Abstract
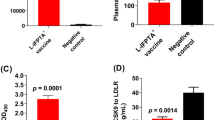
The aim of this study was to study the activity of lysosomal chitinases (chitotriosidase and β-glucosaminidase) in the liver of mice on a model of BCG-induced tuberculous inflammation after intravenous administration of apolipoprotein A-I. The study was carried out on CBA male mice weighing 20–22 g. Disseminated tuberculous inflammation was modeled by a single intraperitoneal administration of 0.5 mg of BCG vaccine. The activity of chitinases was determined using 4-methylumbelliferyl-β-D-N,N',N''-triacetylchitotrioside and 4-methylumbelliferyl-N-acetyl-β-D-glucosaminidine fluorescent substrates. BCG infection of animals after 4 weeks caused a significant increase in the activity of endogenous chitinases as compared with the control group. Thus, the activity of chitotriosidase increased by 3.05 times (p < 0.001), while that of β-glucosaminidase increased by 1.76 times (p < 0.01). The intravenous administration of apolipoprotein A-I to animals against the background of BCG infection inhibited the increased enzyme activity, the values did not differ significantly from the control values. The results of the conducted studies indicate the ability of apolipoprotein A-I to decrease the increased activity of endogenous lysosomal chitinases in the liver of mice with BCG-induced tuberculous inflammation.
Similar content being viewed by others
REFERENCES
Bargagli, E., Margollicci, M., Nikiforakis, N., Luddi, A., Perrone, A., Grosso, S. and Rottoli, G., Chitotriosidase activity in the serum of patients with sarcoidosis and pulmonary tuberculosis, Respiration, 2007, vol. 74, p. 548. https://doi.org/10.1159/000100555
Boot, R.G., Renkema, G.H., Verhoek, M., Strijland, A., Bliek, J., de Meulemeester T.M., Mannens, M.M. and Aerts, J.M., The human chitotriosidase gene. Nature of inherited enzyme deficiency, J. Biol. Chem., 1998, vol. 273, p. 25680. https://doi.org/10.1074/jbc.273.40.25680
Boot, R.G., Blommaart, E.F., Swart, E., Ghauharali-van der Vlugt, K., Bijl, N., Moe, C., Place, A. and Aerts, J.M., Identification of a novel acidic mammalian chitinase distinct from chitotriosidase, J. Biol. Chem., 2001, vol. 276, p. 6770. https://doi.org/10.1074/jbc.M009886200
Burger, D. and Dayer, J.M., High-density lipoprotein-associated apolipoprotein A-I: the missing link between infection and chronic inflammation? Autoimmun. Rev., 2002, vol. 1, p. 111. https://doi.org/10.1016/s1568-9972(01)00018-0
Chua, J., Vergne, I., Master, S. and Deretic, V., A tale of two lipids: Mycobacterium tuberculosis phagosome maturation arrest, Curr. Opin. Microbiol., 2004, vol. 7, p. 71. https://doi.org/10.1016/j.mib.2003.12.011
di Rosa, M., Brundo, V.M., and Malaguarnera, L., New insights on chitinases immunologic activities., World J. Immunol., 2016, vol. 6, p. 96. https://doi.org/10.4049/jimmunol.172.3.181
Elmonem, M.A., van den Heuvel, L.P. and Levtchenko, E.N., Immunomodulatory effects of chitotriosidase enzyme, Enzyme Res., 2016, vol. 2016, p. 2682680. https://doi.org/10.1155/2016/2682680
Hmama, Z., Sendide, K., Talal, A., Garcia, R., Dobos, K., and Reiner, N.E., Quantitative analysis of phagolysosome fusion in intact cells: inhibition by mycobacterial lipoarabinomannan and rescue by an 1alpha,25-dihydroxyvitamin D3-phosphoinositide 3-kinase pathway, J. Cell. Sci., 2004, vol. 117, p. 2131. https://doi.org/10.1242/jcs.01072
Hollak, C.E., van Weely, S., van Oers, M.H., and Aerts, J.M., Marked elevation of plasma chitotriosidase activity. A novel hallmark of Gaucher disease, J. Clin. Invest., 1994, vol. 93, p. 1288. https://doi.org/10.1172/JCI117084
Hyka, N., Dayer, J.M., Modoux, C., Kohno, T., Edwards, C.K., Roux-Lombard, P., and Burger, D., Apolipoprotein A-I inhibits the production of interleukin-1 beta and tumor necrosis factor-alpha by blocking contact-mediated activation of monocytes by T lymphocytes, Blood, 2001, vol. 97, p. 2381. https://doi.org/10.1182/blood.v97.8.2381
Jungbauer, C.G., Uecer, E., Stadler, S., Birner, C., Buchner, S., Maier, L.S., and Luchner A., N-acteyl-ß-D-glucosaminidase and kidney injury molecule-1: new predictors for long-term progression of chronic kidney disease in patients with heart failure, Nephrology (Carlton), 2016, vol. 21, p. 490. https://doi.org/10.1111/nep.12632
