Abstract
Psoriatic arthritis (PsA) is associated with psoriasis, featured by its irreversible joint symptoms. Despite the significant impact on the healthcare system, it is still challenging to leverage machine learning or statistical models to predict PsA and its progression, or analyze drug efficacy. With 3961 patients’ clinical records, we developed a machine learning model for PsA diagnosis and analysis of PsA progression risk, respectively. Furthermore, general additive models (GAMs) and the Kaplan–Meier (KM) method were applied to analyze the efficacy of various drugs on psoriasis treatment and inhibiting PsA progression. The independent experiment on the PsA prediction model demonstrates outstanding prediction performance with an AUC score of 0.87 and an AUPR score of 0.89, and the Jackknife validation test on the PsA progression prediction model also suggests the superior performance with an AUC score of 0.80 and an AUPR score of 0.83, respectively. We also identified that interleukin-17 inhibitors were the more effective drug for severe psoriasis compared to other drugs, and methotrexate had a lower effect in inhibiting PsA progression. The results demonstrate that machine learning and statistical approaches enable accurate early prediction of PsA and its progression, and analysis of drug efficacy.
Similar content being viewed by others
Introduction
Psoriasis is a chronic and incurable inflammatory disease1 with different clinical subtypes, such as psoriasis vulgaris, erythrodermic psoriasis, and pustular psoriasis2. Around 25.0% of psoriasis patients will develop psoriatic arthritis (PsA), which is a chronic inflammatory joint disease associated with psoriasis and is featured by its irreversible joint symptoms in addition to skin lesions1,3,4,5. However, psoriasis can develop many other musculoskeletal symptoms that are unrelated to PsA, including osteoporosis, gout, fibromyalgia, soft tissue rheumatism, osteoarthritis, etc., leading to inevitable misdiagnosis or missed diagnosis6. It is estimated that up to 24.6% of PsA patients suffer from moderate to severe symptoms7. The clinical manifestations of PsA are diverse and can invade peripheral joints of the limbs, the trunk joints, and the attachment points of bones, tendons, and even muscles. Such invasions often lead to different degrees of swelling, pain, and discomfort in the axial joint or limb joint6, bringing great pain and posing a heavy disease burden to the patient’s life. While the early damage to the joints often appears as joint synovitis and progressive cartilage8, the damage may progress to irreversible bone destruction in the middle and late stages, eventually leading to disability4. In addition to the similar skin lesions as psoriasis causes, PsA patients are also at greater risk of cardiovascular disease9, cardiometabolic syndrome10, type 2 diabetes11, and liver disease12 than healthy individuals. The chronic painful muscle and joint stimulation can also lead to increased anxiety and even depression in PsA patients13.
Early diagnosis and control of PsA are therefore critically important2. At **stone for the early diagnosis and treatment of PsA and other types of psoriasis.
Results
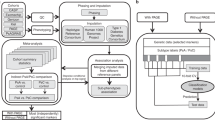
Overview of the patient cohort and study design
We constructed and assembled a large-scale cohort dataset with 104 clinical features of 3961 patients admitted to ** validation test and the predictive results of the new PsA patients’ data. Each star point represents a PsA patient, and its corresponding abscissa value means its predictive probability. BloodRT blood routine test. UrineRT urine routine test. WHR waist-hip ratio.
To further assess the proposed machine learning model, we conducted a prospective study based on six new PsA patients’ clinical data we collected. The six patients all developed from other types of psoriasis to PsA in a short period. We first obtained the predictive probability distribution using the 132 patients’ clinical data based on the bootstrap** validation strategy. The 95% CI of the predictive probability distribution of the positive samples is 0.465–0.701. Then, we tested the six PsA patients’ clinical data on the proposed predictive model and found that five of six (83.3%) predictive probabilities are in 0.465–0.701 (Fig. 4f), suggesting that the proposed model has the potential to perform an accurate risk prediction.
Drug efficacy analysis of psoriasis
We first applied statistical approaches to explore the drug effects on psoriasis treatment in terms of PASI. Six types of drugs are covered in our dataset for psoriasis treatment, including MTX, topical corticosteroid (TCS), acitretin, tumor necrosis factor alpha (TNF-α) inhibitors, interleukin-17 (IL-17) inhibitors, and interleukin-23 (IL-23) inhibitors. The time-dependent log(PASI + 1) for each drug is described in Supplementary Fig. 35. Verifying these time-dependent PASI changes by a Mann-Whitney U test37, we found that there was no significant difference in PASI changes between the patients with PsA and non-PsA, regardless of the drug categories (TCS: p-value = 0.66, IL-17 inhibitors: p-value = 0.43, MTX: p-value = 0.72, TNF-α inhibitors: p-value = 0.62). The statistical p-values of acitretin and IL-23 inhibitors were not calculated due to the lack of PASI data from PsA patients. We therefore focused on the effects of different drug types on the PASI changes within all patients, including PsA and non-PsA. We further split our data into three subgroups, including mild (PASI < 3), moderate (3 ≤ PASI < 10), and severe (PASI ≥ 10) (‘Methods’) according to the PASI values at the patients’ first visits and fitted the GAMs for the time-dependent log(PASI + 1) for each subgroup (Fig. 5a–c). We noticed that acitretin, MTX, and TCS have similar effects on the changes of PASI given their similar gradients of GAM curves; IL-17 inhibitors have a better effect on reducing PASI values compared with other types of drugs in the moderate and severe subgroups, meaning that IL-17 inhibitors are more effective in the treatment of more severe psoriasis. Then, we collected the medication information from 17 new patients (Supplementary Fig. 36), also indicating that the IL-17 inhibitor is a more effective drug than the others tested.
The logarithm of (PASI + 1) vs. time fitting curves of different drugs for those patients with mild psoriasis (PASI < 3) a, moderate psoriasis (3 ≤ PASI < 10) b, and severe psoriasis (PASI ≥ 10) c. The last subplots of a–c show the comparison of fitting curves of different drugs. d The Kaplan–Meier (KM) curve of the non-MTX drug group and MTX drug group. The p-value is obtained by the Log-Rank Test. TCS topical corticosteroid. MTX methotrexate. IL-17 inhibitors interleukin-17 inhibitors. TNF-α inhibitors tumor necrosis factor alpha inhibitors.
In addition, we split the patients into two groups (MTX and non-MTX) to further analyze MTX’s effect on PsA progression (Methods). We utilized the KM method to draw the curves of PsA progression for the two groups (HR: 0.139, 95% CI: 0.040–0.474, Log-Rank test p-value = 0.00024) (Fig. 5d) and further made a subgroup analysis stratified by sex using Cox regression (male, HR: 0.129, 95% CI: 0.028–0.581, Log-Rank test p-value = 0.0016; female, HR: 0.174, 95% CI: 0.021–1.464, Log-Rank test p-value = 0.0701), suggesting that there is statistical significance in PsA progression between MTX drugs and non-MTX drugs and that MTX drugs have a weaker ability to prevent this progression compared with other drugs, especially for males. Note that the drug efficacy analysis was based on the currently available data from the hospital. Prospective studies and more clinical data are therefore required to further validate these models and findings.