Abstract
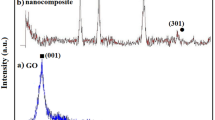
Modified Hummers’ method was employed for the synthesis of graphene oxide, which was used as a precursor for the synthesis of reduced graphene oxide by using chemical reduction method. Sol-gel method was applied for the synthesis of gadolinium oxide and gadolinium oxide/reduced graphene oxide (Gd2O3/rGO) nanocomposite in the presence of dioctyl sulfosuccinate sodium salt. The prepared material was characterized by using UV-Visible spectroscopy, FTIR, TGA, SEM, and XRD. UV-Visible spectroscopy was used to find the band gap, which was found 4.6 eV for Gd2O3/rGO nanocomposite. FTIR helps in identifying the functional groups. TGA was used to determine thermal stability, kinetic and thermodynamic parameters. Crystallite size of Gd2O3/rGO nanocomposite was determined as 4.38 nm using XRD data. SEM was used to study morphology of prepared material, while degradation efficiency of nanocomposite was checked against methylene blue dye. Gadolinium oxide showed only 19.3% photocatalytic activity in 75 min. Gadolinium oxide/reduced graphene oxide nanocomposite displayed higher degradation efficiency (70% in 75 min) as compared with pure gadolinium oxide against methylene blue.













Similar content being viewed by others
REFERENCES
M. S. Usman, M. Z. Hussein, A. U. Kura, et al., Appl. Nanosci. 8, 973 (2018).
S. Perveen and M. A. Farrukh, J. Mater. Sci.: Mater. Electron. 28, 10806 (2017).
G. Sharma, D. Prema, Venkataprasanna, et al., Arab. J. Chem. 13, 7680 (2020).
M. J. Akhtar, M. Ahamed, and H. Alhadlaq, Nanomaterials 10 (9), 1 (2020).
A. Afzaal and M. A. Farrukh, Mater. Sci. Eng. B 223, 167 (2017).
Y. D. Yin, G. Y. Hong, and B. F. **n, Chin. Chem. Lett. 18, 491 (2007).
M. A. Farrukh, I. Muneer, S. Sharif, Z. Munawar, and N. Batool, Mater. Res. Express 5, 1 (2018).
M. A. Farrukh, M. Shahid, I. Muneer, S. Javaid, and M. Khaleeq-ur-Rahman, J. Mater. Sci.: Mater. Electron. 27, 2994 (2015).
T. Saravanan, P. Anan, M. Shanmugam, et al., Solid State Sci. 83, 171 (2018).
S. Javaid, M. A. Farrukh, I. Muneer, et al., Superlatt. Microstruct. 82, 234 (2015).
K. Dědková, L. Kuzníková, L. Pavelek, et al., Mater. Chem. Phys. 197, 226 (2017).
K. Ye, J. Wang, N. Li, et al., Inorg. Chem. Commun. 45, 116 (2014).
S. Jeon, J. W. Ko, and W. B. Ko, Catalysts 11, 742 (2021).
S. Manavalan, U. Rajaji, S.-M. Chen, et al., J. Electroanal. Chem. 837, 167 (2019).
J. Liu, L. Huang, X. Tian, et al., Int. J. Nanomed. Nanosurg. 12, 1 (2017).
L. P. Lingamdinne, S. Lee, J.-S. Choi, et al., Appl. J. Hazard. Mater. 402, 1 (2020).
E. Correa, B. Bosch-Santos, F. Cavalcante, et al., Am. Inst. Phys. Adv. 6 (5), 1 (2016).
J.-G. Kang, B.-K. Min, and Y. Sohn, Ceram. Int. 41, 1243 (2015).
