Abstract
The pandemic of Gompertz virus disease remains a pressing issue in agricultural production. Moreover, the dynamics of various infectious diseases is usually investigated by the method of mathematical modelling. A new mathematical model for dynamics on Gompertz virus disease impulsive system is proposed and analyzed in this paper. We prove the dynamic characteristics about the permanence and globally exponential stability of Gompertz virus disease model. Moreover, we also give the sufficient condition that one positive solution which satisfies \(E(t) \ge q_2\) if \(R_{1*}> 1\) exists. Eventually, numerical simulations are utilized to validate the validity of the theoretically analyzed conclusion in this paper.
Similar content being viewed by others
Introduction
It is well-known that a nonlinear system model usually has a rich dynamic behavior which has been payed attention to by many researchers1,2,3,4,5,6. Moreover, in recent years, many researchers have paid attention to biological control7,8,9,10,11,12,13,14,15,16.
Due to many cases that we need to describe the biological dynamics with sudden changes as well as other phenomena, impulsive differential equations play important part17,18,19. In consideration of hybrid nature, different kinds of evolutionary processes in real-world life are usually described by impulsive systems, and these evolutionary processes usually state a big sudden change at some moments20,21,22,23. Some researchers stated that differential equations with impulse do well in describing biological control, and thought that differential equations with impulsive effect are more practical than ordinary differential equations in stating practical problems24,25. Liang et al.26 proposed the impulsive control of Leslie predator-prey model, and investigated the dynamic properties of the proposed mathematical biology model. The herbivore-plankton model with cannibalism was proposed by Fang et al.27, and the corresponding heteroclinic bifurcations and three order-1 periodic orbits were analyzed. A new impulsive state feedback model was proposed, and the management strategy with impulsive control was developed28. Wang and Chen29 proposed a new microbial pesticide model and analyzed the condition of the existence of corresponding periodic orbit. Li and Chen30 investigated the dynamics of the impulsive turbidostat model, and also studied the characteristic of the proposed mathematical biology model. Li et al.31 proposed a new water hyacinth fish impulsive control model, and investigated the local stability and global dynamics of the proposed ecological system. Li et al.32 proposed a new Filippov predator-prey model, and analyzed the corresponding dynamics using the Filippov theory knowledge. Qin et al.33 proposed a new non-smooth Filippov ecosystem with group defense and also studied the stability of equilibria and the bifurcation phenomenon numerically. Discontinuous plant disease models with a non-smooth separation line were investigated and discussed by Li et al.34. Khan et al., proposed some new hepatitis B epidemic models and novel corona virus disease models and investigated their dynamic characteristics35,36,37,38,39,40.
In fact, exploiting viruses and the release of pest population simultaneously, some research studies have been attached to devote the control of pests9,41. First of all, introducing pathogens into the pest population is expected to produce an epidemic and then become endemic. Moreover, the infected pests are released to the periodic impulsive pest population. However, the crops will not be affected by the infected pest. The vulnerable pests are infected by direct contact with infected pests or being exposed to an infected environment. Then the pest population and its death will be affected to some extent42,43. In recent years, the pandemic of Gompertz virus disease remains a pressing issue in epidemiological ecosystem. Mathematical modelling is an important method in investigating the dynamic characteristics of epidemiological ecosystem. Additionally, due to the hybrid nature, in order to well describe different kinds of real-world evolutionary processes, impulsive systems play important role because it states sudden change at some moments. This article will try to develop a new epidemiological ecosystem with impulsive effect and investigate its dynamics for bio-control. Additionally, it is assumed that the pest population will grow according to the logic curve without the disease9,44,45. Then we investigate the global attractiveness and permanence of the proposed model.
The remainder of this paper is provided as follows. In section “Mathematical model”, the Gompertz virus disease model with impulsive effect is established. In section “Global attractiveness”, utilizing the comparison theorem and Floquet theory of impulsive differential equation, the global attractiveness of the proposed Gompertz virus disease model is strictly proved. In section “Uniform persistence”, we obtain the criteria for the uniform persistence of the proposed Gompertz virus disease model. In section “Numerical simulations”, many numerical results are given to prove the validity of the obtained results. Finally, some conclusions are summarized.
Mathematical model
In this section, we will present the newly developed Gompertz virus disease model with impulsive effect.
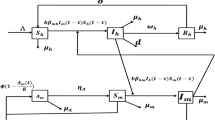
The total population of insect pest is denoted by N(t), and it is divided into five subgroups such as I(t), E(t), S(t), R(t) and A(t). The susceptible pest is denoted by S(t); E(t) denotes the exposed pest; the infected (symptomatic) pest is denoted by I(t); A(t) represents the asymptotically infected pest; the recovered or the removed pest is denoted by R(t). The class M(t) denotes the reservoir or the seafood place or market.
