Abstract
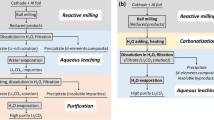
Theoretical foundations of technology for producing lithium carbonate of battery quality by the ammonia method have been developed. This technology is based on the precipitation of lithium carbonate from an aqueous solution of lithium chloride by its direct contact with a gaseous mixture of NH3 and CO2 in a 2 : 1 ratio, followed by NH3 reproduction by the decomposition of ammonium chloride with calcium hydroxide, which is a byproduct of interactions in the LiCl−NH3–CO2−H2O system. The applicability of the ammonia method for obtaining battery-quality Li2CO3 from sulfate, nitrate, and mixed lithium solutions has been shown.





Similar content being viewed by others
REFERENCES
Analiz kon"yunktury mirovogo rynka litiya i ego soedinenii na nachalo XXI v. Otchet po proektu “Litii Rossii” (Analysis of the State of the World Market for Lithium and Its Compounds at the Beginning of the 21st Century: A Report on the Project “Lithium of Russia”), Novosibirsk: Sib. Otd., Ross. Akad. Nauk, 2008.
Kudryavtsev, P.G., Lithium: World’s reserves and prospects for use, Al’tern. Energ. Ekol., 2016, nos. 13–14, p. 62.
Obzor rynka litiya i ego soedinenii v mire i Rossii (A Review of the Market for Lithium and Its Compounds in the World and Russia), Moscow: Infomain, 2017, 2nd ed.
Burmistrova, N.A., Sycheva, V.A., Churikov, A.V., and Ivanishcheva, I.A., Lithium iron phosphate LiFePO4 as a cathode material for lithium-ion batteries, Elektrokhim. Energ., 2009, vol. 9, no. 4, p. 188.
MarketWatch, Global lithium market outlook to 2026. www.marketwatch.com/story/the-global-lithium-market-outlook-to-2026-2026-11-15-4203139. Accessed April 28, 2018.
Kotsupalo, N.P. and Ryabtsev, A.D., Khimiya i tekhnologiya polucheniya soedinenii litiya iz litienosnogo gidromineral’nogo syr’ya (Chemistry and Technology of the Production of Lithium Compounds from Lithium-Containing Hydromineral Raw Materials), Novosibirsk: GEO, 2008.
MOST Marketing, Marketing research on the project “Organization of the production of lithium carbonate and coproducts from natural brines”, MOST Marketing Report. http://www.m-marketing.ru. Accessed April 28, 2018.
Ryabtsev, A.D., Menzheres, L.T., Kurakov, A.A., and Gushchina, E.P., Interaction of ammonium bicarbonate with lithium chloride solutions, Theor. Found. Chem. Eng., 2006, vol. 40, no. 6, pp. 649–654. https://doi.org/10.1134/S0040579506060157
Ryabtsev, A.D., Titarenko, V.I., Kotsupalo, N.P., et al., RF Patent 2544806, 2015.
Plyushchev, V.E. and Stepin, B.D., Khimiya i tekhnologiya soedinenii litiya, rubidiya, tseziya (Chemistry and Technology of Compounds of Lithium, Rubidium, and Cesium), Moscow: Khimiya, 1970.
Zaitsev, I.D., Tkach, G.A., and Stoev, N.D., Proizvodstvo sody (Soda Production), Moscow: Khimiya, 1986.
Eksperimental'nye dannye po rastvorimosti mnogokomponentnykh vodno-solevykh sistem: spravochnik. V 2-kh tomakh, 4-kh knigakh (Experimental Data on the Solubility of Multicomponent Water–Salt Systems: A Handbook in Two Volumes and Four Books), St. Petersburg: Khimizdat, 2003, 3rd ed.
Ryabtsev, A.D., Titarenko, V.I., Kotsupalo, N.P., et al., RF Patent 2560359, 2015.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by D. Kharitonov
Rights and permissions
About this article
Cite this article
Ryabtsev, A.D., Kotsupalo, N.P., Kurakov, A.A. et al. Theoretical Foundations of Technology for the Production of Lithium Carbonate by the Ammonia Method. Theor Found Chem Eng 53, 815–820 (2019). https://doi.org/10.1134/S0040579519040274
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0040579519040274