Abstract
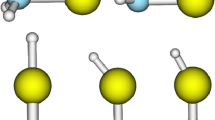
The optimal geometries of (CaO)n and (MgO)n clusters at n = 2–30 have been found and the enthalpies of formation of 1D, 2D, and 3D structures have been determined using quantum-chemical DFT calculations. The calculation demonstrates that the formation of linear chains of Ca and Mg oxides practically does not occur, while the formation of two-dimensional (tiled) and three-dimensional (cubic) structures proceeds with a large release of energy. The competing process of formation of molecular rods consisting of planar six-membered rings (MO)3 has been considered, and it has been shown to proceed not through the stage of preliminary formation of six-membered rings, but directly from monomer units.




Similar content being viewed by others
REFERENCES
J. A. Edmonds, P. Freund, and J. J. Dooley, Greenhouse Gas Control Technologies (2001).
J. P. Mojica-Sánchez, T. I. Zarate-López, J. M. Flores-Álvarez, et al., Phys. Chem. Chem. Phys. 21, 23102 (2019). https://doi.org/10.1039/c9cp05075b
I. K. Garkushin, O. V. Lavrent’eva, and A. M. Shterenberg, Fiz. Khim. Stekla 49, 148 (2023) https://doi.org/10.31857/S0132665122100109
Gu Guoxuan, Li Sheng, Liu **n, et al., Ceram. Silikáty 66, 480 (2022). https://doi.org/10.13168/cs.2022.0044
S. Utamapanya, K. J. Klabunde, and J. R. Schlup, Chem. Mater. 3, 175 (1991).
G. B. Sergeev, Nanochemistry (Izd-vo MGU, Moscow, 2003) [in Russian].
I. P. Suzdalev, Nanotechnology: Physical Chemistry of Nanoclusters, Nanostructures and Nanomaterials (KomKniga, Moscow, 2006) [in Russian].
N. H. Ray, Inorganic Polymers (Acad. Press, London, 1978).
D. S. Sanditov and G. M. Bartenev, Physical Properties of Disordered Structures (Nauka, Novosibirsk, 1982) [in Russian].
A. Fel’ts, Amorphous and Glassy Inorganic Solids (Mir, Moscow, 1986) [in Russian].
R. C. Ropp, Inorganic Polymer Glasses (Elsevier, Amsterdam, 1992).
O. S. Sirotkin, Doctoral Sci. (Eng.) Diss., Kazan’, 1992.
A. Yu. Shaulov, L. V. Vladimirov, A. V. Grachev, et al., Russ. J. Phys. Chem. B 14, 183 (2020). https://doi.org/10.1134/S1990793120010157
M.-J. Malliavin and C. Coudray, J. Chem. Phys. 106, 2323 (1997).
A. Aguado, F. López-Gejo, and J. M. López, J. Chem. Phys. 110, 4788 (1999).
M. Gutowski, P. Skurski, X. Li, et al., Phys. Rev. Lett. 85, 3145 (2000).
R. Dong, X. Chen, X. Wang, et al., J. Chem. Phys. 129, 044705 (2008).
M. L. S. Vasili and D. Felle, et al., J. Phys. Chem. A 114, 9349 (2010).
K. Kwapien, M. Sierka, J. Do?bler, et al., Angew. Chem., Int. Ed. 50, 1716 (2011).
M. Haertelt, A. Fielicke, G. Meijer, et al., Phys. Chem. Phys. 14, 2849 (2012).
L. Hong, H. Wang, J. Cheng, et al., Comput. Theor. Chem. 980, 62 (2012).
Priynka Batra, Ritu Gaba, Upasana Issar, et al., J. Theor. Chem., 720794 (2013). https://doi.org/10.1155/2013/720794
Y. Zhang, H. S. Chen, Y. H. Yin, et al., J. Phys. B: At., Mol. Opt. Phys. 47, 025102 (2014).
FelmyA. R. Mingyang and D. A. Dixon, J. Phys. Chem. A 118, 3136 (2014). https://doi.org/10.1021/jp412820z
Chen Mingyang, Sahan Kanchana Thanthiriwatte, and A. David, J. Phys. Chem. C 121, 23025 (2017). https://doi.org/10.1021/acs.jpcc.7b09062
Motoyoshi Nakano, Daiki Hebiguchi, Shohei Azuma, et al., J. Phys. Chem. A 124, 101 (2020).
E. K. U. Gross and W. Kohn, Adv. Quantum Chem. 21, 255 (1990).
A. D. Becke, J. Chem. Phys. 98, 5648 (1993).
A. Schafer, H. Horn, and R. Ahlrichs, J. Chem. Phys. 97, 2571 (1992).
F. Furche, R. Ahlrichs, C. Hattig, et al., Comput. Mol. Sci. 4, 91 (2014).
A. F. Wells, Structural Inorganic Chemistry, 3rd ed. (Oxford University Press, Clarendon Press, 1962).
M. Vasiliu, D. Feller, J. L. Gole, and D. A. Dixon, J. Phys. Chem. A 114, 9349 (2010).
F. Bawa and I. Panas, Phys. Chem. Chem. Phys. 4, 103 (2002).
Funding
The work was carried out within the framework of the State assignment of the Ministry of Science and Education of the Russian Federation (subject FFZE-2022-0010, GZ no. 122040400099-5, AAAA-A20-120030590042-8).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Additional information
Translated by G. Kirakosyan
Rights and permissions
About this article
Cite this article
Sakovich, R.A., Shaulov, A.Y. Initial Stages of the Formation of the Supramolecular Structure of Ca and Mg Oxides. Russ. J. Inorg. Chem. 68, 1013–1018 (2023). https://doi.org/10.1134/S0036023623601265
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023623601265