How appendages, such as plant leaves or animal limbs, develop asymmetric shapes remains a fundamental question in biology. Although ongoing research has revealed the genetic regulation of organ pattern formation, how gene activity ultimately directs organ shape remains unclear. Here, we show that leaf dorsoventral (adaxial-abaxial) polarity signals lead to mechanical heterogeneity of the cell wall, related to the methyl-esterification of cell-wall pectins in tomato and Arabidopsis. Numerical simulations predicate that mechanical heterogeneity is sufficient to produce the asymmetry seen in planar leaves. Experimental tests that alter pectin methyl-esterification, and therefore cell wall mechanical properties, support this model and lead to polar changes in gene expression, suggesting the existence of a feedback mechanism for mechanical signals in morphogenesis. Thus, mechanical heterogeneity within tissue may underlie organ shape asymmetry.






Similar content being viewed by others
References
Sablowski, R. Coordination of plant cell growth and division: collective control or mutual agreement? Curr. Opin. Plant Biol. 34, 54–60 (2016).
Lecuit, T. & Lenne, P. F. Cell surface mechanics and the control of cell shape, tissue patterns and morphogenesis. Nat. Rev. Mol. Cell Biol. 8, 633–644 (2007).
Sampathkumar, A., Yan, A., Krupinski, P. & Meyerowitz, E. M. Physical forces regulate plant development and morphogenesis. Curr. Biol. 24, R475–R483 (2014).
Louveaux, M., Julien, J. D., Mirabet, V., Boudaoud, A. & Hamant, O. Cell division plane orientation based on tensile stress in Arabidopsis thaliana. Proc. Natl Acad. Sci. USA 113, E4294–E4303 (2016).
Sampathkumar, A. et al. Subcellular and supracellular mechanical stress prescribes cytoskeleton behavior in Arabidopsis cotyledon pavement cells. eLife 3, e01967 (2014).
Peaucelle, A. et al. Pectin-induced changes in cell wall mechanics underlie organ initiation in Arabidopsis. Curr. Biol. 21, 1720–1726 (2011).
Hervieux, N. et al. A mechanical feedback restricts sepal growth and shape in Arabidopsis. Curr. Biol. 26, 1019–1028 (2016).
Braam, J. In touch: plant responses to mechanical stimuli. New Phytol. 165, 373–389 (2005).
Gibson, W. T. et al. Control of the mitotic cleavage plane by local epithelial topology. Cell 144, 427–438 (2011).
Boudon, F. et al. A computational framework for 3D mechanical modeling of plant morphogenesis with cellular resolution. PLoS Comput. Biol. 11, e1003950 (2015).
Cosgrove, D. J. Wall extensibility: its nature, measurement and relationship to plant cell growth. New Phytol. 124, 1–23 (1993).
Cosgrove, D. J. Growth of the plant cell wall. Nat. Rev. Mol. Cell Biol. 6, 850–861 (2005).
Wolf, S., Hematy, K. & Hofte, H. Growth control and cell wall signaling in plants. Annu. Rev. Plant Biol. 63, 381–407 (2012).
Peaucelle, A., Braybrook, S. & Hofte, H. Cell wall mechanics and growth control in plants: the role of pectins revisited. Front. Plant. Sci. 3, 121 (2012).
Peaucelle, A. et al. Arabidopsis phyllotaxis is controlled by the methyl-esterification status of cell-wall pectins. Curr. Biol. 18, 1943–1948 (2008).
Ali, O., Mirabet, V., Godin, C. & Traas, J. Physical models of plant development. Annu. Rev. Cell Dev. Biol. 30, 59–78 (2014).
Waites, R. & Hudson, A. phantastica: a gene required for dorsoventrality of leaves in Antirrhinum majus. Development 121, 2143–2154 (1995).
Barton, M. K. Twenty years on: the inner workings of the shoot apical meristem, a developmental dynamo. Dev. Biol. 341, 95–113 (2010).
Bowman, J. L. & Floyd, S. K. Patterning and polarity in seed plant shoots. Annu. Rev. Plant Biol. 59, 67–88 (2008).
Braybrook, S. A. & Kuhlemeier, C. How a plant builds leaves. Plant Cell 22, 1006–1018 (2010).
Efroni, I., Eshed, Y. & Lifschitz, E. Morphogenesis of simple and compound leaves: a critical review. Plant Cell 22, 1019–1032 (2010).
Husbands, A. Y., Chitwood, D. H., Plavskin, Y. & Timmermans, M. C. Signals and prepatterns: new insights into organ polarity in plants. Genes Dev. 23, 1986–1997 (2009).
Xu, L., Yang, L. & Huang, H. Transcriptional, post-transcriptional and post-translational regulations of gene expression during leaf polarity formation. Cell Res. 17, 512–519 (2007).
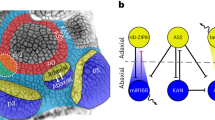
Emery, J. F. et al. Radial patterning of Arabidopsis shoots by class III HD-ZIP and KANADI genes. Curr. Biol. 13, 1768–1774 (2003).
