Abstract
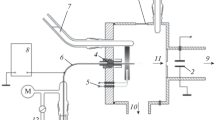
The structure of hydrated ions of dichloroacetic acid (DCA) is established by electrospraying aqueous solutions of DCA into the vacuum chamber of a mass spectrograph. At an acid concentration of 0.01 mol/L, the number of hydrated ions decreases monotonically with an increase in the degree of their hydration. The resulting mass spectrum provides information on the distribution of hydrated ions in solution, which makes the mass spectrographic method of spraying electrolytes in a vacuum applicable for determining the composition of electrolyte solutions in environmental studies.



Similar content being viewed by others
REFERENCES
F. Laturnus, I. Fahimi, M. Gryndler, et al., Environ. Sci. Poll. Res. 12, 233 (2005).
L. Weissflog, G. Krueger, N. Elansky, et al., Chemosphere 65, 975 (2006).
M. R. Frid and Yu. A. Treger, Vinyl Chloride: Chemistry and Technology (Kalvis, Moscow, 2008) [in Russian].
The Chemist’s Handbook, Ed. by B. P. Nikol’skii (Khimiya, Leningrad, 1971) [in Russian].
M. Clemens and H. F. Scholer, Fresenius J. Anal. Chem. 344, 47 (1992).
A. B. De Angelo, F. B. Daniel, B. M. Most, et al., J. Toxicol. Environ. Healt 52, 425 (1997).
Yu. V. Tertyshnaya, S. G. Karpova, and A. A. Popov, Russ. J. Phys. Chem. B 11, 531 (2017).
E. V. Shtamm, Yu. I. Skurlatov, A. V. Roshchin, V. O. Shvydkii, and L. V. Semenyak, Russ. J. Phys. Chem. B 13, 986 (2019).
Harmful Substances in Industry. A Handbook for Chemists, Engineers and Physicians, Ed. by N. V. Lazarev and E. N. Levina (Khimiya, Leningrad, 1976), Vol. 2 [in Russian].
List and Codes of Air Pollutants (AO NII Atmosfera, St. Petersburg, 2015) [in Russian].
R. J. B. Peters, J. Environ. Monitor. 5, 275 (2003).
J. C. Wenger, S. le Calve, H. W. Sidebottom, et al., Environ. Sci. Technol. 38, 831 (2004).
H. Sidebottom and J. Franklin, Pure Appl. Chem. 68, 1757 (1996).
L. L. Lifongo, D. J. Bowden, and P. Brimblecombe, Int. J. Phys. Sci. 5, 738 (2010).
S. Reimann, K. Grob, and H. Frank, Environ. Sci. Technol. 30, 2340 (1996).
Yu. I. Skurlatov, E. V. Vichutinskaya, N. I. Zaitseva, E. V. Shtamm, V. O. Shvydkii, L. V. Semenyak and I. S. Baikova, Russ. J. Phys. Chem. B 11, 576 (2017).
G. V. Karpov, O. A. Vinogradova, E. S. Vasil’ev, and I. I. Morozov, Russ. J. Phys. Chem. B 12, 683 (2018).
G. V. Karpov, I. I. Morozov, E. S. Vasiliev, et al., Chem. Phys. Lett. 586, 40 (2013).
G. V. Karpov, Russ. J. Electrochem. 46, 95 (2010).
G. V. Karpov, E. S. Vasiliev, I. I. Morozov, et al., Int. J. Chem. Kinet. 48, 442 (2016).
Funding
This study was carried out as part of a state assignment of the Ministry of Science and Higher Education of the Russian Federation (topic no. АААА-А20-120021390044-2) and was supported by grant no. 19-05-50076 (Microcosm) from the Russian Foundation for Basic Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karpov, G.V., Vasiliev, E.S., Volkov, N.D. et al. Specific Features of Dichloroacetic Acid Hydration. Russ. J. Phys. Chem. B 14, 829–834 (2020). https://doi.org/10.1134/S1990793120050218
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793120050218