) and local efficiency (Elocal) as well as network centrality including degree centrality (DC), betweenness centrality (BC), eigenvector centrality (EC) and pagerank centrality (PC) based on the binary (unweighted) correlation matrix using the Brain Connectivity Toolbox (http://www.brain-connectivity-toolbox.net) [48] and the CCS scripts [108].
Network efficiency
Global efficiency for network G is defined as:
where N is the number of nodes and Lij is the shortest path length between node i and node j in graph G [52]. Global efficiency is a global measurement of the parallel ability of information transfer within the whole network.
Nodal efficiency of node i is defined as:
where N and Lij are the same as that in Eq. (1), respectively representing the number of nodes and the shortest path length between node i and node j in graph G. Nodal efficiency measures the ability of the node for information transfer within the whole network and is also a global measurement.
Local efficiency of node i is defined as:
where Gi is a subgraph and is composed of the nodes that connect to node i (not including node i) directly and interconnected edges. Local efficiency indicates how well the information is exchanged in the given brain region and hence is a local measurement.
Network centrality
Degree centrality of node i is defined as:
where N is the set of all nodes in the network, and aij is the connection status between i and j: aij = 1 when i and j were connected and aij = 0 when i and j weren’t connected. DC identifies the nodes with the most connected links and is the most common quantifiable local centrality measure [48, 49, 116].
Betweenness centrality of node i is defined as:
where Lkj is the number of shortest paths between node k and node j, and Lkj(i) is the number of shortest paths between k and j that pass through node i. BC represents the fraction of all shortest paths in the network that pass through a given node. High BC indicated the nodes were important in connecting disparate parts of the network [48, 117] and were global measuremens.
Eigenvector centrality of node i is defined as:
where \({\mu }_{j}\left(i\right)\) is the i-th component of the j-th eigenvector of the adjacency matrix aij, and \({\lambda }_{1}\) is the corresponding j-th eigenvalue. N is the set of all nodes in the network, and aij is the connection matrix. EC considers the nodes connecting to other high degree nodes as highly central and indicates a central and important role of the node in the network [118, 119].
Pagerank centrality of node i is defined as:
Pagerank centrality was introduced originally by Google to rank web pages. In graph theory, PC represents the importance of nodes assuming that the importance of a node is the expected sum of the importance of all connected nodes and the direction of edges [120, 121]. The PC algorithm is a variant of EC, which introduces a small probability (1—d = 0.15, d is dam** factor) of random dam** to handle walking traps on a graph [122]. Both EC and PC are global centrality measurements.
Statistics
To investigate the associations between topological measurements (i.e., network efficiency Effi and centrality Cent) of human brain morphological similarity network and different dimensions of well-being, we applied general linear model that took age, sex, education, intracranial volume (ICV), mean cortical thickness (CT) as covariates. The detailed statistical model was shown in Eq. (8).
False discovery rate (FDR, q < 0.05) correction for 32 parcels was used to control type 1 error over multiple tests. And the General Linear Model statistical analysis and FDR correction were performed using MATLAB scripts in the study.
Availability of data and materials
References
Huta V, Waterman AS. Eudaimonia and its distinction from hedonia: develo** a classification and terminology for understanding conceptual and operational definitions. J Happiness Stud. 2013;15(6):1425–56. https://doi.org/10.1007/s10902-013-9485-0.
Lyubomirsky S, King L, Diener E. The benefits of frequent positive affect: Does happiness lead to success? Psychol Bull. 2005;131(6):803–55. https://doi.org/10.1037/0033-2909.131.6.803.
Ryan RM, Deci EL. On happiness and human potentials: a review of research on hedonic and eudaimonic well-being. Annu Rev Psychol. 2001;52(1):141–66. https://doi.org/10.1146/annurev.psych.52.1.141.
Diener E, Seligman MEP. Very happy people. Psychol Sci. 2002;13(1):81–4. https://doi.org/10.1111/1467-9280.00415.
Steptoe A, Wardle J, Marmot M. Positive affect and health-related neuroendocrine, cardiovascular, and inflammatory processes. Proc Natl Acad Sci USA. 2005;102(18):6508–12. https://doi.org/10.1073/pnas.0409174102.
