Abstract
Background
The ATP-binding cassette (ABC) transporter superfamily is the largest transporter gene family responsible for transporting specific molecules across lipid membranes in all living organisms. In insects, ABC transporters not only have important functions in molecule transport, but also play roles in insecticide resistance, metabolism and development.
Results
From the genome of the silkworm, Bombyx mori, we have identified 51 putative ABC genes which are classified into eight subfamilies (A-H) by phylogenetic analysis. Gene duplication is very evident in the ABCC and ABCG subfamilies, whereas gene numbers and structures are well conserved in the ABCD, ABCE, ABCF, and ABCH subfamilies. Microarray analysis revealed that expression of 32 silkworm ABC genes can be detected in at least one tissue during different developmental stages, and the expression patterns of some of them were confirmed by quantitative real-time PCR. A large number of ABC genes were highly expressed in the testis compared to other tissues. One of the ABCG genes, BmABC002712, was exclusively and abundantly expressed in the Malpighian tubule implying that BmABC002712 plays a tissue-specific role. At least 5 ABCG genes, including BmABC005226, BmABC005203, BmABC005202, BmABC010555, and BmABC010557, were preferentially expressed in the midgut, showing similar developmental expression profiles to those of 20-hydroxyecdysone (20E)-response genes. 20E treatment induced the expression of these ABCG genes in the midgut and RNA interference-mediated knockdown of USP, a component of the 20E receptor, decreased their expression, indicating that these midgut-specific ABCG genes are 20E-responsive.
Conclusion
In this study, a genome-wide analysis of the silkworm ABC transporters has been conducted. A comparison of ABC transporters from 5 insect species provides an overview of this vital gene superfamily in insects. Moreover, tissue- and stage-specific expression data of the silkworm ABCG genes lay a foundation for future analysis of their physiological function and hormonal regulation.
Similar content being viewed by others
Background
The ATP-binding cassette (ABC) transporters form one of the largest family of membrane proteins [1]. With 48 members in total, the ABC transporter family from animals was first identified in the human genome [2]. Ranging from 28 to ~200 members, the ABC transporter family is present in all organisms [3]. Based on sequence similarity of the ATP-binding sites, the 48 human ABC transporters can be classified into seven subfamilies (A to G, ABCA to ABCG). The eighth subfamily (H) was defined following the analysis of the genome of the fruitfly, Drosophila melanogaster (Diptera) [2]. In addition to the fruitfly [2], the ABC transporters have been previously analyzed in another insect species, the mosquito, Anopheles gambiae (Diptera), at the genome-wide level [4].
The ABC transporters share highly conserved domains known as nucleotide binding domains (NBDs). Each NBD contains three characteristic motifs, including Walker A box, Walker B box, and ABC signature C which links the two Walker boxes [5]. NBD binds and hydrolyses ATP and provides energy to transport molecules against concentration gradients. In addition to NBD, a eukaryotic ABC transporter usually consists of one or two transmembrane domains (TMD). The ABC transporters which have two NBDs and two TMDs are called full transporters, whereas those with one NBD and one TMD are called half transporters, which often constitute a functional unit by forming a homo- or heterodimer [6].
According to their functions, the ABC proteins can be classified as importers, exporters and non-transport proteins [7]. Importers and exporters are responsible for transport of a wide variety of substances, whereas the third class of ABC proteins are apparently not involved in molecule transport but in cellular processes such as DNA repair, translation or regulation of gene expression [8]. In human, known functions of ABC transporters include cholesterol and lipid transport, multidrug resistance, antigen presentation, mitochondrial iron homeostasis, and the ATP-dependent regulation of ion channels. Mutations in ABC genes have been associated with a range of disorders, including cystic fibrosis, hypercholesterolemia and diabetes [9]. In insects, it has been shown that ABC transporters have roles in uric acid metabolism, development and possibly in insecticide resistance [10]. Due to the importance of ABC transporters, several members of ABC transporters have been extensively studied in several model insects, including the fruitfly and the silkworm, Bombyx mori (Lepidoptera). One of the best studied insect ABC transporters is White, which is a typical ABCG transporter involved in pigment transport in insect eyes [11, 12].
