Abstract
Background
Acute myeloid leukemia (AML) is a highly heterogeneous hematologic malignancy and the most frequently acute leukemia of stem cell precursors and the myeloid derivatives in adult. Longitudinal studies have indicated the therapeutic landscape and drug resistance for patients with AML are still intractable, which largely attribute to the deficiency of detailed information upon the pathogenesis.
Methods
In this study, we compared the cellular phenotype of resident NK cells (rAML-NKs, rHD-NKs) and expanded NK cells (eAML-NKs, eHD-NKs) from bone marrow of AML patients (AML) and healthy donors (HD). Then, we took advantage of the co-culture strategy for the evaluation of the in vitro cytotoxicity of NK cells upon diverse tumor cell lines (e.g., K562, Nalm6, U937). With the aid of RNA-sequencing (RNA-SEQ) and bioinformatics analyses (e.g., GOBP analysis, KEGG analysis, GSEA, volcano plot), we verified the similarities and differences of the omics features between eAML-NKs and eHD-NKs.
Results
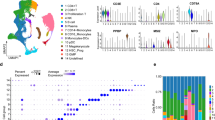
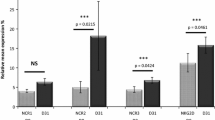
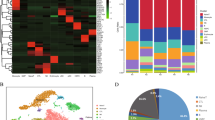
Herein, we verified the sharp decline in the content of total resident NK cells (CD3−CD56+) in rAML-NKs compared to rHD-NKs. Differ from the expanded eHD-NKs, eAML-NKs revealed decline in diverse NK cell subsets (NKG2D+, CD25+, NKp44+, NKp46+) and alterations in cellular vitality but conservations in cytotoxicity. According to transcriptomic analysis, AML-NKs and HD-NKs showed multifaceted distinctions in gene expression profiling and genetic variations.
Conclusions
Collectively, our data revealed the variations in the cytobiological and transcriptomic features between AML-NKs and HD-NKs in bone marrow environment. Our findings would benefit the further development of novel biomarkers for AML diagnosis and NK cell-based cytotherapy in future.
Similar content being viewed by others
Background
Acute myeloid leukemia (AML) has been recognized as a heterogeneous hematological malignancy and the most common acute leukemia in adults, which is mainly caused by multifaceted somatic mutations in myeloid differentiation and monoclonal proliferation of immature progenitors [1, 2]. Despite the approval and application of novel drugs for patients, AML still remains as a major field of unmet medical need among diverse hematologic malignancies attributes to the deficiency of details information upon the cytopathologic and pathogenic features [3]. For decades, considerable progresses have been achieved in elucidating the pathogenesis of AML both at the cellular and molecular levels [4]. For instance, diverse inherited genetic loci and novel biomarkers (e.g., epigenetic, genetic, and protein) involved in myelodysplastic syndrome (MDS) and AML development in patients have been identified for the risk stratification and treatment assessment of AML patients on the basis of epigenetic and omics profiles [3, 5]. According to the World Health Organization (WHO) Classification of leukemia and myeloid neoplasms, AML can be divided into eight categories according to the recurrent genetic abnormalities and into three categories according to the indicated gene mutations. However, the genes with sufficient accuracy for unraveling the risk-stratification schemes and clinically targetable treatment decision-making of AML are still largely obscure [6].
Natural killer (NK) cells are critical innate lymphoid cells (ILCs) for their effect in tumor immunosurveillance and antiviral immunity dispense with presensitization, which have been involved in both innate and adoptive immune responses via the cytotoxic and cytokine-secreting approaches [7,8,9]. NK cells have been considered as heterogeneous populations generated from hematopoietic stem cells (HSCs) in the bone marrow environment (BME) with limited functional and phenotypic diversity, which are composed of two distinct subsets in human, including the cytotoxic CD3−CD56dimCD16high and the IFN-γ-producing CD3−CD56brightCD16+ counterparts [10, 11]. To date, adoptive NK cells and the concomitant chimeric antigen receptor-transduced NK cells (CAR-NKs) have been explored in various cancer settings including AML, and aiming to attain better therapeutic outcomes [12, 13]. For example, Terrén et al. and Albinger et al. reported the application of cytokine-induced memory-like (CIML) NK cells and primary CD33-targeting CAR-NK cells for the treatment of patients with AML, respectively [14, 15]. Furthermore, Soldierer et al. and Xu et al. highlighted the genetic engineering of human cells for the CAR-enhanced immunotherapy of hematological malignancies including AML [16, 17]. Very recently, Gauthier and the colleagues reported the successful control of AML by a trifunctional NKp46−CD16a−NK cell engager (NKCE) with prolonged pharmacodynamic effects and very low inflammatory cytokine induction against CD123 antibody-dependent cell cytotoxicity (ADCC) [18]. Of note, Crinier et al. showed the trajectories of NK cell differentiation from the resident CD56bright NK cells to the CD56dim NK1-like NK cells and the CD56bright NK2-like NK cells, yet the major cytophenotypic and omics features of resident and expanded NK cells in the bone marrow of AML patients (AML-NKs) are largely obscure.