Schutte NS, Malouff JM, Thorsteinsson EB, Bhullar N, Rooke SE. A meta-analytic investigation of the relationship between emotional intelligence and health. Pers Indiv Differ. 2007;42(6):921–33. https://doi.org/10.1016/j.paid.2006.09.003.
Keyes CLM. Promoting and protecting mental health as flourishing—a complementary strategy for improving national mental health. Am Psychol. 2007;62(2):95–108. https://doi.org/10.1037/0003-066x.62.2.95.
Keyes C. Brief description of the mental health continuum short form (MHC-SF). 2009. Available online at: https://www.aacu.org/sites/default/files/MHC-SFEnglish.pdf.
Diener E, Oishi S, Lucas RE. Personality, culture, and subjective well-being: emotional and cognitive evaluations of life. Annu Rev Psychol. 2003;54(1):403–25. https://doi.org/10.1146/annurev.psych.54.101601.145056.
Keyes C. Social well-being. Soc Psychol Q. 1998;61(2):121–40.
Ryff CD, Keyes CLM. The structure of psychological well-being revisited. J Pers Soc Psychol. 1995;69(4):719–27. https://doi.org/10.1037/0022-3514.57.6.1069.
Diener E. Assessing subjective well-being: progress and opportunities. Soc Indic Res. 1994;31(2):103–57. https://doi.org/10.1007/BF01207052.
Gallagher MW, Lopez SJ, Preacher KJ. The hierarchical structure of well-being. J Pers. 2009;77(4):1025–50. https://doi.org/10.1111/j.1467-6494.2009.00573.x.
Lamers SMA, Westerhof GJ, Bohlmeijer ET, ten Klooster PM, Keyes CLM. Evaluating the psychometric properties of the mental health Continuum-Short Form (MHC-SF). J Clin Psychol. 2011;67(1):99–110. https://doi.org/10.1002/jclp.20741.
de Vries LP, van de Weijer MP, Bartels M. A systematic review of the neural correlates of well-being reveals no consistent associations. Neurosci Biobehav Rev. 2023;145:105036. https://doi.org/10.1016/j.neubiorev.2023.105036.
Urry HL, Nitschke JB, Dolski I, Jackson DC, Dalton KM, Mueller CJ, et al. Making a life worth living: neural correlates of well-being. Psychol Sci. 2004;15(6):367–72. https://doi.org/10.1111/j.0956-7976.2004.00686.x.
Kong F, Hu S, Xue S, Song Y, Liu J. Extraversion mediates the relationship between structural variations in the dorsolateral prefrontal cortex and social well-being. NeuroImage (Orlando). 2015;105:269–75. https://doi.org/10.1016/j.neuroimage.2014.10.062.
Kong F, Yang K, Sajjad S, Yan W, Li X, Zhao J. Neural correlates of social well-being: gray matter density in the orbitofrontal cortex predicts social well-being in emerging adulthood. Soc Cogn Affect Neurosci. 2019;14(3):319–27. https://doi.org/10.1093/scan/nsz008.
Goldin PR, McRae K, Ramel W, Gross JJ. The neural bases of emotion regulation: reappraisal and suppression of negative emotion. Biol Psychiat (1969). 2008;63(6):577–86. https://doi.org/10.1016/j.biopsych.2007.05.031.
Golkar A, Lonsdorf TB, Olsson A, Lindstrom KM, Berrebi J, Fransson P, et al. Distinct contributions of the dorsolateral prefrontal and orbitofrontal cortex during emotion regulation. PLoS ONE. 2012;7(11):e48107. https://doi.org/10.1371/journal.pone.0048107.
Kanske P, Heissler J, Schönfelder S, Bongers A, Wessa M. How to regulate emotion? Neural networks for reappraisal and distraction. Cereb Cortex (New York, NY 1991). 2011;21(6):1379–88. https://doi.org/10.1093/cercor/bhq216.
Omar R, Henley SMD, Bartlett JW, Hailstone JC, Gordon E, Sauter DA, et al. The structural neuroanatomy of music emotion recognition: evidence from frontotemporal lobar degeneration. NeuroImage (Orlando). 2011;56(3):1814–21. https://doi.org/10.1016/j.neuroimage.2011.03.002.
Skuse DH, Gallagher L. Dopaminergic-neuropeptide interactions in the social brain. Trends Cogn Sci. 2008;13(1):27–35. https://doi.org/10.1016/j.tics.2008.09.007.