In a microarray study published previously [13, 14], we detected multiple silkworm ABC genes exhibiting possible regulation by the molting hormone, 20-hydroxyecdysone (20E). To this end, we decided to identify and characterize the silkworm ABC transporters at the genome-wide level. Very recently, three silkworm ABC transporter subfamilies, including ABCB, ABCC and ABCG, were analyzed regarding to their possible xenobiotic resistance [10]. In this study, we have identified 51 putative silkworm ABC genes in total and analyzed their phylogenetic relationships. Moreover, we have investigated their temporal and spatial expression patterns using microarray and quantitative real-time PCR (qRT-PCR), with focus on the ABCG subfamily.
Results and Discussion
Identification of the silkworm ABC transporters
The NBDs of all the D. melanogaster ABC transporters were used as queries for BLASTP search against the two silkworm genome databases, SilkDB and KAIKObase. Each potential silkworm ABC transporter was validated by searching its known orthologs from the protein database of NCBI and further searching its NBD and TMD with the Pfam program. As a result, a total of 51 putative ABC genes were identified in the silkworm genome. A previous study has identified 56 ABC genes in D. melanogaster[2]. For comparative analysis, we also identified 68 ABC genes from the flour beetle, Tribolium castaneum (Coleoptera), 52 from A. gambiae (8 more compared to 44 in [4] due to the updated mosquito genome), and 43 from the honeybee, Apis mellifera (Hymenoptera) (Table 1).
In SilkDB, 37 ABC genes have evidences of mRNA expression with EST sequences, which were collected from 36 cDNA libraries of multiple tissues during different developmental stages. Based on the EST information, BmABC010129 shows the highest transcript level with 26 hits. The largest ABC transporter (1794 amino acids) and the smallest one (280 amino acids) are encoded by BmABC012789 and BmABC010825, respectively (Table 2).
Phylogenetic analysis of the silkworm ABC transporters
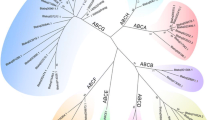
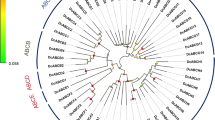
Each silkworm ABC transporter possesses one or two conserved NBDs (Figure 1). We performed a multiple sequence-alignment to construct a phylogenetic tree on the basis of NDBs of the 51 silkworm ABC transporters. Phylogenetic analysis revealed that silkworm ABC transporters can be divided into 8 subfamilies (A-H) with high bootstrap values. The ABCC subfamily can be separated into 2 groups, one of which shares high similarity to the ABCB subfamily (Figure 2). Among the 51 silkworm ABC transporters, there are 11 full transporters and 26 half transporters. The ABCA, ABCB and ABCC subfamilies contain full transporters, whereas half transporters exist in 6 subfamilies rather than ABCE and ABCF (Figure 1). The 4 ABCE and ABCF proteins lack of TMDs. One putative ABCB transporter contains one NDB and two TMDs, while one putative ABCC transporter contains two NDBs and one TMD. In addition, we found that 8 silkworm genes encode obvious NDBs show high similarity to ABC genes in the silkworm and other organisms. For convenience, we referred the 10 putative ABC proteins as incomplete ABC proteins. The incomplete sequences of those ABC genes might be because the genome of silkworm is not at completion and annotation of the entire genome is not complete. For comparative analysis, we also constructed phylogenetic trees for the ABC transporters from T. castaneum (Additional File 1: Fig. S1), A. gambiae (Additional File 2: Fig. S2), and A. mellifera (Additional File 3: Fig. S3). Similarly, the ABC transporters in each insect species can be divided into 8 subfamilies (A-H) (Table 1).
Phylogenetic tree of the silkworm ABC transporters. The phylogenetic tree was constructed using a neighbor-joining technique to analyze the amino acid sequences of the nucleotide binding domain (NBD). Analysis was performed with the program package MEGA4.0. The number at the branch point of the node represents the value resulting from 1000 replications and gaps were deleted with pairwise deletion method.
Genomic distribution of the silkworm ABC transporters
Of the 51 silkworm ABC genes, 50 are dispersed on 17 chromosomes and 1 on an unmapped scaffold. Gene structures of the 51 silkworm ABC transporters show high complexity with exon numbers ranging from 6 to 34 (Table 2). There are two major gene clusters located on chromosomes 12 and 15, which contain 9 and 10 ABC genes, respectively. In addition, chromosomes 10 and 22 each contain 4 ABC genes. Gene duplication is very evident in chromosomes 12 and 15, also occurs in chromosomes 10, 17 and 26 by tandem duplication (Additional File 4: Fig. S4).