In this paper, the impulsive introduction of disinfectants and the pulse vaccination strategy in the environment are investigated for the control of the Gompertz virus epidemic, and we also assume that the proposed model satisfies:
-
1.
\(\mu\) and \(\Pi\) respectively denotes the natural death rate and the birth rate of the pest.
-
2.
The susceptible pest S(t) acquires Gompertz virus infection by ingesting environmental virus from the contaminated reservoir or seafood place or market, through the term given by \(\beta \frac{{S(t)}}{{K + S(t)}},\) where \(\beta\) is the disease transmission coefficient.
-
3.
The proportion of asymptomatic infection is denoted by \(\theta\). The transmission rate of the infected is denoted by \(\rho .\) \(\varpi\) denotes the transmission rate after completing the incubation period. K represents the pathogen concentration.
-
4.
\(\tau _1\) and \(\tau _2\) respectively denote the pest in I(t) and A(t) joining R(t) with the removal or recovery rate, and Gompertz virus induced mortality rate of infected individuals is denoted by \(d_1\).
-
5.
\(\eta M(t)E(t)\) denotes the susceptible pest infected after the interaction with M(t), in which the disease transmission coefficient between S(t) and E(t) is represented by \(\eta .\)
-
6.
The parameters \(\lambda _1\) and \(\lambda _2\) respectively denote the infected symptomatic and the asymptomatically infected contributing the Gompertz virus into the seafood market M(t). The removing rate of the Gompertz virus from the seafood market M(t) is given by the rate d. The natural growth rate of the Gompertz virus is denoted by b. We assume \(b < d\) through this paper.
-
7.
The parameter \(\theta _1(0< \theta _1<1)\) denotes the part of susceptible pest inoculated by the vaccine at times \(t=nT,\) in which n is a positive integer, and the impulsive period between two consecutive pulse vaccinations is denoted by T. nT represents the pulse vaccination at the time of multiple periods. R(t) represents the temporary immunity which contains the recovered individual and the vaccinated individual.
-
8.
The death of the Gompertz virus dues to the environmental sanitation. Therefore, considering the pulse inoculation of disinfectant in the environment at the time of nT, the death rate of the virus is denoted by \(\xi (0<\xi <1).\) The model is expressed as:
$$\begin{aligned} \left\{ {\begin{array}{lll} {\left. {\begin{array}{lll} {\begin{array}{lll} {\dot{S}(t) = \Pi - S(t)M(t) E(t)\eta -\frac{{S(t)\beta E(t)}}{{K+S(t)}} - \mu S(t) } \\ {\dot{E}(t) = \frac{{S(t)\beta E(t)}}{{K+S(t)}}-E(t)\mu - E(t)\theta \rho - E(t)(1-\theta )\varpi +\eta E(t)M(t)S(t)} \\ {\dot{I}(t) = (1 - \theta )\varpi E(t) - (\tau _1 + \mu + d_1 )I(t)} \\ \end{array}} \\ {\begin{array}{lll} {\dot{A}(t) = \theta \rho E(t) - (\tau _2 + \mu )A(t)} \\ {\dot{R}(t) = \tau _1 I(t) + \tau _2 A(t) - \mu R(t)} \\ {\dot{M}(t) = M(t)(b - d)+\lambda _1 I(t) + \lambda _2 A(t)} \\ \end{array}} \\ \end{array}} \right\} \;\;\;n \in N^ +, t \ne nT.} \\ {} \\ {\left. {\begin{array}{lll} {\begin{array}{lll} {S(t^ + ) = S(t)(1 - \theta _1 )} \\ {E(t^ + ) = E(t)} \\ {I(t^ + ) = I(t)} \\ \end{array}} \\ {\begin{array}{lll} {A(t^ + ) = A(t)} \\ {R(t^ + ) = \theta _1S(t)+R(t)} \\ {M(t^ + ) = M(t)(1 - \xi )} \\ \end{array}} \\ \end{array}} \right\} \;\;\; n \in N^ +, t=nT.} \\ \end{array}} \right. \end{aligned}$$(1)
For (1), all the coefficients are greater than 0. Based on
then the population size will change with t. If \(A(t) =I(t) =E(t) = 0\), we have \(\mathop {\lim }\limits _{t \rightarrow \infty } N(t) = \frac{\Pi }{\mu }.\)
Similarly, we have
Then we have that when t takes the sufficiently large value,
By replacing \(N(t)-S(t)-E(t)-I(t)-A(t)\) with R(t), then
We provide the initial condition for (3) as:
Next, we will study the dynamic properties of (3). For biological considerations, (3) is investigated in the following closed set and this set is given as
In fact, it is not difficult to check that \(\Omega\) has a positively invariant property about (3). For further discussion, some important assumptions and definitions are given as follows.