For the purpose, in this study, we isolated resident NK cells (rNKs) from both AML patients (rAML-AMLs) and healthy donors (rHD-AMLs), and conducted ex vivo NK cell expansion and activation from the corresponding rNKs after a 14-day’s induction (eAML-AMLs, eHD-AMLs). With the aid of multifaceted cellular and molecular analyses, we verified the cytophernotic (e.g., cytomorphology, immunophenoty**, cellular vitality, and cytotoxicity against diverse tumor cell lines) and transcriptomic characteristics (e.g., gene expression profiling, genetic variations) between AML-NKs and HD-NKs in the bone marrow microenvironment, which collectively indicated the pathogenic effect of NK cells in patients with AML and would benefit the further development of novel biomarkers for clinical diagnosis and NK cell-based cytotherapy in future.
Methods
Preparation of mononuclear cells (MNCs) from bone marrow
Human bone marrow was obtained with the consent of AML patients and healthy donors (HD) and the approval of the Ethics Committee of Gansu Provincial Hospital and the guideline of Helsinki Declaration (2023-120). For MNCs isolation from bone marrow of AML patients (AML-MNCs) and HD (HD-MNCs), the Ficoll-based density gradient centrifugation were conducted as we described recently with several modifications [40]. The further exploration of the alterations in AML-NK cells would benefit our understanding upon pathogenesis and therapeutic regimens of AML from the aspect of numerical, receptor expression, and maturation defects of NK cells, along with checkpoint overexpression inhibitors and epigenetic modifications [40]. Of note, it’s of great interesting to further explore the potential variations of NK cells in bone marrow environment of AML patients before treatment and remission, which will help illuminate the biofunction and omics features of AML and benefit the development of targeted therapy in clinical practice.
State-of-the-art literatures have highlighted the involvement of dysimmunity in the pathogenesis and drug resistance of AML patients. For instance, Zhang et al. verified the multiple chemoresistant properties in LSCs and the oxidative phosphorylation (OXPHOS) signatures in pediatric AML [41]. Very recently, Naldini et al. reported the longitudinal single-cell profiling and functional validation of chemotherapy response in AML patients, and identified the variations in the stemness and quiescence signatures of the OxPhoslow miR-126high LSCs [42]. Of note, Abbas et al. outlined the T cell receptor repertoire profiling and the T cell landscape in AML patients with PD-1 blockade therapy [43]. Instead, we conducted systematic and detailed characterization of the multifaceted signatures of NK cells and the variations in subsets and gene expression pattern between resident and expanded AML-NKs and HD-NKs. To our knowledge, there were minimal reports upon the specific subsets (e.g., CD3−CD56+CD25+) of AML-NKs in AML patients. Similarly, Stringaris and the colleagues verified the upregulation of NKG2A (a inhibitory receptor) and the downregulation of NKp46 in NK cells of AML patients, which indicated the long-lasting changes and effector function impairment in AML-NK cells induced by AML blasts [44]. Interestingly, Crinier and the colleagues revealed the trajectories and the stress signature of NK cell differentiation in bone marrow, which further indicated the involvement of AML-NKs in the pathogenesis of AML [45]. Therefore, it is of great interesting to further dissect the single-cell mutation and gene expression profiling of clonal evolution as well as cellular hierarchy in AML patients, which will collectively benefit the development of accurate diagnosis and targeted therapy [46, 47].
