Abstract
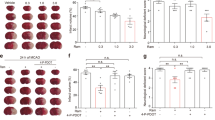
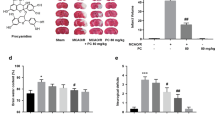
Thrombosis and stroke are major causes of disability and death worldwide. However, the regular antithrombotic drugs may have unsatisfactory results and side effects. Platonin, a cyanine photosensitizing dye, has been used to treat trauma, ulcers and some acute inflammation. Here, we explored the neuroprotective effects of platonin against middle cerebral artery occlusion (MCAO)-induced ischemic stroke in mice. Platonin(200 μg/kg) substantially reduced cerebral infarct volume, brain edema, neuronal cell death and neurological deficit scores, and improved the MCAO-reduced locomotor activity and rotarod performance. Platonin(5–10 μM) potently inhibited platelet aggregation and c-Jun NH2-terminal kinase (JNK) phosphorylation in collagen-activated platelets. The antiaggregation effect did not affect bleeding time but increased occlusion time in platonin(100 and 200 μg/kg)-treated mice. Platonin(2–10 μM) was potent in diminishing collagen- and Fenton reaction-induced ∙OH formation. Platonin(5–10 μM) also suppressed the expression of nitric oxide, inducible nitric oxide synthase, cyclooxygenase-2, interleukin-1β, and JNK phosphorylation in lipopolysaccharide-stimulated macrophages. MCAO-induced expression of 3-nitrotyrosine and Iba1 was apparently attenuated in platonin(200 μg/kg)-treated mice. In conclusion, platonin exhibited remarkable neuroprotective properties against MCAO-induced ischemia in a mouse model through its antiaggregation, antiinflammatory and antiradical properties. The observed therapeutic efficacy of platonin may consider being a novel medcine against ischemic stroke.
Similar content being viewed by others
Introduction
Stroke is the third leading cause of death and the most frequent cause of permanent disability worldwide1, and inflammation appears to be critical in the pathogenesis of ischemic stroke and other forms of ischemic brain injuries. The inflammatory response has a detrimental role in cerebral ischemia/reperfusion (I/R) injury pathogenesis2. The association between inflammation and cerebral I/R outcomes has ensured considerable and continued interest in the development of antiinflammation-oriented therapies for mitigating I/R-induced brain damage. In the brain, microglia and monocyte-derived macrophages are the key players in the immune response after stroke47. The platelet suspensions (3.6 × 108 cells/ml) were treated with 1 μg/ml collagen and 2 μM platonin or PBS for 3 min in a separate vial. The suspensions were incubated for 5 min and 100 μM DMPO was added before the ESR analysis was conducted. The ESR spectrometer was operated at a 20-mW power, 9.78-GHz frequency, 100-G scan range, and receiver gain of 5 × 104. Moreover, a Fenton reaction solution (50 μM FeSO4 and 2 mM H2O2) was pretreated with a solvent control (PBS) or platonin (5 and 10 μM) and vitamin C (100 μM) for 3 min.
Cell culture and platonin treatment
The BV2 and RAW 264.7 cell lines were maintained in DMEM supplemented with 10% heat-inactivated FBS, penicillin G (100 units/ml), streptomycin (100 mg/ml), and L-glutamine (2 mM) and incubated at 37 °C in a humidified atmosphere containing 5% CO2. For platonin treatment, platonin was dissolved in PBS. The cells were pretreated with 5 or 10 μM platonin for 30 min and then stimulated with or without LPS (1 μg/ml) for the indicated time.
Measurement of NO concentration
The nitrite concentration was determined using the Griess reagent (1% sulfanilamide and 0.1% naphthalenediamine in 2.5% phosphoric acid) as an indicator of NO production48. In brief, cells were plated at 8 × 105 cells/well into a 6-cm dish in DMEM with 10% FBS. The cells were pretreated with platonin (5–10 μM) for 30 min and then stimulated with or without LPS (1 μg/ml) for 24 h. After incubation, 150 μl of the culture supernatants was mixed with 150 μl of the Griess reagent. The absorbance of the mixtures was measured at 550 nm on a microplate reader.
