Abstract
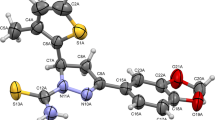
Objective: The tandem Knoevenagel-cyclocondensation reaction of aromatic aldehydes, malonitrile, and phenylhydrazine in water and ethanol at room temperature is described as an effective, one-pot, threecomponent synthesis of many scientifically relevant heterocyclic compounds. Methods: As a part of our efforts, we have synthesized pyrazole-4-carbonitriles and characterized using various spectroscopic methods and subjected for antimicrobial analysis. Results and Discussion: From our analysis, we observed best in vitro candidate as 5-amino-3-(2,5-difluorophenyl)-1-phenyl-1H-pyrazole-4-carbonitrile. The molecular docking analysis on common bacterial target suggested the involvement of 2,2-dialkylglycine decarboxylase (PDB ID: 1D7U) as a target for compound (IVi) (docking score: –9.32 kcal/mol). Furthermore, a molecular dynamic simulation of 100 ns resulted in the stability of best docked candidate, (IVi): 1D7U. Conclusions: We propose that best docked candidate, (IVi) as potential antimicrobial agents.








Similar content being viewed by others
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
Heravi, M.M. and Zadsirjan, V., RSC Adv., 2020,vol. 10, pp. 44247–44311. https://doi.org/10.1039/D0RA09198G
Kaushik, N.K., Kaushik, N., Attri, P., Kumar, N.,Kim, C.H., Verma, A.K., and Choi, E.H., Molecules, 2013, vol. 18, pp. 6620–6662.
Saini, M.S., Kumar, A., Dwivedi, J., and Singh, R.,Int. J. Pharma Sci. Res., 2013, vol. 4, pp. 66–77.
Ameziane El Hassani, I., Rouzi, K., Assila, H., Karrouchi, K., and Ansar, M.H., Reactions, 2023, vol. 4,pp. 478–504. https://doi.org/10.3390/reactions4030029
Fustero, S., Sanchez-Rosello, M., Barrio, P., andSimon-Fuentes, A., Chem. Rev., 2011, vol. 111,pp. 6984–7034. https://doi.org/10.1021/cr2000459
Kumar, V., Kaur, K., Gupta, G.K., and Sharma, A.K.,Eur. J. Med. Chem., 2013, vol. 69, pp. 735–753. https://doi.org/10.1016/j.ejmech.2013.08.053
Küçükgüzel, Ş.G. and Şenkardeş, S., Eur. J. Med. Chem., 2015, vol. 97, pp. 786–815.
Aziz, H., Zahoor, A.F., and Ahmad, S., J. Chilean Chem. Soc., 2020, vol. 65, pp. 4746–4753.
Burgart, Y.V., Agafonova, N.A., Shchegolkov, E.V., Krasnykh, O.P., Kushch, S.O., Evstigneeva, N.P., Gerasimova, N.A., Maslova, V.V., Triandafilova, G.A., Solodnikov, S.Y., and Ulitko, M.V., Eur. J. Med. Chem., 2020, vol. 208, Article ID: 112768. https://doi.org/10.1016/j.ejmech.2020.112768
Neto, J.S. and Zeni, G., Chem. Eur. J., 2020, vol. 26,pp. 8175–8189. https://doi.org/10.1002/chem.201905276
Kosurkar, U.B., Pamanji, R., Janardhan, S., Nanubolu, J., Dadmal, T.L., Mali, S.N., and Kumbhare, R.M., Russ. J. Bioorg. Chem., 2022, vol. 48, pp. S154–S165. https://doi.org/10.1134/S1068162023010144
Shelke, D.E., Thorat, B.R., Mali, S.N., and Dhabarde, S.S., Russ. J. Bioorg. Chem., 2022, vol. 48,pp. S74–S86. https://doi.org/10.1134/S1068162023010259
Mali, S.N. and Pandey, A., Curr. Comput. – Aided Drug Des., 2022, vol. 18, pp. 108–122. https://doi.org/10.2174/1573409918666220929145824
Mali, S.N. and Pandey, A., Chem. Africa, 2022,vol. 5, pp.1215–1236. https://doi.org/10.1007/s42250-022-00462-w