Conclusion
Overall, in this study, we systematically and meticulously dissected the biological phenotypes, cellular vitality and cytotoxicity of both resident and expanded NK cells between AML-NKs and HD-NKs from the landscape of gene expression pattern and somatic variations. Our findings put forward the potential pathogenic role of AML-NK cells in bone marrow microenvironment (BME) in AML patients, which could be conducive to the further exploration of the pathogenesis of AML and the development of NK cell-based targeted therapy in future.
Availability of data and materials
All data of this study are included in the published article. Meanwhile, the datasets analyzed during this study are available from corresponding author upon reasonable request.
Abbreviations
- BM-NKs:
-
Natural killer cells from bone marrow
- MNCs:
-
Mononuclear cells
- GvHD:
-
Graft-versus-host disease
- HSC:
-
Hematopoietic stem cell
- PBS:
-
Phosphate buffer solution
- PCA:
-
Principal component analysis
- GSEA:
-
Gene set enrichment analysis
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- EMT:
-
Epithelial-mesenchymal transition
- NKCE:
-
NK cell engager
- ADCC:
-
Antibody-dependent cytotoxicity
- BME:
-
Bone marrow microenvironment
- VSEs:
-
Variable shear events
- DEGs:
-
Differentially expressed genes
- OXPHOS:
-
Oxidative phosphorylation
- BM-MSCs:
-
Bone marrow-derived mesenchymal stem/stromal cells
- t-AML:
-
Therapy-related AML
- LSCs:
-
Leukemic stem cells
- HSPCs:
-
Haematopoietic stem/progenitor cells
- ILCs:
-
Innate lymphoid cells
References
Saleh K, Khalifeh-Saleh N, Kourie HR. Acute myeloid leukemia transformed to a targetable disease. Future Oncol. 2020;16(14):961–72.
De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: a comprehensive review and 2016 update. Blood Cancer J. 2016;6(7): e441.
Dohner H, Estey EH, Amadori S, Appelbaum FR, Buchner T, Burnett AK, Dombret H, Fenaux P, Grimwade D, Larson RA, et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood. 2010;115(3):453–74.
Prada-Arismendy J, Arroyave JC, Rothlisberger S. Molecular biomarkers in acute myeloid leukemia. Blood Rev. 2017;31(1):63–76.
Li Y, Xu Q, Lv N, Wang L, Zhao H, Wang X, Guo J, Chen C, Li Y, Yu L. Clinical implications of genome-wide DNA methylation studies in acute myeloid leukemia. J Hematol Oncol. 2017;10(1):41.
Kayser S, Levis MJ. The clinical impact of the molecular landscape of acute myeloid leukemia. Haematologica. 2023;108(2):308–20.
Zhang C, Hu Y, Shi C. Targeting natural killer cells for tumor immunotherapy. Front Immunol. 2020;11:60.
Zhang L, Liu M, Yang S, Wang J, Feng X, Han Z. Natural killer cells: of-the-shelf cytotherapy for cancer immunosurveillance. Am J Cancer Res. 2021;11(4):1770–91.
Wu SY, Fu T, Jiang YZ, Shao ZM. Natural killer cells in cancer biology and therapy. Mol Cancer. 2020;19(1):120.
Crinier A, Narni-Mancinelli E, Ugolini S, Vivier E. SnapShot: natural killer cells. Cell. 2020;180(6):1280–1280.
Bjorkstrom NK, Ljunggren HG, Michaelsson J. Emerging insights into natural killer cells in human peripheral tissues. Nat Rev Immunol. 2016;16(5):310–20.
Rahmani S, Yazdanpanah N, Rezaei N. Natural killer cells and acute myeloid leukemia: promises and challenges. Cancer Immunol Immunother. 2022;71(12):2849–67.
Zhang L, Meng Y, Yao H, Zhan R, Chen S, Miao W, Ma S, Xu X, Li Y, Yu M, et al. CAR-NK cells for acute myeloid leukemia immunotherapy: past, present and future. Am J Cancer Res. 2023;13(11):5559–76.
Terren I, Orrantia A, Astarloa-Pando G, Amarilla-Irusta A, Zenarruzabeitia O, Borrego F. Cytokine-induced memory-like NK cells: from the basics to clinical applications. Front Immunol. 2022;13: 884648.