Iwakawa, H. et al. The ASYMMETRIC LEAVES2 gene of Arabidopsis thaliana, required for formation of a symmetric flat leaf lamina, encodes a member of a novel family of proteins characterized by cysteine repeats and a leucine zipper. Plant Cell Physiol. 43, 467–478 (2002).
Siegfried, K. R. et al. Members of the YABBY gene family specify abaxial cell fate in Arabidopsis. Development 126, 4117–4128 (1999).
Sussex, I. M. Experiments on the cause of dorsiventrality in leaves. Nature 167, 651–652 (1951).
Qi, J. et al. Auxin depletion from leaf primordia contributes to organ patterning. Proc. Natl Acad. Sci. USA 111, 18769–18774 (2014).
Braybrook, S. A. & Peaucelle, A. Mechano-chemical aspects of organ formation in Arabidopsis thaliana: the relationship between auxin and pectin. PLoS ONE 8, e57813 (2013).
Cleland, R. Cell wall extension. Annu. Rev. Plant Physiol. 22, 197–222 (1971).
Milani, P. et al. In vivo analysis of local wall stiffness at the shoot apical meristem in Arabidopsis using atomic force microscopy. Plant J. 67, 1116–1123 (2011).
Verhertbruggen, Y., Marcus, S. E., Haeger, A., Ordaz-Ortiz, J. J. & Knox, J. P. An extended set of monoclonal antibodies to pectic homogalacturonan. Carbohydr. Res. 344, 1858–1862 (2009).
Clausen, M. H., Willats, W. G. & Knox, J. P. Synthetic methyl hexagalacturonate hapten inhibitors of anti-homogalacturonan monoclonal antibodies LM7, JIM5 and JIM7. Carbohydr. Res. 338, 1797–1800 (2003).
Liners, F., Thibault, J. F. & Van Cutsem, P. Influence of the degree of polymerization of oligogalacturonates and of esterification pattern of pectin on their recognition by monoclonal antibodies. Plant Physiol. 99, 1099–1104 (1992).
Peaucelle, A., Wightman, R. & Hofte, H. The control of growth symmetry breaking in the Arabidopsis hypocotyl. Curr. Biol. 25, 1746–1752 (2015).
Hayashi, K. et al. Rational design of an auxin antagonist of the SCF(TIR1) auxin receptor complex. ACS Chem. Biol. 7, 590–598 (2012).
Krogan, N. T. & Berleth, T. A dominant mutation reveals asymmetry in MP/ARF5 function along the adaxial-abaxial axis of shoot lateral organs. Plant Signal. Behav. 7, 940–943 (2012).
McConnell, J. R. et al. Role of PHABULOSA and PHAVOLUTA in determining radial patterning in shoots. Nature 411, 709–713 (2001).
Abley, K. et al. An intracellular partitioning-based framework for tissue cell polarity in plants and animals. Development 140, 2061–2074 (2013).
Kuchen, E. E. et al. Generation of leaf shape through early patterns of growth and tissue polarity. Science 335, 1092–1096 (2012).
Lawrence, P. A., Struhl, G. & Casal, J. Planar cell polarity: one or two pathways? Nat. Rev. Genet. 8, 555–563 (2007).
Stopper, G. F. & Wagner, G. P. Of chicken wings and frog legs: a smorgasbord of evolutionary variation in mechanisms of tetrapod limb development. Dev. Biol. 288, 21–39 (2005).
Kennaway, R., Coen, E., Green, A. & Bangham, A. Generation of diverse biological forms through combinatorial interactions between tissue polarity and growth. PLoS Comput. Biol. 7, e1002071 (2011).
Merks, R. M., Guravage, M., Inze, D. & Beemster, G. T. VirtualLeaf: an open-source framework for cell-based modeling of plant tissue growth and development. Plant Physiol. 155, 656–666 (2011).
Kierzkowski, D. et al. Elastic domains regulate growth and organogenesis in the plant shoot apical meristem. Science 335, 1096–1099 (2012).
Juge, N. Plant protein inhibitors of cell wall degrading enzymes. Trends Plant Sci. 11, 359–367 (2006).
Tian, C. et al. An organ boundary-enriched gene regulatory network uncovers regulatory hierarchies underlying axillary meristem initiation. Mol. Syst. Biol. 10, 755 (2014).
Talbert, P. B., Adler, H. T., Parks, D. W. & Comai, L. The REVOLUTA gene is necessary for apical meristem development and for limiting cell divisions in the leaves and stems of Arabidopsis thaliana. Development 121, 2723–2735 (1995).
Nakata, M. et al. Roles of the middle domain-specific WUSCHEL-RELATED HOMEOBOX genes in early development of leaves in Arabidopsis. Plant Cell 24, 519–535 (2012).
Sessions, A., Weigel, D. & Yanofsky, M. F. The Arabidopsis thaliana MERISTEM LAYER 1 promoter specifies epidermal expression in meristems and young primordia. Plant J. 20, 259–263 (1999).