Characterization of the silkworm ABC transporter subfamilies
Since the insect ABC transporters can be classified into 8 subfamilies (A-H), we characterized each subfamily in the silkworm and four other insect species for comparison.
ABCA
The ABCA transporters in mammals perform critical functions in the control of cellular lipid-transport processes [15], yet little is known about their physiological functions in insects. Among the 6 silkworm ABCA genes, BmABC007217, BmABC007218 and BmABC007221 are closely located on chromosome 17. The silkworm ABCA subfamily consists of 2 full transporters, 1 half transporter, and 3 incomplete ABC proteins containing one single NBD in each member. This subfamily includes two of the largest ABC transporters encoded by BmABC012789 (1794 amino acids) and BmABC007221 (1747 amino acids). In human, all ABCA transporters are full transporters [2]. In the plant, Arabidopsis thaliana, 11 are half transporters and only one is a full transporter [16]. No ABCA transporters have been identified in the yeast, Saccharomyces cerevisiae[17]. Interestingly, all the 9 ABCA transporters in the mosquito, A. gambiae, are full transporters, whereas both full and half transporters exist in the ABCA subfamily of the four other insect species, indicating that the ABCA subfamily varies greatly during evolution in insects and other organisms. In addition, phylogenetic analysis reveals that 3 insect ABCA transporters, BmABC012789, AGAP010416 and TcABCA2, are closely related (bootstrap value of 99%), implying that they are orthologs (Additonal File 5: Fig.S5).
ABCB
ABCB1 was the first human ABC transporter to be cloned and characterized through its function to confer a multidrug resistance (MDR) phenotype to cancer, and later studies revealed several more MDRs in the ABCB subfamily [18]. Based on sequence similarity, the insect ABCB transporters are thought to be involved in resistance to insecticides and other chemicals [10]. Among the 8 silkworm ABCB genes, BmABC000725 and BmABC000724 are closely distributed on chromosome 1 suggesting they were arisen by tandem duplication. The hypothesis of gene duplication is supported by phylogenetic analysis of the ABCB subfamily, which indicates that the two genes are closely related in evolution (Additonal File 6: Fig. S6). The silkworm ABCB subfamily consists of 4 full transporters, 3 half transporters, and 1 incomplete ABC protein. The same as other organisms, the ABCB subfamily in all the 5 insect species consists of both full and half transporters. The D. melanogaster ABCB gene, Mdr49, has multiple functions. Disruption of Mdr49 results in change of sensitivity to colchicines [19]. Mdr49 is not only involved in the transport of polycyclic aromatic hydrocarbons [20], but also controls germ cell attractant [21]. The ABCB transporter encoded by BmABC009452 is ~49% identical to that of Mdr49 in sequence (Additonal File 6: Fig. S6), but whether BmABC009452 and other silkworm ABCB genes are involved in xenobiotic resistances requires further investigation [10].
ABCC
The mammalian ABCC family members, including several multidrug resistance-associated proteins (MRP), are involved in ion transport, toxin secretion, signal transduction, and other physiological functions [5]. It has been suggested that the insect ABCC transporters might be important for xenobiotic resistance [10]. With 15 members in total, ABCC is the largest ABC subfamily in the silkworm. Moreover, there are 31 putative ABCC transporters in T. castaneum. Interestingly, 8 of the 15 silkworm ABCC genes are located on chromosome 15 and 4 on chromosome 12. Phylogenetic analysis of the ABCC subfamily within the 5 insect species reveals that many ABCC genes belonging to the same insect species cluster together, indicating gene duplication is an active mechanism that generates high diversity in the ABCC subfamily in insects (Additional File 7: Fig. S7). The silkworm ABCC subfamily consists of 5 full transporters, 7 half transporters, and 3 incomplete ABC proteins. All the 14 ABCC transporters in D. melanogaster are full transporters, whereas both full and half transporters exist in the ABCC subfamily of the four other insect species, supporting the evolutionary divergence of the ABCC subfamily in insects. The ABCC transporter encoded by CG10505 in D. melanogaster is regulated by heavy metals via the metal-responsive transcription factor 1 and involved in biochemical detoxification of zinc and copper [22]. Although several silkworm ABCC transporters have sequence similarity to the protein encoded by CG10505 in D. melanogaster (Additional File 7: Fig. S7), whether they might act as biochemical factors in the defense against toxins is unknown.