Albinger N, Pfeifer R, Nitsche M, Mertlitz S, Campe J, Stein K, Kreyenberg H, Schubert R, Quadflieg M, Schneider D, et al. Primary CD33-targeting CAR-NK cells for the treatment of acute myeloid leukemia. Blood Cancer J. 2022;12(4):61.
Soldierer M, Bister A, Haist C, Thivakaran A, Cengiz SC, Sendker S, Bartels N, Thomitzek A, Smorra D, Hejazi M, et al. Genetic of engineering and enrichment human NK cells for CAR-enhanced immunotherapy of hematological malignancies. Front Immunol. 2022;13: 847008.
Xu J, Niu T. Natural killer cell-based immunotherapy for acute myeloid leukemia. J Hematol Oncol. 2020;13(1):167.
Gauthier L, Virone-Oddos A, Beninga J, Rossi B, Nicolazzi C, Amara C, Blanchard-Alvarez A, Gourdin N, Courta J, Basset A, et al. Control of acute myeloid leukemia by a trifunctional NKp46-CD16a-NK cell engager targeting CD123. Nat Biotechnol. 2023;41(9):1296–306.
**e B, Zhang L, Gao J, Wang T, Liu M, Feng X, Xu X, Ma S, Cai H, Guo T, et al. Decoding the biological properties and transcriptomic landscapes of human natural killer cells derived from bone marrow and umbilical cord blood. Am J Cancer Res. 2023;13(5):2087–103.
Liu M, Meng Y, Zhang L, Han Z, Feng X. High-efficient generation of natural killer cells from peripheral blood with preferable cell vitality and enhanced cytotoxicity by combination of IL-2, IL-15 and IL-18. Biochem Biophys Res Commun. 2021;534:149–56.
Zhang L, Yang S, Chen H, Xue C, Wang T, Chen S, Xu X, Ma S, Yu M, Guo T, et al. Characterization of the biological and transcriptomic signatures of natural killer cells derived from cord blood and peripheral blood. Am J Cancer Res. 2023;13(8):3531–46.
Zhang L, Chi Y, Wei Y, Zhang W, Wang F, Zhang L, Zou L, Song B, Zhao X, Han Z. Bone marrow-derived mesenchymal stem/stromal cells in patients with acute myeloid leukemia reveal transcriptome alterations and deficiency in cellular vitality. Stem Cell Res Ther. 2021;12(1):365.
Huo J, Zhang L, Ren X, Li C, Li X, Dong P, Zheng X, Huang J, Shao Y, Ge M, et al. Multifaceted characterization of the signatures and efficacy of mesenchymal stem/stromal cells in acquired aplastic anemia. Stem Cell Res Ther. 2020;11(1):59.
Zhang L, Liu M, Song B, Miao W, Zhan R, Yang S, Han Z, Cai H, Xu X, Zhao Y, et al. Decoding the multidimensional signatures of resident and expanded natural killer cells generated from perinatal blood. Am J Cancer Res. 2022;12(5):2132–45.
Zhang L, Zou L, Ma Y, Feng C, Zhan R, Yang H, Song B, Han Z. Multifaceted modifications for a cell size-based circulating tumor cell scope technique hold the prospect for large-scale application in general populations. Cell Biol Int. 2021;45(2):345–57.
Wei Y, Zhang L, Chi Y, Ren X, Gao Y, Song B, Li C, Han Z, Zhang L, Han Z. High-efficient generation of VCAM-1(+) mesenchymal stem cells with multidimensional superiorities in signatures and efficacy on aplastic anaemia mice. Cell Prolif. 2020;53(8): e12862.
Zhang L, Wei Y, Chi Y, Liu D, Yang S, Han Z, Li Z. Two-step generation of mesenchymal stem/stromal cells from human pluripotent stem cells with reinforced efficacy upon osteoarthritis rabbits by HA hydrogel. Cell Biosci. 2021;11(1):6.
Zhang L, Wang H, Liu C, Wu Q, Su P, Wu D, Guo J, Zhou W, Xu Y, Shi L, et al. MSX2 initiates and accelerates mesenchymal stem/stromal cell specification of hPSCs by regulating TWIST1 and PRAME. Stem Cell Rep. 2018;11(2):497–513.