Western blotting
To determine the level of COX-2, IL-1β, iNOS, and JNK phosphorylation in the whole cell lysate, protein samples were separated through 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to a PVDF membrane by using a Bio-Rad semidry transfer unit (Hercules, CA, USA). The membranes were then blocked with TBST (10 mM Tris-base, 100 mM NaCl, and 0.01% Tween 20) containing 5% BSA for 1 h at room temperature and probed with the various specific primary antibodies. After several washes, the membranes were incubated with the HRP-linked secondary antibody (1:3000 in TBST) for 1 h. Immunoreactive bands were detected using an enhanced chemiluminescence system. The ratios of the semiquantitative results were obtained by scanning the reactive bands and quantifying the optical densities on a videodensitometer and Bio-profil Biolight software (version V2000.01; Vilber Lourmat, Marne-la-Vallée, France).
Statistical analysis
Data are expressed as the means ± SEM, accompanied by the number of observations. The normality of the data was first tested using the Kolmogorov–Smirnov test. The continuous variables were compared using analysis of variance. When the analysis indicated significant differences among group means, each group was compared using the Newman–Keuls method. P < 0.05 was considered statistically significant.
Additional Information
How to cite this article: Sheu, J.-R. et al. A novel indication of platonin, a therapeutic immunomodulating medicine, on neuroprotection against ischemic stroke in mice. Sci. Rep. 7, 42277; doi: 10.1038/srep42277 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
06 April 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Donnan, G. A., Fisher, M., Macleod, M. & Davis, S. M. Stroke. Lancet 371, 1612–1623 (2008).
Qiao, H. et al. Luteolin downregulates TLR4, TLR5, NF-κB and p-p38MAPK expression, upregulates the p-ERK expression, and protects rat brains against focal ischemia. Brain Res 1448, 71–81 (2012).
**ong, X. Y., Liu, L. & Yang, Q. W. Functions and mechanisms of microglia/macrophages in neuroinflammation and neurogenesis after stroke. Prog Neurobiol 142, 23–44 (2016).
Semenza, G. L. Hypoxia-inducible factors in physiology and medicine. Cell 148, 399–408 (2012).
Laveti, D. et al. Anti-inflammatory treatments for chronic diseases: a review. Inflamm Allergy Drug Targets 12, 349–361 (2013).
Murakami, A. & Ohigashi, H. Targeting NOX, INOS and COX-2 in inflammatory cells: chemoprevention using food phytochemicals. Int J Cancer 121, 2357–2363 (2007).
Kaminska, B. MAPK signaling pathways as molecular targets for anti-inflammatory therapy from molecular mechanisms to therapeutic benefits. Biochim Biophys Acta 1754, 253–262 (2005).
Wong, C. H. & Crack, P. J. Modulation of neuro-inflammation and vascular response by oxidative stress following cerebral ischemia reperfusion injury. Curr Med Chem 15, 1–14 (2008).
Costello, M. J. et al. A census of marine biodiversity knowledge, resources, and future challenges. PLoS One 5, e12110 (2010).
Lopes, R. D. Antiplatelet agents in cardiovascular disease. J Thromb Thrombol 31, 306–309 (2011).
Nutescu, E. A., Shapiro, N. L., Chevalier, A. & Amin, A. N. A pharmacological overview of current and emerging anticoagulants. Clev Clin J Med 72, 2–6 (2005).
Motoyoshi, F. et al. The effect of photosensitive dye platonin on juvenile rheumatoid arthritis. Biotherapy 3, 241 (1991).
Hsiao, G. et al. Platonin, a photosensitizing dye, improves circulatory failure and mortality in rat models of endotoxemia. Biol Pharm Bull 25, 995–999 (2002).
Lee, J. J., Huang, W. T., Shao, D. Z., Liao, J. F. & Lin, M. T. Platonin, a cyanine photosensitizing dye, inhibits pyrogen release and results in antipyresis. J Pharmacol Sci 93, 376–380 (2003).
Chen, C. C. et al. Platonin attenuates LPS-induced CAT-2 and CAT-2B induction in stimulated murine macrophages. Acta Anaesthesiol Scand 50, 604–612 (2006).