We assume that the fixed solution and the arbitrary solution of (3) are respectively given by
and
Definition 1
\(\forall\) \(x_0\), \(\exists\) \(M \ge m > 0,\) we have that when t takes the sufficiently large value, \(x(t)\in [m, M].\) Then (3) has the uniform persistence.
Definition 2
For arbitrary \(x_0\), If \(\mathop {\lim }\limits _{t \rightarrow \infty } \left| {x(t)-x^*(t)}\right| = 0\), then \(x^*(t)\) has the global attractivity.
Definition 3
\(\forall\) \(\eta >0,\) and \(\varepsilon >0,\) \(\left| {x^*(t)-x(t)} \right| < \varepsilon\) when there is \(\delta = \delta (t_0 ,\varepsilon ,\eta ) > 0.\) Moreover, if \(\left| {t_0-t} \right| > \eta\), and \(t\in [ t_0,+\infty ],\) then \(\left| {x_0 - x^*_0 } \right| < \delta .\) Then \(x^*(t)\) has the characteristic of stability.
Definition 4
When the fixed solution \(x^*(t)\) has the characteristic of stability and global attractivity, then \(x^*(t)\) is called globally asymptotical stable. If there is the disease-free periodic solution for (3) which also has the characteristic of globally asymptotical stability, the Gompertz virus will finally disappear.
Definition 5
(Equicontinuty) For \(\forall \varepsilon >0,\) if we can find a positive parameter \(\delta ,\) which satisfies \(|x(t_2)-x(t_1 )|<\varepsilon\) holds true when \(t_i(i=1,2)\in [0,l]\) and \(|t_1-t_2 |<\delta ,\) x(t) is said to be equicontinuous when \(0\le t\le 1.\)
For further analysis conveniently, the following lemma is given as follows.
Lemma 1
46 We consider
where \(0<\theta _1<1,\) \(\mu >0\) and \(\Pi >0.\)
Thus we can obtain that for \(t\in [nT,(n + 1)T],\) the periodic solution is given as
where
exists and is globally asymptotically stable, unique and positive.
Global attractiveness
In this section, we will discuss the global attractiveness of the Gompertz virus disease model with impulsive effect (3).
\(\forall\) \(t\in [0,+\infty ],\)
It means that the asymptotically infectious individuals permanently die out. Based on this prerequisite, we can transform (3) as:
Then we can obtain
Next, we will prove that for the susceptible S(t), there is one periodic solution. Firstly, we consider
It follows from Lemma 1 that a unique globally asymptotical periodic solution of (6) exists, and it is given as
Therefore, there exists one infection-free periodic solution which is given by \((S^*(t),0,0,0,\frac{\Pi }{\mu },0).\)
Theorem 1
For system (3), \(\left( S^*(t),0,0,0,\frac{\Pi }{\mu },0 \right)\) has global attractivity for \(R_1^* < 1\), in which
Proof
Since \(R_1^*< 1\), \(\varepsilon _1^{} > 0\) can be chosen such that
According to \(\dot{N}(t) \le - \mu N(t)+\Pi ,\) then there exists \(n_1>0\) satisfying the following inequality
By (3), then
When \(t > n_1 T\) and \(n > n_1,\) we investigate
For system (10), we can obtain the globally asymptotical stable periodic solution as \(u^*(t) = S^*(t),\) utilizing Lemma 1, and also there is \(n_2> n_1\) such that when \(n > n_2\),
Then
Thus
Therefore, for \(\forall \varepsilon _2> 0\), there is \(n_3>n_2\), satisfying \(E(t) < \varepsilon _2\) when \(t > n_3 T\).
Then, according to (3), we can obtain that if \(t> nT,n> n_3,\)
i.e.
Hence,
Similarly, for \(\forall \varepsilon _3 > 0\), there is \(n_4 > n_3\), satisfying
when \(t > n_4 T\).
Exploiting the fourth one of (3), the following inequality can be obtained as
for all \(t> nT (n > n_4).\)
Obviously
Therefore, for arbitrary \(\forall \varepsilon _4 > 0\), there is \(n_5>n_4\) satisfying \(A(t) < \varepsilon _4\) when \(t > n_5 T.\)
Next, according to the sixth one of (3), we get
Obviously,
Therefore, for \(\forall \varepsilon _5>0\), there is \(n_6 > n_5\) satisfying \(M(t) < \varepsilon _5\) at the case \(t> n_6 T.\)
Eventually, we can obtain
when \(t > n_6 T.\)
When \(t > n_6 T\), the following comparative model will be investigated.