Gao H, Liu M, Zhang Y, Zhang L, **e B. Multifaceted characterization of the biological and transcriptomic signatures of natural killer cells derived from cord blood and placental blood. Cancer Cell Int. 2022;22(1):291.
Shiba N. Comprehensive molecular understanding of pediatric acute myeloid leukemia. Int J Hematol. 2023;117(2):173–81.
Ochs MA, Marini BL, Perissinotti AJ, Foucar CE, Pettit K, Burke P, Bixby DL, Benitez LL. Oncology stewardship in acute myeloid leukemia. Ann Hematol. 2022;101(8):1627–44.
Gruszka AM, Valli D, Alcalay M. Wnt signalling in acute myeloid leukaemia. Cells. 2019;8(11):1403.
Nilsson C, Linde F, Hulegardh E, Garelius H, Lazarevic V, Antunovic P, Cammenga J, Deneberg S, Eriksson A, Jadersten M, et al. Characterization of therapy-related acute myeloid leukemia: increasing incidence and prognostic implications. Haematologica. 2023;108(4):1015–25.
Wang D, Sun Z, Zhu X, Zheng X, Zhou Y, Lu Y, Yan P, Wang H, Liu H, ** J, et al. GARP-mediated active TGF-beta1 induces bone marrow NK cell dysfunction in AML patients with early relapse post-allo-HSCT. Blood. 2022;140(26):2788–804.
Dong H, Ham JD, Hu G, **e G, Vergara J, Liang Y, Ali A, Tarannum M, Donner H, Baginska J, et al. Memory-like NK cells armed with a neoepitope-specific CAR exhibit potent activity against NPM1 mutated acute myeloid leukemia. Proc Natl Acad Sci U S A. 2022;119(25): e2122379119.
Haroun-Izquierdo A, Vincenti M, Netskar H, van Ooijen H, Zhang B, Bendzick L, Kanaya M, Momayyezi P, Li S, Wiiger MT, et al. Adaptive single-KIR(+)NKG2C(+) NK cells expanded from select superdonors show potent missing-self reactivity and efficiently control HLA-mismatched acute myeloid leukemia. J Immunother Cancer. 2022;10(11): e005577.
Parihar R. Memory NK cells to forget relapsed AML. Blood. 2022;139(11):1607–8.
Romee R, Rosario M, Berrien-Elliott MM, Wagner JA, Jewell BA, Schappe T, Leong JW, Abdel-Latif S, Schneider SE, Willey S, et al. Cytokine-induced memory-like natural killer cells exhibit enhanced responses against myeloid leukemia. Sci Transl Med. 2016;8(357): 357ra123.
Paczulla AM, Rothfelder K, Raffel S, Konantz M, Steinbacher J, Wang H, Tandler C, Mbarga M, Schaefer T, Falcone M, et al. Absence of NKG2D ligands defines leukaemia stem cells and mediates their immune evasion. Nature. 2019;572(7768):254–9.
D’Silva SZ, Singh M, Pinto AS. NK cell defects: implication in acute myeloid leukemia. Front Immunol. 2023;14:1112059.
Zhang Y, Jiang S, He F, Tian Y, Hu H, Gao L, Zhang L, Chen A, Hu Y, Fan L, et al. Single-cell transcriptomics reveals multiple chemoresistant properties in leukemic stem and progenitor cells in pediatric AML. Genome Biol. 2023;24(1):199.
Naldini MM, Casirati G, Barcella M, Rancoita PMV, Cosentino A, Caserta C, Pavesi F, Zonari E, Desantis G, Gilioli D, et al. Longitudinal single-cell profiling of chemotherapy response in acute myeloid leukemia. Nat Commun. 2023;14(1):1285.
Abbas HA, Hao D, Tomczak K, Barrodia P, Im JS, Reville PK, Alaniz Z, Wang W, Wang R, Wang F, et al. Single cell T cell landscape and T cell receptor repertoire profiling of AML in context of PD-1 blockade therapy. Nat Commun. 2021;12(1):6071.
Stringaris K, Sekine T, Khoder A, Alsuliman A, Razzaghi B, Sargeant R, Pavlu J, Brisley G, de Lavallade H, Sarvaria A, et al. Leukemia-induced phenotypic and functional defects in natural killer cells predict failure to achieve remission in acute myeloid leukemia. Haematologica. 2014;99(5):836–47.