Yeh, C. T., Kao, M. C., Chen, C. H. & Huang, C. J. Platonin preserves blood-brain barrier integrity in septic rats. Acta Anaesthesiol Taiwan 53, 12–15 (2015).
Huet, O. et al. Plasma-induced endothelial oxidative stress is related to the severity of septic shock. Crit Care Med 35, 821–826 (2007).
Lo, Y. Y., Wong, J. M. & Cruz, T. F. Reactive oxygen species mediate cytokine activation of c-Jun NH2-terminal kinases. J Biol Chem 271, 15703–15707 (1996)
Crack, P. J. & Taylor, J. M. Reactive oxygen species and the modulation of stroke. Free Radic Biol Med 38, 1433–1444 (2005).
Demougeot, C. et al. Cytoprotective efficacy and mechanisms of the liposoluble iron chelator 2, 2-dipyridyl in the rat photothrombotic ischemic stroke model. J Pharmacol Exp Ther 311, 1080–1087 (2004).
Gursoy-Ozdemir, Y., Bolay, H., Saribas, O. & Dalkara, T. Role of endothelial nitric oxide generation and peroxynitrite formation in reperfusion injury after focal cerebral ischemia. Stroke 31, 1974–1980 (2000).
Tominaga, T. & Ohnishi, S. T. Interrelationship of brain edema, motor deficits, and memory impairment in rats exposed to focal ischemia. Stroke 20, 513–518 (1989).
Baigent, C. et al. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomized trials. Lancet 373, 1849–1860 (2009).
Furie, K. L. et al. American Heart Association Stroke Council, Council on Cardiovascular Nursing, Council on Clinical Cardiology, and Interdisciplinary Council on Quality of Care and Outcomes Research. Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 42, 227–276 (2011).
Johnson, S. Known knowns and known unknowns: Risks associated with combination antithrombotic therapy. Thromb Res 123, S7–S11 (2008).
Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomized trials of antiplatelet therapy for prevention of death, myocardial infarction and stroke in high risk patients. BMJ 324, 71–86 (2002).
Helft, G., Gilard, M., Feuvre, Le. C & Zaman, A. G. Drug insight: antithrombotic therapy after percutaneous coronary intervention in patients with an indication for anticoagulation. Nat Clin Pract Cardiovasc Med 3, 673–680 (2006).
Colman, R. W., Clowes, A. W. & George J. N. Overview of hemostasis. In: Colman, R. W. et al. (Eds.), Hemostasis and Thrombosis, 5th ed. Lippincott Williams & Wilkins, Philadelphia, pp. 3–16 (2006).
Lin, K. H. et al. Hinokitiol inhibits platelet activation ex vivo and thrombus formation in vivo . Biochem Pharmacol 85, 1478–1485 (2013).
Brambilla, R., Couch, Y. & Lambertsen, K. L. The effect of stroke on immune function. Mol Cell Neurosci 53, 26–33 (2013).
Lambertsen, K. L., Biber, K. & Finsen, B. Inflammatory cytokines in experimental and human stroke. J Cereb Blood Flow Metab 32, 1677–1698 (2012).
Lee, S. H. et al. Selective expression of mitogen-inducible cyclooxygenase in macrophages stimulated with lipopolysaccharide. J Biol Chem 267, 25934–25938 (1992).
Yenari, M. A., Xu, L., Tang, X. N., Qiao, Y. & Giffard, R. G. Microglia potentiates damage to blood-brain barrier constituents: improvement by minocycline in vivo and in vitro . Stroke 37, 1087–1093 (2006).
Hayashi, T. et al. c-Jun N-terminal kinase (JNK) and JNK interacting protein response in rat brain after transient middle cerebral artery occlusion. Neurosci Lett 284, 195–199 (2000).
Wallace, B. K., Jelks, K. A. & O’Donnell, M. E. Ischemia-induced stimulation of cerebral microvascular endothelial cell Na-K-Cl cotransport involves p38 and JNK MAP kinases. Am J Physiol Cell Physiol 302, 505–517 (2012).