Sato W, Kochiyama T, Uono S, Kubota Y, Sawada R, Yoshimura S, et al. The structural neural substrate of subjective happiness. Sci Rep. 2015;5(1):16891. https://doi.org/10.1038/srep16891.
Matsunaga M, Kawamichi H, Koike T, Yoshihara K, Yoshida Y, Takahashi HK, et al. Structural and functional associations of the rostral anterior cingulate cortex with subjective happiness. NeuroImage (Orlando). 2016;134:132–41. https://doi.org/10.1016/j.neuroimage.2016.04.020.
Takeuchi H, Taki Y, Nouchi R, Hashizume H, Sassa Y, Sekiguchi A, et al. Anatomical correlates of quality of life: evidence from voxel-based morphometry. Hum Brain Mapp. 2014;35(5):1834–46. https://doi.org/10.1002/hbm.22294.
Lewis GJ, Kanai R, Rees G, Bates TC. Neural correlates of the ‘good life’: eudaimonic well-being is associated with insular cortex volume. Soc Cogn Affect Neurosci. 2014;9(5):615–8. https://doi.org/10.1093/scan/nst032.
Kong F, Hu S, Wang X, Song Y, Liu J. Neural correlates of the happy life: the amplitude of spontaneous low frequency fluctuations predicts subjective well-being. NeuroImage (Orlando). 2015;107:136–45. https://doi.org/10.1016/j.neuroimage.2014.11.033.
Kong F, Wang X, Song Y, Liu J. Brain regions involved in dispositional mindfulness during resting state and their relation with well-being. Soc Neurosci. 2016;11(4):331–43. https://doi.org/10.1080/17470919.2015.1092469.
Kong F, Ma X, You X, **ang Y. The resilient brain: psychological resilience mediates the effect of amplitude of low-frequency fluctuations in orbitofrontal cortex on subjective well-being in young healthy adults. Soc Cogn Affect Neurosci. 2018;13(7):755–63. https://doi.org/10.1093/SCAN/NSY045.
**ang G, Li Q, Du X, Liu X, Liu Y, Chen H. Knowing who you are: neural correlates of self-concept clarity and happiness. Neuroscience. 2022;490:264–74. https://doi.org/10.1016/j.neuroscience.2022.03.004.
Ren Z, Shi L, Wei D, Qiu J. Brain functional basis of subjective well-being during negative facial emotion processing task-based fMRI. Neuroscience. 2019;423:177–91. https://doi.org/10.1016/j.neuroscience.2019.10.017.
Luo Y, Huang X, Yang Z, Li B, Liu J, Wei D. Regional homogeneity of intrinsic brain activity in happy and unhappy individuals. PLoS ONE. 2014;9(1):e85181. https://doi.org/10.1371/journal.pone.0085181.
Kong F, Xue S, Wang X. Amplitude of low frequency fluctuations during resting state predicts social well-being. Biol Psychol. 2016;118:161–8. https://doi.org/10.1016/j.biopsycho.2016.05.012.
Won J, Nielson KA, Smith JC. Subjective well-being and bilateral anterior insula functional connectivity after exercise intervention in older adults with mild cognitive impairment. Front Neurosci. 2022;16:834816. https://doi.org/10.3389/fnins.2022.834816.
Barrett LF, Satpute AB. Large-scale brain networks in affective and social neuroscience: towards an integrative functional architecture of the brain. Curr Opin Neurobiol. 2013;23(3):361–72. https://doi.org/10.1016/j.conb.2012.12.012.
Cavanna AE, Trimble MR. The precuneus: a review of its functional anatomy and behavioural correlates. Brain (London, England: 1878). 2006;129(3):564–83. https://doi.org/10.1093/brain/awl004.
Gerlach KD, Spreng RN, Madore KP, Schacter DL. Future planning: default network activity couples with frontoparietal control network and reward-processing regions during process and outcome simulations. Soc Cogn Affect Neurosci. 2014;9(12):1942–51. https://doi.org/10.1093/scan/nsu001.
Spreng RN, Stevens WD, Chamberlain JP, Gilmore AW, Schacter DL. Default network activity, coupled with the frontoparietal control network, supports goal-directed cognition. NeuroImage (Orlando). 2010;53(1):303–17. https://doi.org/10.1016/j.neuroimage.2010.06.016.
