Abstract
Background
Schistosomiasis remains an important public health problem throughout tropical and subtropical countries. Humans are infected through contact with water contaminated with schistosome cercariae. Therefore, issuing early warnings on the risk of infection is an important preventive measure against schistosomiasis. Sentinel mice are used to monitor water body infestations, and identifying appropriate antibody responses to schistosome antigens for early detection of infection would help to improve the efficiency of this system. In this study we explored the potential of detecting antibodies to the hydrophilic domain (HD) of the 23-kDa membrane protein (Sj23HD) and soluble egg antigen (SEA) of Schistosome japonicum for early detection of schistosome infection in sentinel mice.
Results
Development of IgM and IgG antibody levels against Sj23HD and SEA in S. japonicum infected mice was evaluated over the course of 42 days post-infection by enzyme-linked immunosorbent assay (ELISA) and immunoblotting. The Sj23HD and SEA specific IgM and IgG levels in mice all increased gradually over the course of infection, but IgM and IgG antibodies against Sj23HD presented earlier than those against SEA. Furthermore, the rates of positive antibody responses against Sj23HD were higher than those against SEA in the early stage of schistosome infection, suggesting that the likelihood of detecting early infection using anti-Sj23HD responses would be higher than that with anti-SEA responses. The use of immunoblotting could further improve the early detection of schistosome infection due to its greater sensitivity and specificity compared to ELISA. Additionally, the levels of Sj23HD and SEA specific antibodies positively correlated with the load of cercariae challenge and the duration of schistosome infection.
Conclusions
This study demonstrated that antibody responses to the Sj23HD antigen could be monitored for early detection of schistosome infection in mice, especially by immunoblotting which demonstrated greater sensitivity and specificity than ELISA for detection Sj23HD antibodies.
Similar content being viewed by others
Background
Schistosomiasis is an important tropical parasitic disease, with more than 200 million people currently infected among the 779 million people at risk of infection worldwide [1]. In the People's Republic of China (P.R. China), schistosome infection mainly occurs in the marshland and lake regions of Hunan, Hubei, Jiangxi, Anhui and Jiangsu provinces and in the hilly and mountainous regions of Sichuan and Yunnan provinces where the interruption of schistosomiasis transmission has been proven particularly difficult to achieve [17]. Sj23 plays an important role in maintaining growth and development of S. japonicum and is of interest as a potential vaccine candidate [18]. Therefore, detecting antibody responses to the Sj23 protein may be promising for early diagnosis of schistosome infection.
To develop a method for early detection of S. japonicum infection in sentinel mice, the dynamics of specific IgM and IgG antibodies responses to the hydrophilic domain (HD) of the Sj23 membrane protein (Sj23HD) and soluble egg antigen (SEA) in mice over the course of 42 days post-infection were systematically investigated in this study. These antibody levels were correlated with the load of cercariae used for infection and with the infection period. The efficiencies of ELISA and immunoblotting methods for detecting antibodies against Sj23HD and SEA were also compared.
Methods
Snails and cercariae
Snails (Oncomelania hupensis) infected with S. japonicum (typical schistosome species found in China) were provided by the Department of Snail Biology, Jiangsu Institute of Parasitic Diseases, Wuxi, P. R. China. S. japonicum cercariae were induced to hatch from infected snails by immersion in de-chlorinated water with illumination at 25°C for 2.5 h.
Animals
ICR mice (female, 20 g, 6 weeks old) were purchased from the Experimental Animal Center of Yangzhou University, Yangzhou, P.R. China. Japanese white rabbits were provided by the Experimental Animal Facility of Nan**g General Hospital of Nan**g Military Command, Nan**g, P.R. China, and raised at the Department of Experimental Animal, Jiangsu Institute of Parasitic Diseases, Wuxi, P.R. China. The experiments were carried out with approval from the Animal Research Advisory Committee of the Jiangsu Institute of Parasitic Diseases.
Reagents and instruments
Glutathione Sepharose 4B was purchased from GE Healthcare (Piscataway, NJ, USA). Ninety-six-well flat-bottomed ELISA plates were purchased from Greiner Bio-One Company (Frickenhausen, Germany). Bovine serum albumin (BSA) was obtained from BioDev-Tech. Co., Ltd. (Bei**g, P.R. China). Horseradish peroxidase (HRP)-labeled goat anti-mouse IgM antibody and IgG antibody were purchased from Bethyl Laboratories (Montgomery, TX, USA). 3,3',5,5'-tetramethylbenzidine (TMB) substrate was purchased from Genescript Biotech Company (Piscataway, NJ, USA). Nitrocellulose membranes (0.22 μm) were purchased from Whatman (Piscataway, NJ, USA). Diaminobenzidine (DAB) was purchased from Bio Basic Inc. (Ontario, Canada). Skim milk was purchased from Becton, Dickinson and Company (Franklin Lakes, NJ, USA). The ELISA plate reader Anthos Zenyth 340 was manufactured by Biochrom Co., Ltd. (Cambridge, UK) and the Trans-Blot SD Semi-Dry Electrophoretic Transfer Cell by Bio-Rad (Hercules, CA, USA).