Chang, Y. et al. Platonin inhibited PDGF-BB-induced proliferation of rat vascular smooth muscle cells via JNK1/2-dependent signaling. Acta Pharmacol Sin 32, 1337–1344 (2011).
Uto, T., Fujii, M. & Hou, D. X. 6-(Methylsulfinyl) hexyl isothiocyanate suppresses inducible nitric oxide synthase expression through the inhibition of Janus kinase 2-mediated JNK pathway in lipopolysaccharide-activated murine macrophages. Biochem Pharmacol 70, 1211–1221 (2005).
Gilgun-Sherki, Y., Rosenbaum, Z., Melamed, E. & Offen, D. Antioxidant therapy in acute central nervous system injury: current state. Pharmacol Rev 54, 271–284 (2002).
Oliver, C. N. et al. Oxidative damage to brain proteins, loss of glutamine synthetase activity, and production of free radicals during ischemia/reperfusion induced injury to gerbil brain. Proc Natl Acad Sci USA 87, 5144–5147 (1990).
Santabárbara-Ruiz, P. et al. ROS-Induced JNK and p38 Signaling Is Required for Unpaired Cytokine Activation during Drosophila Regeneration. PLoS Genet 11, e1005595 (2015).
Ilmarinen, P., Moilanen, E. & Kankaanranta, H. Mitochondria in the center of human eosinophil apoptosis and survival. Int J Mol Sci 15, 3952–3969 (2014).
Lei, S. et al. Effects of N-acetylcysteine on nicotinamide dinucleotide phosphate oxidase activation and antioxidant status in heart, lung, liver and kidney in streptozotocin-induced diabetic rats. Yonsei Med J 53, 294–303 (2012).
Fiordaliso, F. et al. Antioxidant treatment attenuates hyperglycemia-induced cardiomyocyte death in rats. J Mol Cell Cardiol 37, 959–968 (2004).
Yen, T. L. et al. Andrographolide stimulates p38 mitogen-activated protein kinase-nuclear factor erythroid-2-related factor 2-heme oxygenase 1 signaling in primary cerebral endothelial cells for definite protection against ischemic stroke in rats. Transl Res 170, 57–72 (2016).
Chen, J. et al. Therapeutic benefit of intravenous administration of bone marrow stromal cells after cerebral ischemia in rats. Stroke 32, 1005–1011 (2001).
Lu, W. J. et al. Prevention of arterial thrombosis by nobiletin: in vitro and in vivo studies. J Nutr Biochem 28, 1–8 (2016).
Chou, D. S. et al. ESR spin trap** of a carbon-centered free radical from agonist-stimulated human platelets. Free Radic Biol Med 39, 237–248 (2005).
Sun, J., Zhang, X., Broderick, M. & Fein, H. Measurement of nitric oxide production in biological systems by using Griess reaction assay. Sensors 3, 276–284 (2003).
Acknowledgements
This work was supported by grants provided by the Ministry of Science and Technology of Taiwan (NSC100-2320-B-038-021-MY3, MOST103-2320-B-038-017 and MOST103-2320-B-038-026-MY2), Taipei Medical University (102-AE1-B19), and the Chi-Mei Medical Center-Taipei Medical University (103CM-TMU-05).
Author information
Authors and Affiliations
Contributions
J.R.S., Z.C.C., C.H.Y. and C.Y.H. conceived and designed the experiments. D.S.C, T.L.Y., H.N.L., S.H.P. and C.H.H. performed the experiments and analyzed the data. T.J. and C.Y.H. wrote the manuscript. All authors discussed, edited and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sheu, JR., Chen, ZC., Jayakumar, T. et al. A novel indication of platonin, a therapeutic immunomodulating medicine, on neuroprotection against ischemic stroke in mice. Sci Rep 7, 42277 (2017). https://doi.org/10.1038/srep42277
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep42277
- Springer Nature Limited
This article is cited by
-
NLRP3 inflammasome as a potential treatment in ischemic stroke concomitant with diabetes
Journal of Neuroinflammation (2019)
-
HDAC6 dysfunction contributes to impaired maturation of adult neurogenesis in vivo: vital role on functional recovery after ischemic stroke
Journal of Biomedical Science (2019)