Abstract
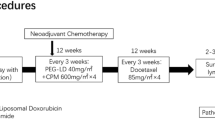
The present study aimed to investigate the efficacy and toxicity of pegylated liposomal doxorubicin (PLD) in preoperative neoadjuvant chemotherapy for patients with breast cancer by comparing with conventional anthracycline. This study is a non-randomized controlled trial. Prospective analysis was conducted after matching as required. A total of 146 patients with confirmed diagnosis of breast cancer by histopathological examinations were enrolled into the observation group and control group in 1:1 ratio. Each of the cases in the observation group was required to correspond to another in the control group according to the requirements including age, molecular subtype, axillary node status, and regimen of the preoperative neoadjuvant chemotherapy. The chemotherapy was based on regimens consisting of anthracyclines, paclitaxel or docetaxel, and/or platinum. PLD was used at least twice in the observation group, with traditional anthracycline as a contrast in the control group. Clinical responses as well as cardiac side effects and other adverse reactions were evaluated by clinical and imaging examinations such as electrocardiogram (ECG) and color Doppler ultrasound during the chemotherapy. Pathologic examinations were performed following the surgeries after preoperative neoadjuvant chemotherapy. All the patients in both groups completed the preoperative neoadjuvant chemotherapy according to their original regimens. The postoperative pathological evaluation revealed a higher pathologic complete response (PCR) rate and significantly more patients of grade V of the Miller-Payne grading system in the observation group as compared to the control group (p = 0.047). In addition, the observation group recorded an evidently lower occurrence of the adverse cardiac events (p = 0.014), ECG changes (p = 0.048), and the relatively severe adverse reactions such as myelosuppression. Compared with conventional anthracycline drugs, PLD has a better pathologic response and safety performance, as well as a similar clinical effectiveness in preoperative neoadjuvant chemotherapy for breast cancer.
Similar content being viewed by others
Introduction
Breast cancer is one of the most common malignant tumors in women worldwide, with a mortality rate only lower than that of lung cancer among all the malignancies. Every year, more than 1.3 million women in the world are newly diagnosed with breast cancer, and about 500,000 cases die of this life-threatening disease1. Breast cancer management towards a favorable prognosis requires comprehensive measures, among which chemotherapy has always been used as a key part due to its substantial clinical effects in disease control.
Neoadjuvant therapy is typically used for the management of local malignant tumors prior to surgery and other treatment2. There are three types of neoadjuvant therapy for breast cancer—preoperative neoadjuvant chemotherapy, targeted therapy and endocrine therapy. A number of clinical trials showed that 80% of the patients with breast cancer achieved a significant reduction in tumor size, and about 10–20% obtained pathologic complete response (PCR) by preoperative neoadjuvant chemotherapy3. National Comprehensive Cancer Network (NCCN) guidelines also recommend preoperative neoadjuvant chemotherapy as a routine for stage II and part of stage III breast cancer4.
However, a variety of side effects, even toxic responses concomitant with chemotherapy have put a limit to the clinical application of the antineoplastics, resulting in the restricted use of some potent chemotherapeutic drugs despite of their promising future in improving the survival rate for patients with breast cancer. Toxic reactions caused by chemotherapy varies with the agents, but most commonly nausea, vomiting, myelosuppression, and even some severer side effects that may discontinue the treatment. Cardiotoxicity of a drug is usually confirmed when congestive heart failure occurs accompanied by relevant clinical symptoms after treatment, or left ventricular ejection fraction (LVEF) is less than 55% or decreases by more than 10% compared with the reference value although absent of clinical symptoms5,6. The clinical symptoms suggesting congestive heart failure include but not limited to lung moist rale, pretibial edema of both legs, and cyanosis of lip or mouth.
There are many kinds of antineoplastics used for chemotherapy. Anthracyclines, taxanes and targeted drugs are frequently employed as adjuvant drugs in breast cancer treatment. Anthracyclines, for instance, has been constantly playing an irreplaceable role in chemotherapy, particularly for the patients with locally advanced breast cancer19, was used for all the patients treated with anthracyclines to prevent the occurrence of cardiac toxic events, and thus may compromise the results of our study to some extent. Moreover, as the period for observation is not long enough to evaluate the accumulation of the agents, long-term follow-ups are necessary to trace the long-term survival rate and the adverse reactions.
References
Siegel, R. et al. Cancer statistics, 2014. CA Cancer J. Clin. 64(1), 9–29 (2014).
Curigliano, G. et al. De-escalating and escalating treatments for early-stage breast cancer: The St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann. Oncol. 28, 1700–1712. https://doi.org/10.1093/annonc/mdx308 (2017).
Bear, H. D. et al. Sequential preoperative or postoperative docetaxel added to preoperative doxorubicin plus cyclophosphamide for operable breast cancer National Surgical Adjuvant Breast and Bowel Project Protocol B—27. J. Clin. Oncol. 24, 24019–24202 (2006).