Kim, D.H., Park, H.J., Lim, S., Lee, H.G., Koo, J.H., Lee, H.G., Choi, J.O., Oh, J.H., Ha, S.J., Min-Jong, K., and Choi, J.M., Regulation of chitinase-3-like-1 in T cell elicites Th1 nad cytotoxic responses to inhibit lung metastasis, Nat. Commun., 2018, vol. 9, p. 503. https://doi.org/10.1038/s41467-017-02731-6
Pandit, S., Roy, S., Pillai, J., and Banerjee, S., Formulation and intracellular trafficking of lipid-drug conjugate nanoparticles containing a hydrophilic antitubercular drug for improved intracellular delivery to human macrophages, ACS Omega, 2020, vol. 5, p. 4433. https://doi.org/10.1021/acsomega.9b03523
Polyakov, L.M., Sumenkova, D.V., and Panin, L.E., Effect of plasma lipoproteins and their complexes with polysaccharides on interleukin-1β concentration in macrophages of mice with HA-1 ascitic hepatoma, Bull. Exp. Biol. Med., 2009, vol. 147, p. 466. https://doi.org/10.1007/s10517-009-0557-4
Polyakov, L.M., Knyazev, R.A., Kotova, M.V., Russkikh, G.S., Solov’eva, E.I., and Ryabchenko, A.V., Apolipoprotein A-I Iincreases the activity of lysosomal glycosidases in the liver of mice with BCG-induced tuberculosis inflammation, Sib. Nauchn. Med. Zh., 2021, vol. 41, p. 51. https://doi.org/10.18699/SSMJ20210605
Sumenkova, D.V., Polyakov, L.M., and Panin L.E., Influence of isoniazid complex with A-I apolipoprotein on activity of lysosomal enzymes in mice with tuberculous inflammation model, Eksp. Klin. Farmakol., 2012, vol. 75, p. 29.
Tada, N., Sakamoto, T., Kagami, A., Mochizuk, K., and Kurosaka, K., Antimicrobial activity of lipoprotein particles containing apolipoprotein A-I
l., Mol. Cell. Biochem., 1993, vol. 119, p. 171. https://doi.org/10.1007/BF00926868Tasci, C., Tapan, S., Ozkaya, S., Demirer, E., Deniz, O., Balkan, A., Ozkan, M., Inan, I., Kurt, I., and Bilgic, H., Efficacy of serum chitotriosidase activity in early treatment of patients with active tuberculosis and a negative sputum smear, Ther. Clin. Risk Manage., 2012, vol. 8, p. 369. https://doi.org/10.2147/tcrm.s31752
Tronsmo, A., and Harman, G.E., Detection and quantification of N-acetyl-beta-D-glucosaminidase, chitobiosidase, and endochitinase in solutions and on gels, Anal. Biochem., 1993, vol. 208, p. 74. https://doi.org/10.1006/abio.1993.1010
Turk, J., ŞahutoĞlu, A.S., Hatice, H., Frese, S.A., and Karav, S., Structural insights of two novel N-acetyl-glucosaminidase enzymes through in silico methods, Turk. J. Chem., 2020, vol. 44, p. 1703. https://doi.org/10.3906/kim-2006-19
Vergne, I., Chua, J., Lee, H., Lucas, M., Belisle, J., and Deretic, V., Mechanism of phagolysosome biogenesis block by viable Mycobacterium tuberculosis, Proc. Natl. Acad. Sci. U. S. A., 2005, vol. 102, p. 4033. https://doi.org/10.1073/pnas.0409716102
Wadham, C., Albanese, N., Roberts, J., Wang, L., Bagley, C. J., Gamble, J.R., Rye, K.A., Barter, P.J., Vadas, M.A., and **a, P., High-density lipoproteins neutralize C-reactive protein proinflammatory activity, Circulation, 2004, vol. 109, p. 2116. https://doi.org/10.1161/01.CIR.0000127419.45975.26
Yin, K., Chen, W.J., Zhou, Z.G., Zhao, G.J., Lv, Y.C., Ouyang, X.P., Yu, X.H., Fu, Y., Jiang, Z.S., and Tang, C.K., Apolipoprotein A-I inhibits CD40 proinflammatory signaling via ATP-binding cassette transporter A1-mediated modulation of lipid raft in macrophages, J. Atheroscler. Thromb., 2012, vol. 19, p. 823. https://doi.org/10.5551/jat.12823
Zhu, Z., Homer, R.J., Kim, Y.K., Chen, N.Y., Cohn, L., Qutayba, H.Q., and Elias, J.A., Acidic mammalian chitinase in asthmatic Th2 inflammation and IL-13 pathway activation, Science, 2004, vol. 304, p. 1678. https://doi.org/10.1126/science.109533
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflicts of interest.
Statement on the welfare of animals. The kee**, nutrition, and care of animals and their removal from the experiment were performed according to the principles of humane treatment set out in the “Rules for Conducting Works Using Experimental Animals.”
Additional information
Translated by A. Barkhash
Rights and permissions
About this article
Cite this article
Polyakov, L.M., Kotova, M.V., Trifonova, N.V. et al. Apolipoprotein A-I Inhibits Increased Activity of Chitotriosidase and β-Glucosaminidase in the Liver of Mice with BCG-Induced Tuberculous Inflammation. Cell Tiss. Biol. 17, 436–440 (2023). https://doi.org/10.1134/S1990519X23040090
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X23040090