Obviously,
Exploiting the corresponding lemma of25, we can obtain that if \(t> n_7 T\), there is \(n_7 > n_6\) such that
Because \(\varepsilon _3>0\) and \(\varepsilon _6>0,\) and they are extremely small, we can obtain by (9) and (17) that
From (9) and (12)–(15), we can get the following conclusion:
For \(\forall \varepsilon _7>0\), there is \(n_8 > n_7\) satisfying
when \(t > n_8 T.\)
When \(t > n_8 T\), it follows from (3) that
when \(t > nT\) and \(n > n_8.\)
Considering
based on Lemma 1, its unique asymptotical stable periodic solution can be computed as
when \(nT<t\le (n + 1)T,\) in which
According to25, there exists \(n_9 > n_8\) satisfying
for
Because \(\varepsilon _7\) and \(\varepsilon _1\) take the very small value, then we have
when \(nT<t\le (n + 1)T.\)
By (12)–(15), (18) and (21), the infection-free periodic solution \((S^*(t),0,0,0,\frac{\Pi }{\mu },0)\) is the globally attractive. Based on Theorem 1, we can obtain some assertions as follows.
Proposition 1
-
(a)
The infection-free periodic solution which is given by \((S^*(t),0,0,0,\frac{\Pi }{\mu },0)\) is globally attractive, when
$$\begin{aligned} \Pi \left( {\frac{{\beta \mu }}{{\Pi + \mu K}} + \frac{{\eta \Pi (\lambda _1 + \lambda _2 )}}{{\mu (d-b)}}} \right) < \mu ((1 - \theta ) + \theta \rho + \mu ); \end{aligned}$$ -
(b)
If \(T<T^*\) and
$$\begin{aligned} \Pi \left( {\frac{{\beta \mu }}{{\Pi + \mu K}} + \frac{{\eta \Pi (\lambda _1 + \lambda _2 )}}{{\mu (d-b)}}} \right) > \mu ((1 - \theta ) + \theta \rho + \mu ), \end{aligned}$$\(\left( S^*(t),0,0,0,\frac{\Pi }{\mu },0\right)\) is globally attractive, where
$$\begin{aligned} \begin{array}{*{20}c} \begin{array}{l} T^* = \frac{1}{\mu }\ln \left( {1 + \frac{{\upsilon \theta _1 \mu }}{{\Pi - \upsilon \mu }}} \right) , \\ A = \eta \pi (\lambda _1 + \lambda _1 ), \\ B = \mu (d-b), \\ C = (1 - \theta )\varpi + \theta \rho + \mu , \\ \end{array} \\ {\upsilon = \frac{{ - \left( {B + KA - BC} \right) + \sqrt{\left( {B + KA - BC} \right) ^2 + 4KABC} }}{{2A}}}. \\ \end{array} \end{aligned}$$
Proposition 2
If
\(\left( S^*(t),0,0,0,\frac{\Pi }{\mu },0 \right)\) is globally attractive.
Uniform persistence
In this section, we will discuss the sufficient conditions for the Gompertz virus disease model with impulsive effect. In order to obtain uniform persistence, the following lemma needs to be proved first.
Lemma 2
If \(R_{1*}> 1,\) then for arbitrary \(t_0 > 0, E(t) < \mathop E\limits ^\_\) doesn’t hold true for all \(t > t_0 ,\) where
Proof
Herein we use the method of contradiction. We assume that there is \(t_0 > 0,\) which satisfies when \(t > t_0,\)
When \(t_0<t,\)
and
By (23) and (24), then \(\exists \varepsilon ^* > 0\) and when \(t_1 > t_0 ,\) then
and
for arbitrary \(t > t_1.\)
Thus, when \(t > t_1,\)
It is not difficult to check that there is \(t_2> t_1,\) such that
Hence, when \(t \ne nT (n \in N),\) and \(t > t_2,\) we can obtain
For the case of \(n_{10} T > t_2\) and \(t> n_{10} T,\)
It follows from Lemma 1 that
is globally attractive, in which
Thus, there exists a constant \(t_3 > t_2,\) satisfying that when \(t> t_3,\)
According to (26) and (3), we can obtain
Integrating (27) from nT to \((n+1)T,\) considering that \(\varepsilon ^*\) is arbitrary small and \({\bar{E}}>0\), then we can get
Using \(R_{1*} > 1,\) we get
Thus
This is the contradiction with that E(t) is bounded. Thus, there is \(t^* > t_0,\) such that \(E(t^*) \ge \mathop E\limits ^\_,\) and the above prediction is proved.