Coen, E., Rolland-Lagan, A. G., Matthews, M., Bangham, J. A. & Prusinkiewicz, P. The genetics of geometry. Proc. Natl Acad. Sci. USA 101, 4728–4735 (2004).
Wang, J., Lu, D., Mao, D. & Long, M. Mechanomics: an emerging field between biology and biomechanics. Protein Cell 5, 518–531 (2014).
Kutschera, U. & Niklas, K. J. The epidermal-growth-control theory of stem elongation: an old and a new perspective. J. Plant Physiol. 164, 1395–1409 (2007).
Reinhardt, D., Frenz, M., Mandel, T. & Kuhlemeier, C. Microsurgical and laser ablation analysis of leaf positioning and dorsoventral patterning in tomato. Development 132, 15–26 (2005).
McConnell, J. R. & Barton, M. K. Leaf polarity and meristem formation in Arabidopsis. Development 125, 2935–2942 (1998).
Semiarti, E. et al. The ASYMMETRIC LEAVES2 gene of Arabidopsis thaliana regulates formation of a symmetric lamina, establishment of venation and repression of meristem-related homeobox genes in leaves. Development 128, 1771–1783 (2001).
Heisler, M. G. et al. Patterns of auxin transport and gene expression during primordium development revealed by live imaging of the Arabidopsis inflorescence meristem. Curr. Biol. 15, 1899–1911 (2005).
Wang, Y. et al. The stem cell niche in leaf axils is established by auxin and cytokinin in Arabidopsis. Plant Cell 26, 2055–2067 (2014).
Takada, S. & Jurgens, G. Transcriptional regulation of epidermal cell fate in the Arabidopsis embryo. Development 134, 1141–1150 (2007).
Shapiro, B. E., Tobin, C., Mjolsness, E. & Meyerowitz, E. M. Analysis of cell division patterns in the Arabidopsis shoot apical meristem. Proc. Natl Acad. Sci. USA 112, 4815–4820 (2015).
Acknowledgements
We thank K.-I. Hayashi (Okayama University of Science) for providing auxinole, N. Li (Institute of Mechanics, Chinese Academy of Sciences) and Z. Huang (Bruker Nano Surfaces Business, Bei**g) for assistance with AFM measurement, the Core Facilities of Life Sciences of Peking University for use of the TEM and S.-N. Bai (Peking University) and S. Poethig (University of Pennsylvania) for discussions. This work was supported by National Natural Science Foundation of China grants 31430010 and 31627804, National Basic Research Program of China (973 Program) grants 2014CB943500 and 2011CB710900, National Key Research and Development Program of China grant 2016YFA0501601, the National Program for Support of Top-Notch Young Professionals, China Postdoctoral Science Foundation grant 2015M570171 and the State Key Laboratory of Plant Genomics.
Author information
Authors and Affiliations
Contributions
Y.J. conceived and designed experiments. J.Q. and B.W. carried out most of the experiments. S.F., S.L. and M.L. carried out numerical simulations. C.G. contributed to phenotypic analysis. X.Z. and D.Q. contributed to AFM experiments. Y.H. performed TEM experiments. Y.Z. and C.L. provided materials/reagents. Y.J. and M.L. wrote the manuscript, with contributions from all the authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1–11, Supplementary Table 1, Supplementary Table 3, Supplementary Table 4, Supplementary Video Legends, Supplementary Methods, Supplementary References.
Supplementary Table 2
Raw AFM measurements for Figure 1 and Supplementary Figures 4–6.
Supplementary Source Code
Supplementary source code.
Supplementary Video 1
Normal leaf growth, related to Figure 2i–l.
Supplementary Video 2
Hastened adaxial cell wall loosening leads to reduced asymmetry, related to Figure 3c.
Supplementary Video 3
Two-domain partition leads to reduced asymmetry, related to Figure 3d.
Supplementary Video 4
Reduced epidermal restriction leads to reduced asymmetry, related to Figure 6a.
Supplementary Video 5
Enhanced epidermal restriction leads to reduced asymmetry, related to Figure 6b.
Rights and permissions
About this article
Cite this article
Qi, J., Wu, B., Feng, S. et al. Mechanical regulation of organ asymmetry in leaves. Nature Plants 3, 724–733 (2017). https://doi.org/10.1038/s41477-017-0008-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-017-0008-6
- Springer Nature Limited
This article is cited by
-
MorphoSim: an efficient and scalable phase-field framework for accurately simulating multicellular morphologies
npj Systems Biology and Applications (2023)
-
The evolving views of the simplest pectic polysaccharides: homogalacturonan
Plant Cell Reports (2022)
-
Identification of the PmWEEP locus controlling wee** traits in Prunus mume through an integrated genome-wide association study and quantitative trait locus map**
Horticulture Research (2021)
-
An on-demand plant-based actuator created using conformable electrodes
Nature Electronics (2021)
-
Plant multiscale networks: charting plant connectivity by multi-level analysis and imaging techniques
Science China Life Sciences (2021)