Animal infection and serum collection
Experiment I
ICR mice were divided into a control group (n = 5) which received no treatment and an experimental group (n = 10) in which each animal was infected with 50 cercariae of S. japonicum by abdominal skin exposure [
Discussion
The current emphasis of schistosomiasis management in P.R. China is to survey areas where the transmission has been stopped and to control schistosomiasis epidemics in the areas where the prevalence of this parasitic infection has not been controlled effectively. Finding the susceptible, high risk environments for schistosome infection, issuing warnings of infection risk rapidly and taking more targeted measures for prevention and intervention to eliminate the incidence of infection are key goals for effective schistosomiasis control [22]. Therefore, develo** an early diagnostic technique to meet the requirements of the current schistosomiasis prevention and control program is paramount [23, 24].
The rapid development of modern genetic engineering techniques [25] makes it possible to isolate and produce specific antigens for detection of schistosome infections, and recombinant antigens can be used to improve the sensitivity and specificity of diagnostic methods. The recombinant Sj23 membrane protein has been shown to induce strong humoral immune responses and thus can be used as a diagnostic antigen to detect schistosome specific antibody responses [26, 27]. Lu et al. used the recombinant large HD of Sj23 to diagnose S. japonicum infection in buffalo [28]. Yu et al. had used the recombinant fusion protein of GST and large HD of Sj23 (GST-HD) to diagnose S. japonicum infection in humans [21]. The Sj23 membrane protein is present in all stages of the schistosome life cycle [17]; therefore, Sj23 specific antibodies should theoretically appear in the early stage of schistosome infection and would be valuable for early clinical diagnosis, as well as for determining the infection status of sentinel mice.
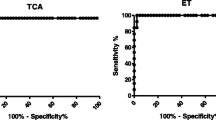
In this study, the utilities of Sj23 and SEA, a widely used diagnostic antigen, were compared for early detection of schistosome infection in sentinel mice. The recombinant fusion protein GST-Sj23HD and SEA were used to detect specific serum IgM and IgG antibodies in S. japonicum infected mice at different times post-infection by ELISA and immunoblotting. The results showed that both anti-SEA and anti-Sj23HD IgM and IgG could be found at days 7 to 10 post-infection, and the antibody titers increased gradually over the course of infection. However, the titers of Sj23HD antibodies increased more quickly than those of the SEA specific antibodies. The positive rates of Sj23HD antibodies at different times post-infection were higher than those of the antibodies against SEA. At day 21 post-infection, the positive rates of Sj23HD specific IgG and IgM reached 80 and 90%, respectively, but the positive rates of both SEA specific IgG and IgM only reached 70%. These results suggested that the antibody responses against Sj23HD would be more valuable for early diagnosis of schistosome infection than those against SEA, which may be attributed to the fact that Sj23 is a dominant schistosomula antigen. We speculate that there are epitopes within SEA which can induce specific antibodies in the early stage of infection in mice; however, since these are non-dominant antigens in schistosomula, they are unable to induce high titer antibodies in the early stage of schistosome infection and result in only weak immuno-reactivity and a low positive rate of detection.
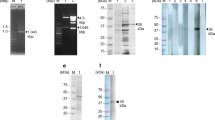
The specificity of an antibody detection method depends on the nature of the antigen used to capture the antibody. If the non-specific epitopes in the antigen are dominant, it would result in non-specific reactivity. In this study, the recombinant antigen Sj23HD was prepared from E. coli, and SEA was prepared from infected rabbit liver tissues, which could be contaminated with non-specific proteins. Thus, these pooled antigens used to detect antibodies by ELISA may result in cross-reactive or false positive responses. However, the immunoblotting assay can separate various components of the pooled antigens, and the specificity of a reaction can be evaluated based upon whether the serum antibodies recognize the protein band(s) of expected size. When using the purified GST-HD fusion protein, only the expected 33.5 kDa protein recognized by sera of mice post-infection could be considered as a positive response. For SEA, five main bands of 55, 73, 78, 84 and 121 kDa recognized by sera of mice post-infection could be judged as positive, especially the 73 and 78 kDa protein bands, which are present in nearly every stage of schistosome infection. Thus, the ability of the immunoblotting method to separate the antigens by size can help to avoid or reduce false positives due to non-specific protein reactions. For these reasons, the specificity of immunoblotting was determined to be higher than that of ELISA for detection of schistosome specific antibodies.
The results of this study also showed that the sensitivity of immunoblotting to detection of specific IgM and IgG was higher than that of ELISA, especially for detecting serum antibodies of mice in the early stage of infection. The lower detection efficiency of ELISA may be ascribed to the low titers of specific antibodies against schistosome antigens in the early stage of infection as well as the low antigen amounts coated on the wells of the microtiter plates. However, the amount of antigen transferred to the nitrocellulose membrane could be adjusted in accordance with the requirement of the immunoblotting conditions. For example, appropriately increasing the amount of antigen could be helpful in detecting low-titer schistosome specific serum antibodies. Therefore, the more efficient immunoblotting method would be more suitable than ELISA for early detection of schistosome infections.
Antibody levels are generally positively correlated with the amount and duration of antigen stimulation [29]. Indeed, the anti-Sj23 antibody levels in serum of schistosomiasis patients have been positively associated with the schistosome adult burden and infection duration [30–33]. Similarly, the results of this study also showed that the positive rates of anti-Sj23HD antibodies in mouse sera positively correlated with the load of S. japonicum cercariae used for the challenge and the duration of infection.
Conclusions
This study demonstrated that the antibody responses to the 23 kDa membrane protein of S. japonicum are useful for early detection of schistosome infection in mice. Compared to the ELISA based method, immunoblotting using the recombinant Sj23HD antigen has improved sensitivity and specificity for detecting specific IgM or IgG antibodies.