Theorem 2
When \(R_{1*} > 1,\) for system (3), there will be \(q_2\) satisfying that there exists some positive solution which satisfies \(q_2\le E(t).\)
Proof
Define
According to Lemma 2, \(\left( {S(t),E(t),I(t),A(t),N(t),M(t)} \right)\) is discussed based on the following cases.
Firstly, \(E(t) \ge \mathop E\limits ^\_\) when t takes sufficiently large value.
Secondly, E(t) vibrates about \(\mathop E\limits ^\_\) when t takes sufficiently large value.
For case 1, we have \(\mathop {\lim }\limits _{t \rightarrow \infty } \inf E(t)> q_2 .\) Immediately, we have the conclusion of Theorem 2.
For case 2, let \(t_4> t_3\) and \(t_5\) be sufficiently large satisfying
If \(t_5- t_4\le T\), since
and
we have \(E(t) \ge q_2\) (\(t\in [t_4,t_5 ]\)).
When \(t_5-t_4> T,\) then
if \(t \in [t_4,t_5]\).
Similarly, we can prove
when \(t \in [t_4,t_4 + T]\) and
when \(t \in [t_4 + T,t_5]\).
In the following, the conclusion that \(E(t) \ge q_2\) holds true when \(t_4 + T\le t\le t_5,\) will be proved.
Herein we use the method of contradiction, and it means there is a positive constant \(T^\prime\) satisfying \(q_2\le E(t)\) if \(t_4\le t\le T'+ T+t_4,\)
and
Exploiting (3), and \(t =T+T'+t_4\), then
This is a contradiction. So \(E(t) \ge q_2\) when \(t \in [t_4 ,t_5].\)
Theorem 3
If \(R_{1*} > 1,\) we have the conclusion that model (3) is permanent.
Proof
We assume the arbitrary solution of (3) is denoted by \(\left( {S(t),E(t),I(t),A(t),N(t),M(t)} \right) .\)
If t takes a sufficiently large value, we can obtain
Using the same method, we have
where
(\(\varepsilon ^*\) is sufficiently small).
When t takes a sufficiently large value, we derive
Therefore
where
When t is sufficiently large, we can obtain
So
where
According to Theorem 2, when t takes sufficiently large value, then
Therefore
where
( \(\varepsilon ^*\) is sufficiently small).
Set
It follows from Theorem 3 and the analysis above that \(\Omega _0\) belongs to the globally attractive region of \(\Omega .\) For the system (3), the positive solution satisfying the condition (*) will finally enter and remain in \(\Omega _0,\) i.e., the system will be permanent.
According to Theorem 2 and Theorem 3, the following assertions can be easily obtained.
Proposition 3
If \(\theta _1 < \theta _1^*\), system (3) will be permanent, i.e., the Gompertz virus will be an endemic disease, in which
When \(T > \mathop T\limits ^\_\), system (3) will be permanent, and the Gompertz virus will be an endemic disease, in which
It follows from Theorem 1 and Theorem 3 that the Gompertz virus will not appear when \(R_1^*< 1\), and when \(R_{1*}>1,\) the Gompertz virus will be uniformly persistent.
Numerical simulations
In this section, the Runge-Kutta method is exploited to perform numerical simulations of Gompertz virus disease model with impulsive effect. Numerical simulations are presented to validate the analyzed conclusion from the view of impulsive differential equation theory.
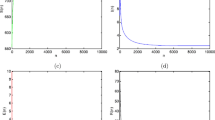
Dynamical behavior of system (3) with \(\beta =0.1, \eta =0.2, \lambda _1=0.1, \Pi = 5.1, \mu =1, b=0.3, \lambda _2=0.15, \rho = 1, T= 1, d= 1, K= 2, \varpi = 1, \tau _1= 1.5, \tau _2= 1, \xi = 0.02, \theta _1=0.1, \theta =0.5, d_1= 0.2,\) and all the parameters satisfy \(R_1^*=0.6498<1.\) (a) (I(t), E(t), S(t)); (b) trajectory of the susceptible pest S(t); (c) trajectory of the exposed pest E(t); (d) trajectory of the infected pest I(t); (e) trajectory of the asymptotically infected pest A(t); (f) trajectory of the total population of pest N(t); (g) trajectory of the reservoir or the seafood place or market M(t).