Kringelbach ML, Berridge KC. Towards a functional neuroanatomy of pleasure and happiness. Trends Cogn Sci. 2009;13(11):479–87. https://doi.org/10.1016/j.tics.2009.08.006.
Luo Y, Kong F, Qi S, You X, Huang X. Resting-state functional connectivity of the default mode network associated with happiness. Soc Cogn Affect Neurosci. 2016;11(3):516–24. https://doi.org/10.1093/scan/nsv132.
Shi L, Sun J, Wu X, Wei D, Chen Q, Yang W, et al. Brain networks of happiness: dynamic functional connectivity among the default, cognitive and salience networks relates to subjective well-being. Soc Cogn Affect Neurosci. 2018;13(8):851–62. https://doi.org/10.1093/scan/nsy059.
Waytz A, Hershfield HE, Tamir DI. Mental simulation and meaning in life. J Pers Soc Psychol. 2015;108(2):336–55. https://doi.org/10.1037/a0038322.
Weathersby FL, King JB, Fox JC, Loret A, Anderson JS. Functional connectivity of emotional well-being: overconnectivity between default and attentional networks is associated with attitudes of anger and aggression. Psychiat Res-Neuroim. 2019;291:52–62. https://doi.org/10.1016/j.pscychresns.2019.08.001.
Bullmore E, Sporns O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009;10(3):186–98. https://doi.org/10.1038/nrn2575.
Sporns O. The human connectome: a complex network. Ann N Y Acad Sci. 2011;1224(1):109–25. https://doi.org/10.1111/j.1749-6632.2010.05888.x.
Li C, Qiao K, Mu Y, Jiang L. Large-scale morphological network efficiency of human brain: cognitive intelligence and emotional intelligence. Front Aging Neurosci. 2021;13:605158. https://doi.org/10.3389/fnagi.2021.605158.
Rubinov M, Sporns O. Complex network measures of brain connectivity: uses and interpretations. Neuroimage. 2010;52(3):1059–69. https://doi.org/10.1016/j.neuroimage.2009.10.003.
Zuo XN, Ehmke R, Mennes M, Imperati D, Castellanos FX, Sporns O, et al. Network centrality in the human functional connectome. Cereb Cortex. 2012;22(8):1862–75. https://doi.org/10.1093/cercor/bhr269.
Joyce KE, Laurienti PJ, Burdette JH, Hayasaka S. A new measure of centrality for brain networks. PLoS ONE. 2010;5(8):e12200. https://doi.org/10.1371/journal.pone.0012200.
Latora V, Marchiori M. Efficient behavior of small-world networks. Phys Rev Lett. 2001;87(19):198701/4-4. https://doi.org/10.1103/PhysRevLett.87.198701.
Latora V, Marchiori M. Economic small-world behavior in weighted networks. EPJ B Condens Matter Phys. 2003;32(2):249–63. https://doi.org/10.1140/epjb/e2003-00095-5.
Bullmore E, Sporns O. The economy of brain network organization. Nat Rev Neurosci. 2012;13(5):336–49. https://doi.org/10.1038/nrn3214.
Wheelock MD, Rangaprakash D, Harnett NG, Wood KH, Orem TR, Mrug S, et al. Psychosocial stress reactivity is associated with decreased whole-brain network efficiency and increased amygdala centrality. Behav Neurosci. 2018;132(6):561–72. https://doi.org/10.1037/bne0000276.
Diener E, Emmons RA, Larsen RJ, Griffin S. The satisfaction with life scale. J Pers Assess. 1985;49(1):71–5.
Tang G-Q, Huang M-E. Diverse consequences of negative emotional responses between high and low happiness people. Acta Psychol Sin. 2012;44(8):1086–99.
Lyubomirsky S, Boehm JK, Kasri F, Zehm K. The cognitive and hedonic costs of dwelling on achievement-related negative experiences: implications for enduring happiness and unhappiness. Emotion (Washington). 2011;11(5):1152–67. https://doi.org/10.1037/a0025479.
Lyubomirsky S. Why are some people happier than others? The role of cognitive and motivational processes in well-being. Am Psychol. 2001;56(3):239–49. https://doi.org/10.1037/0003-066X.56.3.239.