Pandey, A., Shyamal, S.S., Shrivastava, R., Ekka, S.,and Mali, S.N., Chem. Africa, 2022, vol. 5, pp. 1469–1491. https://doi.org/10.1007/s42250-022-00449-7
Bhosale, D., Mali, S.N., Thorat, B.R., Wavhal, S.S., Bhagat, D.S., and Borade, R.M., Recent Adv. AntiInfect. Drug Dis. Formerly Recent Patents AntiInfect. Drug Dis., 2022, vol. 17, pp. 69–83. https://doi.org/10.2174/1570193X19666220531154544
Mali, S.N. and Pandey, A., J. Comput. Biophys. Chem.,2022, vol. 21, pp. 857–882. https://doi.org/10.1142/S2737416522500387
Desale, V.J., Mali, S.N., Thorat, B.R., and Yamgar, R.S.,Curr. Comput. – Aided Drug Des., 2021, vol. 17,pp. 493–503. https://doi.org/10.2174/1573409916666200615141047
Kshatriya, R., Shelke, P., Mali, S., Yashwantrao, G.,Pratap, A., and Saha, S., ChemistrySelect, 2021, vol. 6,pp. 6230–6239. https://doi.org/10.1002/slct.202101083
Mali, S.N. and Pandey, A., J. Indian Chem. Soc., 2021,vol. 98, Article ID: 100082. https://doi.org/10.1016/j.jics.2021.100082
Chopade, A.R., Pol, R.P., Patil, P.A., Dharanguttikar, V.R., Naikwade, N.S., Dias, R.J., and Mali, S.N.,Comb. Chem. High Throughput Screen., 2021, vol. 24,pp. 415–422. https://doi.org/10.2174/1386207323666200605150915
Nagre, D.T., Mali, S.N., Thorat, B.R., Thorat, S.A., Chopade, A.R., Farooqui, M., and Agrawal, B., Curr. Enzym.Inhibit., 2021, vol. 17, pp. 127–143. https://doi.org/10.2174/1573408017666210203203735
Anuse, D.G., Thorat, B.R., Sawant, S., Yamgar, R.S., Chaudhari, H.K., and Mali, S.N., Curr. Comput. – Aided Drug Des., 2020, vol. 16, pp. 530–540. https://doi.org/10.2174/1573409915666190902143648
Kapale, S.S., Mali, S.N., and Chaudhari, H.K., Med. Drug Dis., 2019, vol. 2, Article ID: 100008. https://doi.org/10.1016/j.medidd.2019.100008
Thorat, B.R., Mali, S.N., Rani, D., and Yamgar, R.S., Curr. Comput. – Aided Drug Des., 2021, vol. 17,pp. 294–306. https://doi.org/10.2174/1573409916666200302120942
Mali, S.N. and Pandey, A., J. Comput. Biophys. Chem.,2021, vol. 20, pp. 267–282. https://doi.org/10.1142/S2737416521500125
Mali, S.N. and Pandey, A., J. Comput. Biophys. Chem.,2022, vol. 21, pp. 83–114. https://doi.org/10.1142/S2737416521410015
Mali, S.N., Tambe, S., Pratap, A.P., and Cruz, J.N.,Essential Oils, Springer, 2022, vol. 1, pp. 417–442. https://doi.org/10.1007/978-3-030-99476-1_18
Rout, S., Tambe, S., Deshmukh, R.K., Mali, S., Cruz, J., Srivastav, P.P., Amin, P.D., Gaikwad, K.K., de Aguiar Andrade, E.H., and de Oliveira, M.S., Trend. Food Sci. Technol., 2022, vol. 129, pp. 421–439. https://doi.org/10.1016/j.tifs.2022.10.012
Mali, S.N., Pandey, A., Bhandare, R.R., and Shaik, A.B.,Sci. Rep., 2022, vol. 12, pp. 1–21. https://doi.org/10.1038/s41598-022-20325-1
Mali, S.N., Pandey, A., Thorat, B.R., and Lai, C.H., Struct. Chem., 2022, vol. 33, pp. 679–694. https://doi.org/10.1007/s11224-022-01879-2
Ghosh, S., Mali, S.N., Bhowmick, D.N., and Pratap, A.P.,J. Indian Chem. Soc., 2021, vol. 98, Article ID: 100088. https://doi.org/10.1016/j.jics.2021.100088
Thorat, B.R., Mali, S.N., Wagh, R.R., and Yamgar, R.S.,Curr. Comput. – Aided Drug Des., 2022, vol. 18,pp. 247–257. https://doi.org/10.2174/1573409918666220610162158
Mali, S.N., Thorat, B.R., Gupta, D.R., and Pandey, A., Engineer. Proceedings, 2021, vol. 11, p. 21. https://doi.org/10.3390/ASEC2021-11157