ABCD
The ABCD transporters are located to the peroxisome at the subcellular level and involved in the import of fatty acids and/or fatty acyl-CoAs into this organelle [23]. Each of the 5 insect species consists of 2 half transporters in the ABCD subfamily. The insect ABCD transporters are separated into two distinct groups by phylogenetic analysis (Additional File 8: Fig. S8), indicating that they are evolutionarily conserved in insects.
ABCE and ABCF
The ABCE and ABCF proteins lack TMD and each member contains a pair of linked NBDs [24]. Most eukaryotes have only one ABCE gene that is highly conserved during evolution (Additional File 9: Fig. S9). Different from most ABC transporters, their functions in cell biological processes are not related to transport. In human, ABCE1 was identified as an inhibitor of RNase L [25], whereas in yeast, ABCE proteins play key roles in translation initiation [26]. The silkworm ABCE gene, BmABC010129 which shows the highest expression level based on the EST information, is preferentially expressed in testis, ovary and fat body. However, RNA interference (RNAi)-mediated knockdown of BmABC010129 has no obvious effects on RNase L expression [27].
ABCF proteins function in ribosome biogenesis, translational control, and mRNA export, and are not involved in molecule transport [28]. The best-characterized ABCF protein is the yeast GCN20 protein, which is involved in the initiation and control of translation. In addition, mutations in GCN20 gene reduce eIF2α phosphorylation and thus translation in the ribosome [29]. Later studies revealed that ABCF1 physically interacts with eIF2 and associates with the ribosome in an ATP-dependent manner [28]. Until now, the ABCF subfamily has not been studied in insects. The same as human and most of other organisms, each of the 5 insect species consists of 3 ABCF proteins, which are separated into 3 distinct groups by phylogenetic analysis and genes in each group have high similarity (Additional File 9: Fig. S9). These data suggest that ABCE and ABCF subfamilies are highly conserved during evolution in all living organisms.
ABCG
The reported ABCG transporters are half transporters. Importantly, each ABCG transporter has a TMD at the C-terminal region of NBD, showing a distinct structure [1]. In human, members of the ABCG subfamily play key roles in lipid transport across membranes. For example, ABCG2 is an essential MDR and its activity is associated with decreased efficacy of anticancer agents in several carcinomas. Apart from its role in cancer, ABCG2 has a broad substrate specificity and its ability to transport numerous diverse pharmaceuticals has implications for the absorption, distribution, metabolism, excretion, and toxicity profile of these compounds. In addition, ABCG2 plays a role in the normal physiological transport of urate and haem [30]. The silkworm ABCG subfamily has 13 members, and 11 of them encode half transporters. Gene duplication is evident in this subfamily by the localization of gene paralogues or orthologs on chromosomes 5, 10, 12 and 22, which is further supported by phylogenetic analysis (Additional File 10: Fig. S10). The best-studied ABCG genes are those encoding eye pigment precursor transporters, white, scarlet, and brown, in D. melanogaster[11]. BmABC002922 (Bmwh3) encodes an ortholog of D. melanogaster white. Polyadenylated Bmwh3 transcript of about 2.7 kb length is detected in eggs, Malpighian tubule and pupal heads, but not in testes, posterior silk glands or fat body cells [31]. Bmwh3 is responsible for the transportation of ommochrome precursors and uric acid into pigment granules and urate granules, respectively, and Bmwh3 mutations cause white eyes, white eggs, and translucent larval skin [32]. BmABC002924 encodes an ortholog of D. melanogaster scarlet. In the silkworm, Scarlet forms a heterodimer with White to transport ommochrome precursors [33]. BmABC002581 encodes an ortholog of D. melanogaster brown. In addition, phylogenetic analysis revealed that BmABC010557 is orthologous to D. melanogaster E23 (CG3327) (Additional File 10: Fig. S10), which is a 20E primary response gene that represses 20E-mediated gene activation [12]. The potential physiological function of the silkworm ABCG subfamily is of interest.
ABCH
The ABCH subfamily was first identified in D. melanogaster and exists solely in arthropods [2]. The structure organizations of the ABCH and ABCG transporters are similar. Each of the 5 insect genomes studied in this report has 3 ABCH genes. Phylogenetic analysis suggests that the ABCH genes are originated from a common ancestor (Additional File 11: Fig. S11), yet their physiological functions remain unknown.