Kaufmannm, M. et al. Recommendations from an international expert panel on the use of neoadjuvant (primary) systemic treatment of operable breast cancer: An update. J. Clin. Oncol. 24, 1940–1949 (2006).
Bird, B. R. & Swain, S. M. Cardiac toxicity in breast cancer survivors: Review of potential cardiac problems. Clin. Cancer Res. 14(1), 14–24 (2008).
Chen, S. et al. Clinical and pathological response to neoadjuvant chemotherapy based on primary tumor reduction is correlated to survival in hormone receptor-positive but not hormone receptor-negative locally advanced breast cancer. Ann. Surg. Oncol. 22(1), 32–39 (2015).
**ng, M., Yan, F. & Yu, S. Efficacy and cardiotoxicity of liposomal doxorubicin-based chemotherapy in advanced breast cancer: A meta-analysis of ten randomized controlled trials. PLoS ONE 10, e0133569. https://doi.org/10.1371/journal.pone.0133569 (2015).
Shah, A. N. Adjuvant anthracyclines in breast cancer: What is their role?. Oncologist 23, 1153–1161. https://doi.org/10.1634/theoncologist.2017-0672 (2018).
Gil-Gil, M. J. et al. Pegylated liposomal doxorubicin plus cyclophosphamide followed by paclitaxel as primary chemotherapy in elderly or cardiotoxicity prone patients with high-risk breast cancer: Results of the phase II CAPRICE study. Breast Cancer Res. Treat. 151, 597–606 (2015).
Gabizon, A. et al. An open-label study to evaluate dose and cycle dependence of the pharmacokinetics of pegylated liposomal doxorubicin. Cancer Chemother. Pharmacol. 61, 695–702 (2008).
Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0: National Cancer Institute of Health (2009).
Bear, H. D. et al. The effect on tumor response of adding sequential preoperative docetaxel to preoperative doxorubicin and cyclophosphamide: Preliminary results from National Surgical Adjuvant Breast and bowel project protocol B-27. J. Clin. Oncol. 21, 4165–4174. https://doi.org/10.1200/JCO.2003.12.005 (2003).
Khokher, S. & Qureshi, M. U. Comparison of WHO and RECIST criteria for evaluation of clinical response to chemotherapy in patients with advanced breast cancer. Asian Pac. J. Cancer Prev. 13, 3213–3218 (2012).
Cortazar, P. Pathological complete response in neoadjuvant treatment of breast cancer. Ann. Surg. Oncol. 22, 1441–1446. https://doi.org/10.1245/s10434-015-4404-8 (2015).
**e, F. et al. Efficacy of two-weekly nanoparticle albumin-bound paclitaxel as neoadjuvant chemotherapy for breast cancer. Nanomedicine (Lond.) 14, 1595–1603. https://doi.org/10.2217/nnm-2018-0485 (2019).
Suzuki, C. et al. Radiologic measurements of tumor response to treatment: Practical approaches and limitations. Radiographics 28, 329–344 (2008).
Partridge, S. C. et al. Accuracy of MR imaging for revealing residual breast cancer in patients who have undergone neoadjuvant chemotherapy. AJR 179, 1193–1199 (2002).
Airoldi, M. et al. Clinical activity and cardiac tolerability of non-pegylated liposomal doxorubicin in breast cancer: A synthetic review. Tumori 97(6), 690–692 (2011).
Asselin, B. L. et al. Cardioprotection and safety of dexrazoxane in patients treated for newly diagnosed T-cell acute lymphoblastic leukemia or advanced-stage lymphoblastic non-hodgkin lymphoma: A report of the children’s oncology group randomized trial pediatric oncology group 9404. J. Clin. Oncol. 34, 854–862. https://doi.org/10.1200/JCO.2015.60.8851 (2016).
Acknowledgements
The authors are grateful to the patients for their contribution to the study. The statistical analysis was performed under the guidance of the Medical Statistics Department of Jilin University. The present study was financially supported by the Bethune Plan B (Jilin, China; Grant No. 2012217), Department of finance of Jilin Province (Grant Nos. 2018SCZWSZX-035 and JLSWSRCZX2020-0042), and Department of science and technology of Jilin Province (Grant No. 20190701041GH).
Author information
Authors and Affiliations
Contributions
W.L., P.Z., G.Z., X.Z. and A.S. wrote the main manuscript text, others prepared all figures and tables. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, W., Chen, W., Zhang, X. et al. Higher efficacy and reduced adverse reactions in neoadjuvant chemotherapy for breast cancer by using pegylated liposomal doxorubicin compared with pirarubicin. Sci Rep 11, 199 (2021). https://doi.org/10.1038/s41598-020-80415-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-80415-w
- Springer Nature Limited
This article is cited by
-
Ultrasound-based radiomics for early predicting response to neoadjuvant chemotherapy in patients with breast cancer: a systematic review with meta-analysis
La radiologia medica (2024)
-
Neoadjuvant pegylated liposomal doxorubicin- and epirubicin-based combination therapy regimens for early breast cancer: a multicenter retrospective case–control study
Breast Cancer Research and Treatment (2023)