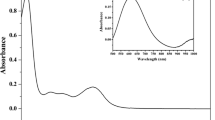
Abstract
Using a computer modeling approach, we proposed a structure for a potential GC-specific DNA ligand, which could form a complex with DNA in the minor groove similar to that formed by Hoechst 33258 at DNA AT-enriched sites. According to this model, MBoz2A, a bisbenzoxazole ligand, was synthesized. The results of spectrophotometric methods supported the complex formation of the compound under study with DNA differing in the nucleotide composition.






Similar content being viewed by others
REFERENCES
Geierstanger B.H., Wemmer D.E. 1995. Complexes of the minor groove of DNA. Annu. Rev. Biophys. Biomol. Struct. 24, 463‒493.
Krey A.K., Hahn F.E. 1970. Studies on the complex of distamycin A with calf thymus DNA. FEBS Lett. 10, 175‒178.
Harshman K.D., Dervan P.B. 1985. Molecular recognition of B-DNA by Hoechst 33258. Nucl. Acids Res. 13, 4825‒48354.
Dervan P.B. 2001. Molecular recognition of DNA by small molecules. Bioorg. Med. Chem. 9, 2215‒2235.
Dervan P.B., Buerli R.W. 1999. Sequence-specific DNA recognition by polyamides. Curr. Opin. Chem. B-iol. 3, 688‒693.
Wemmer D.E., Dervan P.B. 1997. Targeting the minor groove of DNA. Curr. Opin. Struct. Biol. 7, 355‒361.
Dervan P.B., Doss R.M., Marques M.A. 2005. Programmable DNA binding oligomers for control of transcription. Curr. Med. Chem.—Anti-Cancer Agents. 5, 373‒387.
Guo P., Paul A., Kumar A., Farahat A.A., Kumar D., Wang S., Boykin D.W., Wilson D.W. 2016. The thiophene “sigma-hole” as a concept for preorganized, specific recognition of G.C base pairs in the DNA minor groove. Chem. Eur. J. 22, 15404–15412.
Guo P., Farahat A.A., Paul A., Hanka N.K., Boykin D.V., Wilson W.D. 2018. Compound shape effects in minor groove binding affinity and specificity for mixed sequence DNA. J. Am. Chem. Soc. 140, 14761‒14769.
Wilson W.D., Paul A. 2022. Compound shape and substituent effects in DNA minor groove interactions. In Handbook of Chemical Biology of Nucleic Acids. Sugimoto N., Ed. Springer Nature Singapore Pte Ltd., p. 1‒39.
Paul A., Nanjunda R., Wilson W.D. 2023. Binding to the DNA minor groove by heterocyclic dications from AT specific to GC recognition compounds. Curr. Protocols. 3 (4), e729.
O’Boyle N.M., Banck M., Craig A.J., Morley C., Vandermeersch T., Hutchison G.R. 2011. Open Babel: An open chemical toolbox. J. Cheminf. 3, 1‒14.
Korb O., Stützle T., Exner T.E. 2007. An ant colony optimization approach to flexible protein–ligand docking. Swarm Intelligence. 1, 115134.
Teng M.K., Usman N., Frederick C.A., Wang A.H. 1988. The molecular structure of the complex of Hoechst 33258 and the DNA dodecamer d (CGCGAATTCGCG. Nucleic Acids Res. 16, 2671‒2690.
Finlay A.C., Hochstein F.A., Sobin B.A., Murphy F.X. 1951. Netropsin, a new antibiotic produced by a Streptomyces. J. Am. Chem. Soc. 73, 341‒344.
Zasedatelev A.S., Borodulin V.B., Grokhovsky S.L., Nikitin A.M., Salmanova D.V., Zhuze A.L., Gursky G.V., Shafer R.H. 1995. Mono-, di- and trimeric binding of a bis-netropsin to DNA. FEBS Lett. 375, 304‒306.
Makarska-Bialokoz M. 2014. Fluorescence quenching effect of guanine interacting with water-soluble cationic porphyrin. J. Lumin. 47, 27‒33.
Funding
This work was supported by the Russian Science Foundation, project no. 23-24-00082.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors of this work declare that they have no conflicts of interest.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This work does not contain any studies involving human and animal subjects.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Modifications have been made to the author’s name and Fig. 1. Full information regarding the corrections made can be found in the erratum/correction for this article.
Rights and permissions
About this article
Cite this article
Arutyunyan, A.F., Aksenova, M.S., Kostyukov, A.A. et al. Synthesis of a Bisbenzoxazole Analogue of Hoechst 33258 as a Potential GC-Selective DNA Ligand. Mol Biol 58, 547–556 (2024). https://doi.org/10.1134/S0026893324700171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893324700171