Tissue distribution analysis reveals some tissue-specific ABC genes
In order to understand the possible physiological function and hormonal regulation of the silkworm ABC genes in the future, we here analyzed the expression patterns of all the silkworm ABC genes by microarray data. With the exception of BmABC010557, all other ABC genes have corresponding probes in the oligonucleotide chip [44].
We used SilkMap tool in silkworm genome database to map the loci of each ABC genes on the 28 chromosomes. Genes in clusters, for example, BmABC000724 and BmABC000725 on Chromosome 1, are indicated by a vertical line (Additional File 4: Fig. S4).
Microarray analysis
We used silkworm genome-wide microarray data to profile the expression patterns of ABC genes in multiple larval tissues and during the larval-pupal-adult metamorphosis. The expression patterns of ABC transporter genes were estimated from intensity values [33]. From SilkDB, we downloaded normalized microarray data (GSE17571) for genome-wide gene expression in the anterior/middle silk gland (A/MSG), posterior silk gland (PSG), testis, ovary, fat body, midgut, integument, hemocyte, Malpighian tubule, and head on day 3 of the fifth larval instar. Normalized microarray data for gene expression at 19 time points during silkworm metamorphosis was recently performed (unpublished data from Southwest University). The sequential time points include V4 (day 4 of the fifth larval instar), V5, V6, V7, W0 (0 h after wandering), W12, W24, W36, W48, W60 (just after pupation), W72, W96, W120, W144, W168, W192, W216, W240, and adult (moth). The expression pattern of the ABC genes was estimated from intensity values. An ABC gene was considered to be expressed if its normalized intensity value exceeded 0 [34].
qRT-PCR
Total RNA was extracted from the whole body or selected tissues of different developmental stages and used for qRT-PCR analysis as previously described [13, 14]. Primers used here and somewhere else in this paper are listed in Additional File 13, Table S1.
Hormone treatments
As described previously [13, 14], day 2 of the fifth instar larvae (48 hrs after the fourth molting) was chosen for 20E injection (Sigma Aldrich, USA) (5 μg/larva) and the controls were injected with the same volume of control solvent. Six hours after 20E treatment, larvae were sacrificed to dissect tissues for qRT-PCR analysis. Ten animals were used for each group and 3 biological replicates were conducted.
RNAi
dsRNA was generated using the T7 RiboMAX™ Express RNAi system (Promega, USA). At the initiation of the early wandering stage, each individual larva was injected with GFP dsRNA (10 μg) or USP dsRNA (10 μg). Twenty four hours after RNAi treatment, the larvae were sacrificed for qRT-PCR analysis [13, 14]. Thirty animals were used for each group and 3 biological replicates were conducted.
References
Holland IB, Cole SPC, Kuchler K, Higgins CF: ABC proteins: from bacteria to man. 2003, London, UK:Academic Press
Dean M, Rzhetsky A, Allikmets R: The human ATP-binding cassette (ABC) transporter superfamily. Genome Res. 2001, 11: 1156-1166. 10.1101/gr.GR-1649R.
Dassa E, Bouige P: The ABC of ABCS: a phylogenetic and functional classification of ABC systems in living organisms. Res Microbiol. 2001, 152: 211-229. 10.1016/S0923-2508(01)01194-9.
Roth CW, Holm I, Graille M, Dehoux P, Rzhetsky A, Wincker P, Weissenbach J, Brey PT: Identification of the Anopheles gambiae ATP binding cassette transporter superfamily genes. Mol Cells. 2003, 15: 150-158.
Dean M, Hamon Y, Chimini G: The human ATP-binding cassette (ABC) transporter superfamily. J Lipid Res. 2001, 42: 1007-1017.
Hollenstein K, Dawson RJP, Locher KP: Structure and mechanism of ABC transporter proteins. Curr Opin Struc Biol. 2007, 17: 412-418. 10.1016/j.sbi.2007.07.003.
Saurin W, Hofnung M, Dassa E: Getting in or out: early segregation between importers and exporters in the evolution of ATP-binding cassette (ABC) transporters. J Mol Evol. 1999, 48: 22-41. 10.1007/PL00006442.
ABCISSE database (ABC systems: Information on Sequence, Structure and Evolution). [http://www1.pasteur.fr/recherche/unites/pmtg/abc/database.iphtml]
Dean M, Annilo T: Evolution of the ATP-binding cassette (ABC) transporter superfamily in vertebrates. Annu Rev Genomics Hum Genet. 2005, 6: 123-142. 10.1146/annurev.genom.6.080604.162122.