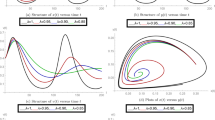
Dynamical behavior of system (3) with \(\beta =5, \eta =2, \lambda _1=0.1, \Pi = 5.1, \mu =0.3, b=0.3, \lambda _2=0.15, \rho =1.3, T= 1, d= 1, K=3, \varpi = 2, \tau _1= 25, \tau _2=0.2, \xi = 0.02, \theta _1=0.13, \theta =0.5, d_1= 0.2,\) and all the parameters satisfy \(R_{1*}=2.0644>1.\) (a) (I(t), E(t), S(t)); (b) trajectory of the susceptible pest S(t); (c) trajectory of the exposed pest E(t); (d) trajectory of the infected pest I(t); (e) trajectory of the asymptotically infected pest A(t); (f) trajectory of the total population of pest N(t); (g) trajectory of the reservoir or the seafood place or market M(t).
Firstly, when we choose \(\Pi = 5.1, \lambda _1=0.1, \eta =0.2, \beta =0.1, \mu =1, b=0.3, \lambda _2=0.15, \rho = 1, T= 1, d= 1, K= 2, \varpi = 1, \tau _1= 1.5, \tau _2= 1, \xi = 0.02, \theta _1=0.1, \theta =0.5, d_1= 0.2,\) we compute \(R_1^*=0.6498<1,\) which satisfies the condition of Theorem 1. By Theorem 1, the infection-free periodic solution of (3) is globally asymptotically stable. Figure 1 also demonstrates this conclusion: The periodic solution has the characteristic of global attractivity. It can be shown from Fig. 1 that S(t) goes to oscillatory; E(t), I(t), A(t) and M(t) go to extinction; N(t) goes to some stable value. It implies that the susceptible pest population oscillate with a positive amplitude, i.e., they will not go extinct. Additionally, the infective pests almost will go extinct and have little effect on the crop, which can be shown in Fig. 1.
Moreover, when we choose \(\Pi = 5.1, \lambda _1=0.1, \eta =2, \beta =5, \mu =0.3, b=0.3, \lambda _2=0.15, \rho =1.3, T= 1, d= 1, K=3, \varpi = 2, \tau _1= 25, \tau _2=0.2, \xi = 0.02, \theta _1=0.13, \theta =0.5, d_1= 0.2,\) we compute \(R_{1*}=2.01>1,\) which satisfies the condition of Theorem 3. By Theorem 3, we can obtain that the proposed model is permanent, which can be shown in Fig. 2. Figure 2 also demonstrates this conclusion: The periodic solution has the characteristic of permanence. It can be shown in Fig. 2 that M(t), A(t), I(t), E(t) and S(t) go to oscillatory; N(t) goes to some stable value. It means that all the species will not go extinct and will almost maintain a stable value, which can be shown in Fig. 2.
From the view of ecology, infective pests almost have no effect on crops, but the pests that have a significant impact on the crops will become extinct. This will also demonstrate the effectiveness of scientific control. Actually, the purpose of scientific control is not to eradicate pests, but to control the number of pests to a certain level.
Conclusions
A new mathematical biological model on the Gompertz virus impulsive system is proposed in this paper. This proposed model considers the factors of the recovered or the removed population, asymptotically infected, infected (symptomatic), exposed and susceptible population. Utilizing the Floquet theory, we strictly prove the infection-free periodic solution of the proposed model is globally attractive if \(R_1^* < 1.\) It means that the infective pests almost will go extinct and have little effect on the crop. Moreover, we also obtain that the system (3) is permanent if \(R_{1*}> 1.\) Hence, we exploit the impulsive control method based on the effect of the Gompertz virus on pest such that \(R_1^* < 1,\) and then drive the virus to extinction. In addition, we can also control that the susceptible, exposed, infected (symptomatic), asymptotically infected population and the Gompertz virus in the market or seafood place or the reservoir oscillate with a positive amplitude. Actually, the scientific impulsive control is not to eradicate pests, but to control the number of pests to a certain level. Therefore, the proposed impulsive control strategy in this paper is very effective in impulsive control for the Gompertz virus model. Considering that time delays unavoidably exist in the transmission of the impulsive information and sampling in lots of practical cases, the proposed system will be extended to the delayed impulsive control for the Gompertz virus disease model in the future.
Data availibility
Data will be made available on request from the corresponding author (Youxiang **e).
References
Guo, S. J., Chen, Y. M. & Wu, J. H. Two-parameter bifurcations in a network of two neurons with multiple delays. J. Differ. Equ. 244, 444–486 (2008).
Ji, W., Zhang, H. & Qiu, J. Fuzzy affine model-based output feedback controller design for nonlinear impulsive systems. Commun. Nonlinear Sci. Numer. Simul. 79, 104894 (2019).
Driessche, P. V. & Zou, X. Global attractivity in delayed Hopfield neural network models. SIAM J. Appl. Math. 58, 1878–1890 (1998).