Crinier A, Dumas PY, Escaliere B, Piperoglou C, Gil L, Villacreces A, Vely F, Ivanovic Z, Milpied P, Narni-Mancinelli E, et al. Single-cell profiling reveals the trajectories of natural killer cell differentiation in bone marrow and a stress signature induced by acute myeloid leukemia. Cell Mol Immunol. 2021;18(5):1290–304.
Wu J, **ao Y, Sun J, Sun H, Chen H, Zhu Y, Fu H, Yu C, Weigao E, Lai S, et al. A single-cell survey of cellular hierarchy in acute myeloid leukemia. J Hematol Oncol. 2020;13(1):128.
Miles LA, Bowman RL, Merlinsky TR, Csete IS, Ooi AT, Durruthy-Durruthy R, Bowman M, Famulare C, Patel MA, Mendez P, et al. Single-cell mutation analysis of clonal evolution in myeloid malignancies. Nature. 2020;587(7834):477–82.
Acknowledgements
The co-authors would like to thank the members in The Fourth People’s Hospital of **an (The Teaching Hospital of Shandong First Medical University), The Second Hospital of Shandong University, Peking University Cancer Hospital Yunnan & Yunnan Cancer Hospital & The Third Affiliated Hospital of Kunming Medical University, Chinese Academy of Medical Sciences and Peking Union Medical College, The First Affiliated Hospital of Soochow University, and Gansu Provincial Hospital for their assistance. We also thank Professor Lugui Qiu in Chinese Academy of Medical Sciences and Peking Union Medical College for scientific suggestions.
Funding
The work was supported by the project Youth Fund supported by the National Natural Science Foundation of China (82260031), SDU-KI Collaborative Research Project of Qilu Medical College, Shandong University (SDU-KI-2019-15), Natural Science Foundation of Shandong Province (ZR2023MH341), Fujian Provincial Ministerial Finance Special Project (2021XH018), Postdoctoral Program of Natural Science Foundation of Gansu Province (23JRRA1319), Joint Major Project of Science and Technology Innovation in Fujian Province (2021Y9083), Natural Science Foundation of Fujian Province (2022J01266), Natural Science Foundation of Jiangxi Province (20224BAB206077), Science and Technology Research and Development Project of Langfang City (2023013147), Natural Science Foundation of Gansu Province (21JR7RA594), Gansu Provincial Hospital Intra-Hospital Research Fund Project (22GSSYB-6), The 2022 Master/Doctor/Postdoctoral program of NHC Key Laboratory of Diagnosis and Therapy of Gastrointestinal Tumor (NHCDP2022004, NHCDP2022008), project funded by China Postdoctoral Science Foundation (2023M730723), project funded by Lugui Qiu Yunnan Expert Workstation (202105AF150051).
Author information
Authors and Affiliations
Contributions
Leisheng Zhang, Yunyan Sun, and Chun-e Xue: performed the experiments, collection and assembly of data, manuscript writing; Yunyan Sun, Chengyun Zheng, Shuling Wang, and **anghong Xu: helped with collection or assembly of data; Leisheng Zhang, Cunrong Chen and Dexiao Kong: conception and design, data analysis and interpretation, manuscript writing, final approval of manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study was conducted with the approval of the Ethics Committee of Gansu Provincial Hospital and the guideline of Helsinki Declaration (2023-120), and informed consent were obtained from the participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1
: Table S1. The detail data of AML. Table S2. The cytokines used in this study. Table S3. Antibodies for flow cytometry assay in the study.
Additional file 2: Figure S1.
Representative FCS raw data files for rHD-NKs and rAML-NKs.
Additional file 3: Figure S2.
Antibody matching tables and representative FCS Raw Data files for cell apoptosis analysis of eHD-NKs and eAML-NKs.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, L., Sun, Y., Xue, Ce. et al. Uncovering the cellular and omics characteristics of natural killer cells in the bone marrow microenvironment of patients with acute myeloid leukemia. Cancer Cell Int 24, 106 (2024). https://doi.org/10.1186/s12935-024-03300-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12935-024-03300-w