Labbé R, Caveney S, Donly C: Genetic analysis of the xenobiotic resistance associated ABC gene subfamilies of the Lepidoptera. Insect Mol Biol. 2011, 20: 243-256. 10.1111/j.1365-2583.2010.01064.x.
Mackenzie SM, Brooker MR, Gill TR, Cox GB, Howells AJ, Ewart GD: Mutations in the white gene of Drosophila melanogaster affecting ABC transporters that determine eye colouration. Biochim Biophys Acta. 1999, 1419: 173-185. 10.1016/S0005-2736(99)00064-4.
Hock T, Cottrill T, Keegan J, Garza D: The E23 early gene of Drosophila encodes an ecdysone-inducible ATP-binding cassette transporter capable of repressing ecdysone-mediated gene activation. Proc Natl Acad Sci USA. 2000, 97: 9519-9524.
Tian L, Guo E, Diao Y, Zhou S, Peng Q, Cao Y, Ling E, Li S: Genome-wide regulation of innate immunity by juvenile hormone and 20-hydroxyecdysone in the Bombyx fat body. BMC Genomics. 2010, 11: 549-10.1186/1471-2164-11-549.
Tian L, Guo E, Diao Y, Wang S, Liu S, Cao Y, Jiang RJ, Ling E, Li S: Developmental regulation of glycolysis by 20-hydroxyecdysone and juvenile hormone in fat body tissues of the silkworm, Bombyx mori. J Mol Cell Biol. 2010, 2: 255-263. 10.1093/jmcb/mjq020.
Wenzel JJ, Piehler A, Kaminski WE: ABC A-subclass proteins: gatekeepers of cellular phospho- and sphingolipid transport. Front Biosci. 2007, 12: 3177-3193. 10.2741/2305.
Sanchez-Fernandez R, Davies TGE, Coleman JOD, Rea PA: The Arabidopsis thaliana ABC protein superfamily, a complete inventory. J Biol Chem. 2001, 276: 30231-30244. 10.1074/jbc.M103104200.
Decottignies A, Goffeau A: Complete inventory of the yeast ABC proteins. Nat Genet. 1997, 15: 137-145.
Gottesman MM, Fojo T, Bates SE: Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer. 2002, 2: 48-58. 10.1038/nrc706.
Wu CT, Budding M, Griffin MS, Croop JM: Isolation and characterization of Drosophila multidrug resistance gene homologs. Mol Cell Biol. 1991, 11: 3940-3948.
Vache C, Camares O, Cardoso-Ferreira MC, Dastugue B, Creveaux I, Vaury C, Bamdad M: A potential genomic biomarker for the detection of polycyclic aromatic hydrocarbon pollutants: multidrug resistance gene 49 in Drosophila melanogaster. Environ Toxicol Chem. 2007, 26: 1418-1424. 10.1897/06-552R.1.
Ricardo S, Lehmann R: An ABC transporter controls export of a Drosophila germ cell attractant. Science. 2009, 323: 943-946. 10.1126/science.1166239.
Yepiskoposyan H, Egli D, Fergestad T, Selvaraj A, Treiber C, Multhaup G, Georgiev O, Schaffner W: Transcriptome response to heavy metal stress in Drosophila reveals a new zinc transporter that confers resistance to zinc. Nucleic Acids Res. 2006, 34: 4866-4877. 10.1093/nar/gkl606.
Theodoulou FL, Holdsworth M, Baker A: Peroxisomal ABC transporters. FEBS Lett. 2006, 580: 1139-1155. 10.1016/j.febslet.2005.12.095.
Kerr ID: Sequence analysis of twin ATP binding cassette proteins involved in translational control, antibiotic resistance, and ribonuclease L inhibition. Biochem Biophys Res Commun. 2004, 315: 166-173. 10.1016/j.bbrc.2004.01.044.
Zhou A, Hassel BA, Silverman RH: Expression cloning of 2-5Adependent RNAase: a uniquely regulated mediator of interferon action. Cell. 1993, 72: 753-765. 10.1016/0092-8674(93)90403-D.
Chen ZQ, Dong J, Ishimura A, Daar I, Hinnebusch AG, Dean M: The essential vertebrate ABCE1 protein interacts with eukaryotic initiation factors. J Biol Chem. 2006, 281: 7452-7457. 10.1074/jbc.M510603200.
