Abstract
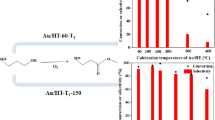
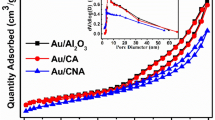
Synthesis of glyoxalic acid by selective oxidation of glyoxal at ambient temperatures with O2 as an oxidant is an important problem. We found that gold nanoparticles supported on hydrotalcite (Au/HT) exhibit an appreciable catalytic activity for this reaction in the liquid phase. Moreover, Au-Pd/HT, prepared by the deposition-precipitation method is superior in the catalytic behavior to monometallic Au/HT and Pd/HT catalysts. Introduction of palladium enhances ability of the catalysts to oxidize carbonyl to carboxyl, weakens the power to rupture C-C bond and in this way improves the catalytic performance. Furthermore, the Au: Pd ratio also influences the properties of the alloy catalysts. The 1.5Au-1.5Pd/HT catalysts show the highest activity for the selective oxidation at ambient temperature producing glyoxalic acid in 13.4% yield at pH 7.7. Moreover, due to basic properties of hydrotalcite, glyoxalic acid could be synthesized over 1.5Au-1.5Pd/HT in 8.0% yield without adding a base. It is hoped that results of this study can fuel further research in designing new catalysts with alloy nanoparticles supported by hydrotalcite that can be used for the selective oxidation of other useful compounds.
Similar content being viewed by others
References
Sun, Z., Eli, W., Xu, T., and Zhang, Y., Ind. Eng. Chem. Res., 2006, vol. 45, p. 1849.
Zhang, N. Chen, X., Cao, G., and Yuan, W., React. Kinet. Mech. Catal., 2010, vol. 99, p. 325.
Yadav. G. and Gupta, V., Process, Biochem., 2000, vol. 36, p. 73.
Alardin, F., Ruiz, P., Delmon, B., and Devillers, M., Appl. Catal., A, 2001, vol. 215, p. 125.
Alardin, F., Delmon, B., Ruiz, P., and Devillers, M., Catal. Today, 2000, vol. 61, p. 255.
Mattioda, G. and Christidis, Y., Ullmann’s Encyclopedia of Industrial Chemistry, New York: Wiley, 1989, p. 495.
Niu, Y. and Li, R., Sci. China., Ser. B: Chem., 2009, vol. 52, p. 1057.
Niu, Y., Xu, Z., Li, M., and Li, R., Chin. Chem. Lett., 2008, vol. 19, p. 245.
Hermans, S., Deffernez, A., and Devillers, M., Appl. Catal., A, 2011, vol. 395, p. 19.
Gallezot, P., Mesanstourne, R., Christidis, Y., Mattioda, G., and Schouteeten, A., J. Catal., 1992, vol. 133, p. 479.
Gallezot, P., de Mesanstourne, R., Christidist, Y., and Mattioda, G., J. Catal., 1992, vol. 133, p. 479.
Deffernez, A., Hermans, S., and Devillers, M., Appl. Catal., A, 2005, vol. 282, p. 303.
Alardin, F., Wullens, H., Hermans, S., and Devillers, M., J. Mol. Catal. A: Chem., 2005, vol. 225, p. 79.
Manolis, S. and Hermenegildo, G., Chem. Rev., 2012, vol. 112, p. 4469.
Cavani, F., Trifir, F., and Vaccari, A., Catal. Today, 1991, vol. 11, p. 173.
Akifumi, N., Tomoo, M., Koichiro, J., and Kiyotomi, K., Green Chem., 2009, vol. 11, p. 793.
Orthman, J., Zhu, H., and Lu, G., Sep. Purif. Technol., 2003, vol. 31, p. 53.
Kushwaha, P., Srivastava, V., Bhatt, S., and Jasra, R., Ind. Eng. Chem. Res., 2007, vol. 46, p. 4856.
Hermans, S. and Devillers, M., Catal. Lett., 2005, vol. 99, p. 55.
Bolognini, M., Cavania, F., Scagliarini, D., Flego, C., Perego, C., and Saba, M., Catal. Today, 2002, vol. 75, p. 103.
Castro, C., Cardoso, D., Nascente, P., and Assaf, J., Catal. Lett., 2011, vol. 141, p. 1316.
Wang, D., Alberto, V., Francesca, P., Laura, P., and Su, D., J. Phys. Chem. C, 2008, vol. 112, p. 8617.
Zhu, Y., Chen, X., Zheng, Z., Ke, X., Esa, J., Zhao, J., Guo, C., **e, T., and Wang, D., Chem. Commun., 2009, vol. 48, p. 7524.
Tuzovskaya, I., Bogdanchikova, N., Simakov, A., Gurin, V., Pestryakov, A., Avalos, M., and Farias, M., Chem. Phys., 2007, vol. 338, p. 23.
Boccuzzi, F., Cerrato, G., Pinna, F., and Strukul, G., J. Phys. Chem. B, 1998, vol. 102, p. 5733.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Jia, M.L., Liu, C.X., Wang, J. et al. Catalytic oxidation of glyoxal to glyoxalic acid over Au-Pd alloy nanoparticles on hydrotalcite. Kinet Catal 55, 671–675 (2014). https://doi.org/10.1134/S0023158414050061
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158414050061