Wang, L. J. & Han, X. Stability and Hopf bifurcation analysis in bidirectional ring network model. Commun. Nonlinear Sci. Numer. Simul. 16, 3684–3695 (2011).
Wang, B. X. & Jian, J. G. Stability and Hopf bifurcation analysis on a four-neuron BAM neural network with distributed delays. Commun. Nonlinear Sci. Numer. Simul. 15, 189–204 (2010).
Jiang, X. W. et al. Bifurcation, chaos, and circuit realisation of a new four-dimensional memristor system. De Gruyter. https://doi.org/10.1515/ijnsns-2021-0393 (2021).
Gao, L., Wang, D. & Zong, G. Exponential stability for generalized stochastic impulsive functional differential equations with delayed impulses and Markovian switching. Nonlinear Anal. Hybrid Syst. 30, 199–212 (2018).
Yu, H. G., Zhong, S. M., Agarwal, R. P. & Sen, S. K. Effect of seasonality on the dynamical behavior of an ecological system with impulsive control strategy. J. Franklin Inst. 348, 652–670 (2011).
Wang, L. J., **e, Y. X. & Fu, J. Q. The dynamics of natural mortality for pest control model with impulsive effect. J. Franklin Inst. 350, 1443–1461 (2013).
Zou, L., **ong, Z. L. & Shu, Z. P. The dynamics of an eco-epidemic model with distributed time delay and impulsive control strategy. J. Franklin Inst. 348, 2332–2349 (2011).
**e, Y. X., Yuan, Z. H. & Wang, L. J. Dynamic analysis of pest control model with population dispersal in two patches and impulsive effect. J. Comput. Sci. 5, 685–695 (2014).
**e, Y. X., Wang, L. J., Deng, Q. C. & Wu, Z. J. The dynamics of an impulsive predator-prey model with communicable disease in the prey species only. Appl. Math. Comput. 292, 320–335 (2017).
Liang, J. H., Tang, S. Y. & Cheke, R. A. Beverton-Holt discrete pest management models with pulsed chemical control and evolution of pesticide resistance. Commun. Nonlinear Sci. Numer. Simul. 36, 327–341 (2016).
Shah, K., Abdeljawad, T., Jarad, F. & Al-Mdallal, Q. On nonlinear conformable fractional order dynamical system via differential transform method. Comput. Model. Eng. Sci. 136, 1457–1472 (2023).
Li, B., Eskandari, Z. & Avazzadeh, Z. Strong resonance bifurcations for a discrete-time preyCpredator model. J. Appl. Math. Comput. 69, 2421–2438 (2023).
Li, B., Eskandari, Z. & Avazzadeh, Z. Dynamical behaviors of an SIR epidemic model with discrete time. Fract. Fract. 659, 1–17 (2022).
Shah, K., Abdalla, B., Abdeljawad, T. & Gul, R. Analysis of multipoint impulsive problem of fractional-order differential equations. Bound. Value Probl.https://doi.org/10.1186/s13661-022-01688-w (2023).
Sitthiwirattham, T. et al. Study of implicit-impulsive differential equations involving Caputo-Fabrizio fractional derivative. AIMS Math. 7(3), 4017–4037 (2021).
Shah, K., Ahmad, I., Nieto, J. J., Rahman, G. U. & Abdeljawad, T. Qualitative investigation of nonlinear fractional coupled pantograph impulsive differential equations. Qual. Theory Dyn. Syst. 21, 131 (2022).
Wang, Y. Q. & Lu, J. Q. Some recent results of analysis and control for impulsive systems. Commun. Nonlinear Sci. Numer. Simul. 80, 104862 (2020).
Guan, Z., Chen, G. & Jian, M. On delayed impulsive Hopfield neural networks. Neural Netw. 12(2), 273–280 (1999).
Li, X., Shen, J. & Rakkiyappan, R. Persistent impulsive effects on stability of functional differential equations with finite or infinite delay. Appl. Math. Comput. 329, 14–22 (2018).
Lakshmikantham, V. & Simeonov, P. Theory of Impulsive Differential Equations (World Scientific, 1989).
Bainov, D.D. & Simeonov, P.S. Impulsive differential equations: Periodic solutions and application. In Pitman Monographs and Surveys in Pure and Applied Mathematics, vol. 66, Longman Science and Technical, Harlow, UK (1993).
Lakshmikantham, V., Bainov, D. & Simeonov, P. Theory of Impulsive Differential Equations (World Scientific Publisher, 1989).
Liang, Z. et al. Periodic solution of a Leslie predator-prey system with ratio-dependent and state impulsive feedback control. Nonlinear Dyn. 89(4), 2941–2955 (2017).