Maeda T, Lee JM, Miyagawa Y, Koga K, Kawaguchi Y, Kusakabe T: Cloning and characterization of a ribonuclease L inhibitor from the silkworm, Bombyx mori. DNA Seq. 2005, 16: 21-27. 10.1080/10425170400028871.
Tyzack JK, Wang X, Belsham GJ, Proud CG: ABC50 interacts with eukaryotic initiation factor 2 and associates with the ribosome in an ATP-dependent manner. J Biol Chem. 2000, 275: 34131-34139.
Marton MJ, Vazquez de Aldana CR, Qu H, Chakraburtty K, Hinnebusch AG: Evidence that GCN1 and GCN20, translational regulators of GCN4, function on elongating ribosomes in activation of eIF2α kinase GCN2. Mol Cell Biol. 1997, 17: 4474-4489.
Kerr ID, Haider AJ, Gelissen IC: The ABCG family of membrane-associated transporters: you don't have to be big to be mighty. Br J Pharmacol. 2010
Abraham EG, Sezutsu H, Kanda T, Sugasaki T, Shimada T, Tamura T: Identification and characterisation of a silkworm ABC transporter gene homologous to Drosophila white. Mol Gen Genet. 2000, 264: 11-19. 10.1007/s004380000283.
Komoto N, Quan GX, Sezutsu H, Tamura T: A single-base deletion in an ABC transporter gene causes white eyes, white eggs, and translucent larval skin in the silkworm w-3(oe) mutant. Insect Biochem Mol Biol. 2009, 39: 152-156. 10.1016/j.ibmb.2008.10.003.
Tatematsu KI, Yamamoto K, Uchino K, Narukawa J, Iizuka T, Banno Y, Katsuma S, Shimada T, Tamura T, Sezutsu H, Daimon T: Positional cloning of silkworm white egg 2 (w-2) locus shows functional conservation and diversification of ABC transporters for pigmentation in insects. Genes Cell. 2011, 16: 331-342. 10.1111/j.1365-2443.2011.01490.x.
**a Q, Cheng D, Duan J, Wang G, Cheng T, Zha X, Liu C, Zhao P, Dai F, Zhang Z, He N, Zhang L, **ang Z: Microarray-based gene expression profiles in multiple tissues of the domesticated silkworm, Bombyx mori. Genome Biol. 2007, 8: R162-10.1186/gb-2007-8-8-r162.
Neira Oviedo M, Corena-McLeod MD, Linser PJ: A microarray-based analysis of transcriptional compartmentalization in the alimentary canal of Anopheles gambiae (Diptera:Culicidae) larvae. Insect Mol Biol. 2008, 17: 61-72. 10.1111/j.1365-2583.2008.00779.x.
Velamakanni S, Janvilisri T, Shahi S, van Veen HW: A functional steroid-binding element in an ATP-binding cassette mutidrug transporter. Mol Pharmacol. 2008, 73: 12-17. 10.1124/mol.108.038299.
Imamura M, Nakai J, Inoue S, Quan GX, Kanda T, Tamura T: Targeted gene expression using the GAL4/UAS system in the silkworm Bombyx mori. Genetics. 2003, 165: 1329-1340.
Tan AJ, Tanaka H, Tamura T, Shiotsuki T: Precocious metamorphosis in transgenic silkworms overexpressing juvenile hormone esterase. Proc Natl Acad Sci USA. 2005, 102: 11751-11756. 10.1073/pnas.0500954102.
Ma L, Xu H, Zhu J, Ma S, Liu Y, Jiang RJ, **a Q, Li S: Ras1CA overexpression in the posterior silk gland improves silk yield. Cell Res. 2011, 21: 934-943. 10.1038/cr.2011.36.
International Silkworm Genome Consortium: The genome of a lepidopteran model insect, the silkworm Bombyx mori. Insect Biochem Mol Biol. 2008, 38: 1036-1045. 10.1016/j.ibmb.2008.11.004.
Duan J, Li RQ, Cheng DJ, Fan W, Zha XF, Cheng TC, Wu YQ, Wang J, Mita K, **ang ZH, **a QY: SilkDB v2.0: a platform for silkworm (Bombyx mori) genome biology. Nucleic Acids Res. 2010, 38: D453-D456. 10.1093/nar/gkp801.
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Lipman DJ: Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 1997, 25: 3389-3402. 10.1093/nar/25.17.3389.