Pyrkov, T.V., Ozerov, I.V., Balitskaya, E.D., and Efremov, R.G., Russ. J. Bioorg. Chem., 2010, vol. 36,pp. 446–455. https://doi.org/10.1134/S1068162010040023
Maksimenko, A.V. and Beabealashvili, R.S., Russ. J. Bioorg. Chem., 2018, vol. 44, pp. 165–172. https://doi.org/10.1134/S1068162018020048
Renuka, N., Vivek, H.K., Pavithra, G., and Ajay Kumar, K., Russ. J. Bioorg. Chem., 2017, vol. 43,pp.197–210. https://doi.org/10.1134/S106816201702011X
Savateev, K.V., Ulomsky, E.N., Fedotov, V.V., Rusinov, V.L., Sivak, K.V., Lyubishin, M.M., Kuzmich, N.N., and Aleksandrov, A.G., Russ. J. Bioorg. Chem., 2017, vol. 43,pp. 421–428. https://doi.org/10.1134/S1068162017040094
Sun, X., Belal, A., Elanany, M.A., Alsantali, R.I.,Alrooqi, M.M., Mohamed, A.R., and Hasabelnaby, S., Russ. J. Bioorg. Chem., 2022, vol. 48, pp. 438–456. https://doi.org/10.1134/S1068162022330019
Glide, Schrodinger, LLC, NY, 2022. https://www.schrodinger.com/platform/drug-discovery
Nikpassand, M., Fekri, L.Z., and Khalatbari, S., Russ. J. Bioorg. Chem., 2023, vol. 49, pp. 1112–1118. https://doi.org/10.1134/S1068162023050187
Sudhakar, M., Gutam, M., Yerrabelli, J.R., Irlapati, V.K., Gorityala, N., Sagurthi, S.R., and Chitneni, P.R., Russ. J.Bioorg. Chem., 2020, vol. 46, pp. 845–855. https://doi.org/10.1134/S1068162020050179
Srinivas, K., Kumar, P.V., Joshi, H., Velidandi, A., and Manchal, R., Russ. J. Bioorg. Chem., 2023, vol. 49,pp. 1068–1075. https://doi.org/10.1134/S106816202305014X
Dag, B., Tenekecioğlu, Y., Aral, T., Kızılkaya, H.,Erenler, R., and Genc, N., Russ. J. Bioorg. Chem., 2023,vol. 49, pp. 861–866. https://doi.org/10.1134/S1068162023040106
ACKNOWLEDGMENTS
The authors are grateful to the Principal and Head, CS’s S.S. and L.S. Patkar College of Arts and Science, and V.P. Varde College of Commerce and Economics, Goregaon, Mumbai, 400104 India for his constant encouragement.
The author SM is thankful to Department of Pharmaceutical Sciences and Technology, Birla Institute of Technology, Mesra for usage of facility of Schrodinger Drug Discovery module, 2023.
Funding
This work was supported by regular institutional funding, and no additional grants were obtained.
Author information
Authors and Affiliations
Contributions
The authors NJ, BT, SY, ST, and RY—selected the literature data on the research topic. The authors SM, NJ, and BT—contributed to manuscript preparation.
All authors participated in the discussions.
Corresponding authors
Ethics declarations
This article does not contain any studies involving patients or animals as test objects.
Informed consent was not required for this article. No conflict of interest was declared by the authors.
Additional information
Publisher's Note. Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Jha, N.N., Thorat, B.R., Yadav, S. et al. Catalyst-Free, One-Pot, Three-Component Synthesis of 5-Amino-1,3-diphenyl-1H-pyrazole-4-carbonitriles in Green Media, and Evaluation of Their Biological Activities. Russ J Bioorg Chem 50, 949–961 (2024). https://doi.org/10.1134/S1068162024030208
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162024030208