S. A. Khan, S. Gambhir, and A. Ahmad, Beilstein J. Nanotechnol. 5, 249 (2014).
H. N. Sheikh, H. Khajuria, J. Ladol, et al., Sloven. Chem. Soc. 62, 849 (2015).
M. A. Farrukh, F. Naseem, A. Imtiaz, et al., Russ. J. Phys. Chem. A 90, 1231 (2016).
F. Iskandar, U. Hikmah, E. Stavila, and A. H. Aimon, R. Soc. Chem. Adv. 7, 52391 (2017).
J. Liu, Z. Wang, L. Liu, and W. Chen, Phys. Chem. Chem. Phys. 13, 13216 (2011).
Q. Gao, Z. Yuan, L. Dong, et al., Electrochim. Acta 270, 417 (2018).
L. Wang, L. Miao, H. Yang, et al., Electrochim. Acta 281, 638 (2018).
V. Vinesh, A. Shaheer, and B. Neppolian, Ultrason. Sonochem. 50, 302 (2019).
M. Govindasamy, S. Manavalan, S.-M. Chen, et al., Sens. Actuat., B 272, 274 (2018).
H. Li, G. Wang, F. Zhang, et al., R. Soc. Chem. Adv. 2, 12413 (2012).
M. S. Bakshi, Cryst. Growth Des. 16, 1104 (2015).
H. Perveen, M. A. Farrukh, M. Khaleeq-ur-Rahman, et al., Russ. J. Phys. Chem. A 89, 99 (2014).
N. Aslam, M. A. Farrukh, and S. Karim, Russ. J. Phys. Chem. A 93, 1610 (2019).
A. Imtiaz, M. A. Farrukh, M. Khaleeq-ur-Rahman, and R. Adnan, Sci. World J. 2013, 1 (2013).
M. A. Farrukh, P. Tan, and R. Adnan, Turk. J. Chem. 36, 303 (2012).
A. Javaid and M. A. Farrukh, J. Chin. Chem. Soc. 70, 32 (2023).
I. Muneer, M. A. Farrukh, D. Ali, and F. Bashir, Mater. Sci. Eng. B 265, 115028 (2021).
H. Liu, C. Wang, and G. Wang, Chem. Asian J. 15, 3239 (2020).
V. L. E. Siong, X. H. Tai, K. M. Lee, et al., R. Soc. Chem. Adv. 10, 37905 (2020).
P. Kumar, R. Agnihotri, K. L. Wasewar, et al., Desalin. Water Treatm. 50, 226 (2012).
S. Gnanam and V. Rajendran, J. Alloys Compd. 735, 1854 (2018).
I. Muneer, M. A. Farrukh, S. Javaid, et al., Superlatt. Microstruct. 77, 256 (2015).
S. Yang, H. Gao, Y. Wang, et al., Mater. Res. Bull. 48, 37 (2013).
A. T. M. Anishur Rahman, K. Vasilev, and P. Majewski, J. Colloid Interface Sci. 354, 592 (2011).
E. Aliyev, V. Filiz, M. M. Khan, et al., Nanomaterials 9, 1180 (2019).
S. Thakur and N. Karak, Carbon 50, 5331 (2012).
P. Chand, S. Vaish, and P. Kumar, Phys. B (Amsterdam, Neth.) 524, 53 (2017).
F. Anwar and M. A. Farrukh, Asian J. Chem. 27, 3571 (2015).
M. A. Farrukh, K. M. Butt, K.-K. Chong, and W. S. Chang, J. Saudi Chem. Soc. 23, 561 (2018).
J. Chen, B. Yao, C. Li, and G. Shi, Carbon 64, 225 (2013).
Z. Fan, K. Wang, T. Wei, et al., Carbon 48, 1686 (2010).
D. Xu, Y. Zhang, D. Zhang, and S. Yang, Cryst. Eng. Commun. 17, 1106 (2015).
I. Ilyas, I. Bashir, and M. A. Farrukh, Russ. J. Phys. Chem. A 95, 1023 (2021).
M. Ahmadi, M. S. Seyed Dorraji, M. H. Rasoulifard, and A. R. Amani-Ghadim, Sep. Purif. Technol. 228, 1 (2019).
A. R. Rajan, A. Rajan, A. John, et al., Ceram. Int. 45, 21947 (2019).
A. Ishtiaq, M. A. Farrukh, A. U. Rehman, et al., J. Chin. Chem. Soc. 69, 269 (2022).
ACKNOWLEDGMENTS
The authors are grateful to FCCU for financial support to complete this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare that they have no conflicts of interest.
DECLARATION OF INTERESTS
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Rights and permissions
About this article
Cite this article
Akbar, S., Farrukh, M.A. & Chong, KK. Facile Synthesis of Gd2O3/rGO Nanocomposite: Optical, Thermal, Morphological, Structural, and Catalytic Studies. Russ. J. Phys. Chem. 97, 722–734 (2023). https://doi.org/10.1134/S0036024423040283
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024423040283