Thompson JD, Higgins DG, Gibson TJ: CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22: 4673-4680. 10.1093/nar/22.22.4673.
Tamura K, Dudley J, Nei M: MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol. 2007, 24: 1596-1599. 10.1093/molbev/msm092.
Acknowledgements
This study was supported by the 973 program (2012CB114605), the Natural Science Foundation of China (30770271), the Shanghai Municipal Education Commission (10JC1416700), and Chinese Academy of Sciences (KSCX2-EW-J-12) to SL. SL received the Outstanding Youth Investigator award (31125025) and the SA-SIBS award. It was also supported by the Natural Science Foundation of China (31101670) and Shanghai Institutes for Biological Sciences (2011KIP303) to LT. We highly appreciate Drs. Qingyou **a and Daojun Chen (Southwest University, Chongqing, China) for sharing and analyzing the unpublished microarray data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors' contributions
SL performed most of the experiments and analyzed the data. SZ, LT, EG helped with qRT-PCR, hormone treatment and RNAi. YL and JZ helped writing the paper and constructing the phylogenetic trees. SL designed the experiments, analyzed the data, wrote the paper, and coordinated the whole study. All authors approved the final manuscript.
Electronic supplementary material
12864_2011_3627_MOESM2_ESM.PDF
Additional file 2:Figure S2. Phylogenetic tree of the ABC transporters from the mosquito, Anopheles gambiae. (PDF 458 KB)
12864_2011_3627_MOESM3_ESM.PDF
Additional file 3:Figure S3. Phylogenetic tree of the ABC transporters from the flour beetle, Tribolium castaneum. (PDF 505 KB)
12864_2011_3627_MOESM4_ESM.PDF
Additional file 4:Figure S4. Locations of the silkworm ABC genes on chromosomes. Genes in clusters, for example, BmABC000724 and BmABC000725 on Chromosome 1, are indicated by a vertical line. (PDF 309 KB)
12864_2011_3627_MOESM5_ESM.PDF
Additional file 5:Figure S5. Phylogenetic tree of ABCA transporters from five insect spcies and the human. The phylogenetic tree was constructed using a neighbor-joining technique to analyze the amino acid sequences of the nucleotide binding domain (NBD). Analysis was performed with the program package MEGA4.0. The number at the branch point of the node represents the value resulting from 1000 replications and gaps were deleted with pairwise deletion method. Am, Apis mellifera; Ag, Anopheles gambiae; Bm, Bombyx mori; Dm, Drosophila melanogaster; Hs, Homo sapiens; Tc, Tribolium castaneum. (PDF 419 KB)
12864_2011_3627_MOESM6_ESM.PDF
Additional file 6:Figure S6. Phylogenetic tree of ABCB transporters from five insect species and the human. (PDF 395 KB)
12864_2011_3627_MOESM7_ESM.PDF
Additional file 7:Figure S7. Phylogenetic tree of ABCC transporters from five insect species and the human. (PDF 582 KB)
12864_2011_3627_MOESM8_ESM.PDF
Additional file 8:Figure S8. Phylogenetic tree of ABCD transporters from five insect species and the human. (PDF 252 KB)
12864_2011_3627_MOESM9_ESM.PDF
Additional file 9:Figure S9. Phylogenetic tree of ABCE and ABCF proteins from five insect species and the human. (PDF 269 KB)
12864_2011_3627_MOESM10_ESM.PDF
Additional file 10:Figure S10. Phylogenetic tree of ABCG transporters from five insect species and the human. (PDF 410 KB)
12864_2011_3627_MOESM11_ESM.PDF
Additional file 11:Figure S11. Phylogenetic tree of ABCH transporters from five insect species and the human. (PDF 254 KB)
12864_2011_3627_MOESM12_ESM.PDF
Additional file 12:Figure S12. Tissue distribution and developmental expression profiles of the 2 silkworm ABCG genes, including BmABC002581 and BmABC012035 in the brain from day 2 of 4th instar to day 2 of prepupae. M, molting; PP1, day 1 of prepupae; PP2, day 2 of prepupae. (PDF 397 KB)
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Liu, S., Zhou, S., Tian, L. et al. Genome-wide identification and characterization of ATP-binding cassette transporters in the silkworm, Bombyx mori. BMC Genomics 12, 491 (2011). https://doi.org/10.1186/1471-2164-12-491
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1471-2164-12-491