Tomasi D, Volkow ND. Association between functional connectivity hubs and brain networks. Cereb Cortex (New York, NY 1991). 2011;21(9):2003–13. https://doi.org/10.1093/cercor/bhq268.
Carlson JM, Reinke KS, Habib R. A left amygdala mediated network for rapid orienting to masked fearful faces. Neuropsychologia. 2009;47(5):1386–9. https://doi.org/10.1016/j.neuropsychologia.2009.01.026.
Lai C-H, Hsu Y-Y, Wu Y-T. First episode drug-naïve major depressive disorder with panic disorder: gray matter deficits in limbic and default network structures. Eur Neuropsychopharm. 2010;20(10):676–82. https://doi.org/10.1016/j.euroneuro.2010.06.002.
Keightley ML, Chiew KS, Winocur G, Grady CL. Age-related differences in brain activity underlying identification of emotional expressions in faces. Soc Cogn Affect Neurosci. 2007;2(4):292–302. https://doi.org/10.1093/scan/nsm024.
Kitada R, Johnsrude IS, Kochiyama T, Lederman SJ. Brain networks involved in haptic and visual identification of facial expressions of emotion: an fMRI study. NeuroImage (Orlando). 2010;49(2):1677–89. https://doi.org/10.1016/j.neuroimage.2009.09.014.
Schwarzkopf DS, Song C, Rees G. The surface area of human V1 predicts the subjective experience of object size. Nat Neurosci. 2011;14(1):28–30. https://doi.org/10.1038/nn.2706.
Zhang M, Ma C, Luo Y, Li J, Li Q, Liu Y, et al. Neural basis of uncertain cue processing in trait anxiety. Sci Rep-UK. 2016;6(1):21298. https://doi.org/10.1038/srep21298.
Yang X, Liu J, Meng Y, **a M, Cui Z, Wu X, et al. Network analysis reveals disrupted functional brain circuitry in drug-naive social anxiety disorder. NeuroImage (Orlando). 2019;190:213–23. https://doi.org/10.1016/j.neuroimage.2017.12.011.
Killgore WDS, Yurgelun-Todd DA. Social anxiety predicts amygdala activation in adolescents viewing fearful faces. NeuroReport. 2005;16(15):1671–5. https://doi.org/10.1097/01.wnr.0000180143.99267.bd.
Derrfuss J, Vogt VL, Fiebach CJ, von Cramon DY, Tittgemeyer M. Functional organization of the left inferior precentral sulcus: dissociating the inferior frontal eye field and the inferior frontal junction. NeuroImage (Orlando). 2012;59(4):3829–37. https://doi.org/10.1016/j.neuroimage.2011.11.051.
Berman RA, Colby CL, Genovese CR, Voyvodic JT, Luna B, Thulborn KR, et al. Cortical networks subserving pursuit and saccadic eye movements in humans: an FMRI study. Hum Brain Mapp. 1999;8(4):209–25. https://doi.org/10.1002/(SICI)1097-0193(1999)8:4%3c209::AID-HBM5%3e3.0.CO;2-0.
Luna B, Thulborn KR, Strojwas MH, McCurtain BJ, Berman RA, Genovese CR, et al. Dorsal cortical regions subserving visually guided saccades in humans: an fMRI study. Cereb Cortex (New York, NY 1991). 1998;8(1):40–7. https://doi.org/10.1093/cercor/8.1.40.
Petit L, Orssaud C, Tzourio N, Crivello F, Berthoz A, Mazoyer B, et al. Functional anatomy of a prelearned sequence of horizontal saccades in humans. J Neurosci. 1996;16(11):3714–26. https://doi.org/10.1523/jneurosci.16-11-03714.1996.
Fusser F, Linden DEJ, Rahm B, Hampel H, Haenschel C, Mayer JS. Common capacity-limited neural mechanisms of selective attention and spatial working memory encoding. Eur J Neurosci. 2011;34(5):827–38. https://doi.org/10.1111/j.1460-9568.2011.07794.x.
Hagler DJ, Sereno MI. Spatial maps in frontal and prefrontal cortex. NeuroImage (Orlando). 2006;29(2):567–77. https://doi.org/10.1016/j.neuroimage.2005.08.058.