Fang, D. et al. Periodicity induced by state feedback controls and driven by disparate dynamics of a herbivore-plankton model with cannibalism. Nonlinear Dyn. 90(4), 2657–2672 (2017).
Zhang, T. et al. Periodic solution of a pest management Gompertz model with impulsive state feedback control. Nonlinear Dyn. 78(2), 921–938 (2014).
Wang, T. & Chen, L. Nonlinear analysis of a microbial pesticide model with impulsive state feedback control. Nonlinear Dyn. 65(1–2), 1–10 (2011).
Li, Z. & Chen, L. Periodic solution of a turbidostat model with impulsive state feedback control. Nonlinear Dyn. 58(3), 525–538 (2009).
Li, W. J., Ji, J. C. & Huang, L. H. Global dynamics analysis of a water hyacinth fish ecological system under impulsive control. J. Franklin Inst. 359, 10628–10652 (2022).
Li, W. X., Chen, Y. M., Huang, L. H. & Wang, J. F. Global dynamics of a filippov predator-prey model with two thresholds for integrated pest management. Chaos Solit. Fract. 157, 111881 (2022).
Qin, W. J., Tan, X. W., Tosato, M. & Liu, X. Z. Threshold control strategy for a non-smooth filippov ecosystem with group defense. Appl. Math. Comput. 362, 1–18 (2019).
Li, W. X., Huang, L. H. & Wang, J. F. Dynamic analysis of discontinuous plant disease models with a non-smooth separation line. Nonlinear Dyn. 99(2), 1675–1697 (2020).
Khan, T., Khan, A. & Zaman, G. The extinction and persistence of the stochastic hepatitis B epidemic model. Chaos Solit. Fract. 108, 123–128 (2018).
Khan, T. et al. The transmission dynamics of hepatitis B virus via the fractional-order epidemiological model. Complexity 2021, 8752161 (2021).
Khan, T., Zaman, G. & Chohan, M. I. The transmission dynamic and optimal control of acute and chronic hepatitis B. J. Biol. Dyn. 11, 172–189 (2017).
Khan, T., Zaman, G. & El-Khatib, Y. Modeling the dynamics of novel coronavirus (COVID-19) via stochastic epidemic model. Results Phys. 24, 104004 (2021).
Khan, T., Ullah, R., Zaman, G. & Alzabut, J. A mathematical model for the dynamics of SARS-CoV-2 virus using the Caputo-Fabrizio operator. Math. Biosci. Eng. 18(5), 6095–6116 (2021).
Ullah, R. et al. The dynamics of novel corona virus disease via stochastic epidemiological model with vaccination. Sci. Rep. 13, 3805 (2023).
Wang, L. M., Chen, L. S. & Nieto, J. J. The dynamics of an epidemic model for pest control with impulsive effect, Journal of. Nonlinear Anal. Real World Appl. 11, 1374–1386 (2010).
Liu, J. N., Qi, Q., Liu, B. & Gao, S. J. Pest control switching models with instantaneous and non-instantaneous impulsive effects. Math. Comput. Simul. 205, 926–938 (2023).
Djuikem, C., Grognard, F. & Touzeau, S. Impulsive modelling of rust dynamics and predator releases for biocontrol. Math. Biosci. 356, 108968 (2023).
**ao, Y. & Bosch, F. V. D. The dynamics of an eco-epidemic model with biological control. Ecol. Model. 168, 203–214 (2003).
Rhodes, C. J. & Anderson, R. M. Forest-fire as a model for the dynamics of disease epidemics. J. Franklin Inst. 335, 199–211 (1998).
Sisodiya, O. S., Misraa, O. P. & Dharb, J. Dynamics of cholera epidemics with impulsive vaccination and disinfection. Math. Biosci. 298, 46–57 (2018).
Acknowledgements
This work was supported by the Open Fund of Hubei Key Laboratory of Hydroelectric Machinery Design and Maintenance (2019KJX12).
Author information
Authors and Affiliations
Contributions
L.W. contributed to the conception of the study. A.S. contributed significantly to data analysis and manuscript preparation; Y.X. helped to perform the analysis with constructive discussions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, L., She, A. & **e, Y. The dynamics analysis of Gompertz virus disease model under impulsive control. Sci Rep 13, 10180 (2023). https://doi.org/10.1038/s41598-023-37205-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37205-x
- Springer Nature Limited
This article is cited by
-
Towards a crop pest control system based on the Internet of Things and fuzzy logic
Telecommunication Systems (2024)
-
Nonparaxial solitons and the dynamics of solitary waves for the coupled nonlinear Helmholtz systems
Optical and Quantum Electronics (2023)