Noyce AL, Cestero N, Michalka SW, Shinn-Cunningham BG, Somers DC. Sensory-biased and multiple-demand processing in human lateral frontal cortex. J Neurosci. 2017;37(36):8755–66. https://doi.org/10.1523/JNEUROSCI.0660-17.2017.
Jaun-Frutiger K, Cazzoli D, Müri RM, Bassetti CL, Nyffeler T. The frontal eye field is involved in visual vector inversion in humans—a theta burst stimulation study. PLoS ONE. 2013;8(12):83297. https://doi.org/10.1371/journal.pone.0083297.
Nagel M, Sprenger A, Lencer R, Kömpf D, Siebner H, Heide W. Distributed representations of the “preparatory set” in the frontal oculomotor system: a TMS study. BMC Neurosci. 2008;9(1):89. https://doi.org/10.1186/1471-2202-9-89.
Nyffeler T, Bucher O, Pflugshaupt T, Von Wartburg R, Wurtz P, Hess CW, et al. Single-pulse transcranial magnetic stimulation over the frontal eye field can facilitate and inhibit saccade triggering. Eur J Neurosci. 2004;20(8):2240–4. https://doi.org/10.1111/j.1460-9568.2004.03667.x.
Yang Q, Kapoula Z. Distinct control of initiation and metrics of memory-guided saccades and vergence by the FEF: a TMS study. PLoS ONE. 2011;6(5):e20322. https://doi.org/10.1371/journal.pone.0020322.
Grosbras M-H, Laird AR, Paus T. Cortical regions involved in eye movements, shifts of attention, and gaze perception. Hum Brain Mapp. 2005;25(1):140–54. https://doi.org/10.1002/hbm.20145.
Muggleton NG, Juan C-H, Cowey A, Walsh V. Human frontal eye fields and visual search. J Neurophysiol. 2003;89(6):3340–3. https://doi.org/10.1152/jn.01086.2002.
Quentin R, Chanes L, Migliaccio R, Valabrègue R, Valero-Cabré A. Fronto-tectal white matter connectivity mediates facilitatory effects of non-invasive neurostimulation on visual detection. NeuroImage (Orlando). 2013;82:344–54. https://doi.org/10.1016/j.neuroimage.2013.05.083.
Smith DT, Jackson SR, Rorden C. Transcranial magnetic stimulation of the left human frontal eye fields eliminates the cost of invalid endogenous cues. Neuropsychologia. 2005;43(9):1288–96. https://doi.org/10.1016/j.neuropsychologia.2004.12.003.
Andrews-Hanna JR, Reidler JS, Sepulcre J, Poulin R, Buckner RL. Functional-anatomic fractionation of the brain’s default network. Neuron (Cambridge). 2010;65(4):550–62. https://doi.org/10.1016/j.neuron.2010.02.005.
Andrews-Hanna JR, Smallwood J, Spreng RN. The default network and self-generated thought: component processes, dynamic control, and clinical relevance: the brain’s default network. Ann N Y Acad Sci. 2014;1316(1):29–52. https://doi.org/10.1111/nyas.12360.
Utevsky AV, Smith DV, Huettel SA. Precuneus is a functional core of the default-mode network. J Neurosci. 2014;34(3):932–40. https://doi.org/10.1523/JNEUROSCI.4227-13.2014.
Irish M, Halena S, Kamminga J, Tu S, Hornberger M, Hodges JR. Scene construction impairments in Alzheimer’s disease—a unique role for the posterior cingulate cortex. Cortex. 2015;73:10–23. https://doi.org/10.1016/j.cortex.2015.08.004.
Maddock RJ, Garrett AS, Buonocore MH. Posterior cingulate cortex activation by emotional words: fMRI evidence from a valence decision task. Hum Brain Mapp. 2003;18(1):30–41. https://doi.org/10.1002/hbm.10075.
Wagner AD, Shannon BJ, Kahn I, Buckner RL. Parietal lobe contributions to episodic memory retrieval. Trends Cogn Sci. 2005;9(9):445–53. https://doi.org/10.1016/j.tics.2005.07.001.
Abraham A, Schubotz RI, von Cramon DY. Thinking about the future versus the past in personal and non-personal contexts. Brain Res. 2008;1233:106–19. https://doi.org/10.1016/j.brainres.2008.07.084.
Maddock RJ, Garrett AS, Buonocore MH. Remembering familiar people: the posterior cingulate cortex and autobiographical memory retrieval. Neuroscience. 2001;104(3):667–76. https://doi.org/10.1016/S0306-4522(01)00108-7.
Northoff G, Bermpohl F. Cortical midline structures and the self. Trends Cogn Sci. 2004;8(3):102–7. https://doi.org/10.1016/j.tics.2004.01.004.
Kjaer TW, Nowak M, Lou HC. Reflective self-awareness and conscious states: PET evidence for a common midline parietofrontal core. NeuroImage (Orlando). 2002;17(2):1080–6. https://doi.org/10.1016/S1053-8119(02)91230-9.
Lou HC, Luber B, Crupain M, Keenan JP, Nowak M, Kjaer TW, et al. Parietal cortex and representation of the mental self. P Natl Acad Sci USA. 2004;101(17):6827–32. https://doi.org/10.1073/pnas.0400049101.
Kjaer TW, Nowak M, Kjaer KW, Lou AR, Lou HC. Precuneus-prefrontal activity during awareness of visual verbal stimuli. Conscious Cogn. 2001;10(3):356–65. https://doi.org/10.1006/ccog.2001.0509.
Dörfel D, Werner A, Schaefer M, von Kummer R, Karl A. Distinct brain networks in recognition memory share a defined region in the precuneus—a functional connectivity study. NeuroImage (Orlando). 2009;47:S53. https://doi.org/10.1016/S1053-8119(09)70164-8.
Wenderoth N, Debaere F, Sunaert S, Swinnen SP. The role of anterior cingulate cortex and precuneus in the coordination of motor behaviour. Eur J Neurosci. 2005;22(1):235–46. https://doi.org/10.1111/j.1460-9568.2005.04176.x.
Dickerson BC, Sperling RA. Large-scale functional brain network abnormalities in Alzheimer’s disease: insights from functional neuroimaging. Behav Neurol. 2009;21(1–2):63–75. https://doi.org/10.1155/2009/610392.
Miners JS, Palmer JC, Love S. Pathophysiology of hypoperfusion of the precuneus in early Alzheimer’s disease: hypoperfusion of precuneus in Alzheimer’s disease. Brain Pathol (Zurich). 2016;26(4):533–41. https://doi.org/10.1111/bpa.12331.
Ryu S-Y, Kwon MJ, Lee S-B, Yang DW, Kim T-W, Song I-U, et al. Measurement of precuneal and hippocampal volumes using magnetic resonance volumetry in Alzheimer’s disease. J Clin Neurol (Seoul). 2010;6(4):196–203. https://doi.org/10.3988/jcn.2010.6.4.196.
Rosas HD, Salat DH, Lee SY, Zaleta AK, Pappu V, Fischl B, et al. Cerebral cortex and the clinical expression of Huntington’s disease: complexity and heterogeneity. Brain (London, England: 1878). 2008;131(4):1057–68. https://doi.org/10.1093/brain/awn025.
Bailly M, Destrieux C, Hommet C, Mondon K, Cottier J-P, Beaufils E, et al. Precuneus and cingulate cortex atrophy and hypometabolism in patients with Alzheimer’s disease and mild cognitive impairment: MRI and 18F-FDG PET quantitative analysis using freesurfer. Biomed Res Int. 2015. https://doi.org/10.1155/2015/583931.
Dai W, Lopez OL, Carmichael OT, Becker JT, Kuller LH, Gach HM. Mild cognitive impairment and alzheimer disease: patterns of altered cerebral blood flow at MR imaging. Radiology. 2009;250(3):856–66. https://doi.org/10.1148/radiol.2503080751.
Matsuda H. The role of neuroimaging in mild cognitive impairment. Neuropathology. 2007;27(6):570–7. https://doi.org/10.1111/j.1440-1789.2007.00794.x.
Baumeister RF, Vohs KD, Aaker JL, Garbinsky EN. Some key differences between a happy life and a meaningful life. J Posit Psychol. 2013;8(6):505–16. https://doi.org/10.1080/17439760.2013.830764.
Huta V. Linking peoples’ Pursuit of eudaimonia and hedonia with characteristics of their parents: parenting styles, verbally endorsed values, and role modeling. J Happiness Stud. 2011;13(1):47–61. https://doi.org/10.1007/s10902-011-9249-7.
Keyes CLM. Mental illness and/or mental health? Investigating axioms of the complete state model of health. J Consult Clin Psychol. 2005;73(3):539–48. https://doi.org/10.1037/0022-006X.73.3.539.
Yin K, He J. Reliability and validity of the mental health continuum short form in adults. Chin Mental Health J. 2012;26(5):388–92.
Xu T, Yang Z, Jiang L, **ng X-X, Zuo X-N. A connectome computation system for discovery science of brain. Sci Bull. 2015;60(1):86–95. https://doi.org/10.1007/s11434-014-0698-3.
Cox RW. AFNI: what a long strange trip it’s been. Neuroimage. 2012;62(2):743–7. https://doi.org/10.1016/j.neuroimage.2011.08.056.
Jenkinson M, Beckmann CF, Behrens TE, Woolrich MW, Smith SM. FSL. Neuroimage. 2012;62(2):782–90. https://doi.org/10.1016/j.neuroimage.2011.09.015.
Fischl B. FreeSurfer. Neuroimage. 2012;62(2):774–81. https://doi.org/10.1016/j.neuroimage.2012.01.021.
Zuo X-N, Xu T, Jiang L, Yang Z, Cao X-Y, He Y, et al. Toward reliable characterization of functional homogeneity in the human brain: preprocessing, scan duration, imaging resolution and computational space. NeuroImage (Orlando). 2013;65:374–86. https://doi.org/10.1016/j.neuroimage.2012.10.017.
Manjon JV, Coupe P. Volbrain: an online MRI brain volumetry system. Front Neuroinform. 2016;10:30. https://doi.org/10.3389/fninf.2016.00030.
Yeo BT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106(3):1125–65. https://doi.org/10.1152/jn.00338.2011.
Dimitriadis SI, Salis C, Tarnanas I, Linden DE. Topological filtering of dynamic functional brain networks unfolds informative chronnectomics: a novel data-driven thresholding scheme based on orthogonal minimal spanning trees (OMSTs). Front Neuroinform. 2017;11:28. https://doi.org/10.3389/fninf.2017.00028.
Sabidussi G. The centrality index of a graph. Psychometrika. 1966;31(4):581–603. https://doi.org/10.1007/BF02289527.
Freeman LC. Centrality in social networks conceptual clarification. Soc Netw. 1979;1(3):215–39. https://doi.org/10.1016/0378-8733(78)90021-7.
van Duinkerken E, Schoonheim MMI, Jzerman RG, Moll AC, Landeira-Fernandez J, Klein M, et al. Altered eigenvector centrality is related to local resting-state network functional connectivity in patients with longstanding type 1 diabetes mellitus. Hum Brain Mapp. 2017;38(7):3623–36. https://doi.org/10.1002/hbm.23617.
Bonacich P. Factoring and weighting approaches to status scores and clique identification. J Math Sociol. 1972;2(1):113–20. https://doi.org/10.1080/0022250X.1972.9989806.
Gleich DF. PageRank beyond the web. SIAM Rev. 2015;57(3):321–63. https://doi.org/10.1137/140976649.
Henni K, Mezghani N, Gouin-Vallerand C. Unsupervised graph-based feature selection via subspace and pagerank centrality. Expert Syst Appl. 2018;114:46–53. https://doi.org/10.1016/j.eswa.2018.07.029.
Boldi P, Santini M, Vigna S. PageRank: functional dependencies. ACM Trans Inform Syst. 2009. https://doi.org/10.1145/1629096.1629097.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 11674388).
Author information
Authors and Affiliations
Contributions
Conceptualization and Funding acquisition: LJ; Methodology, Software and Visualization: YL, CL and LJ; Writing, review and editing: YL, LJ and CL.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The institutional review board of the Institute of Psychology Chinese Academy of Sciences approved this study, and written informed consent was obtained from individual participants prior to data acquisition.
Consent to publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Li, Y., Li, C. & Jiang, L. Well-being is associated with cortical thickness network topology of human brain. Behav Brain Funct 19, 16 (2023). https://doi.org/10.1186/s12993-023-00219-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12993-023-00219-6