Abstract
Molecular ferroelectrics are attracting great interest due to their light weight, mechanical flexibility, low cost, ease of processing and environmental friendliness. These advantages make molecular ferroelectrics viable alternatives or supplements to inorganic ceramics and polymer ferroelectrics. It is expected that molecular ferroelectrics with good performance can be fabricated, which in turns calls for effective chemical design strategies in crystal engineering. To achieve so, we propose a hydrogen bond modification method by introducing the hydroxyl group, and successfully boost the phase transition temperature (Tc) by at least 336 K. As a result, the molecular ferroelectric 1-hydroxy-3-adamantanammonium tetrafluoroborate [(HaaOH)BF4] can maintain ferroelectricity until 528 K, a Tc value much larger than that of BTO (390 K). Meanwhile, micro-domain patterns, in stable state for 2 years, can be directly written on the film of (HaaOH)BF4. In this respect, hydrogen bond modification is a feasible and effective strategy for designing molecular ferroelectrics with high Tc and stable ferroelectric domains. Such an organic molecule with varied modification sites and the precise crystal engineering can provide an efficient route to enrich high-Tc ferroelectrics with various physical properties.
Similar content being viewed by others
Introduction
Due to the unique feature of switchable spontaneous polarization, ferroelectric are widely used in industrial and commercial applications, such as ferroelectric random access memories, piezoelectric sonar, sensors, and electromechanical transformers1,2,3,4. A great majority of excellent and advanced ferroelectrics are based on inorganic ceramics and polymers, like BaTiO3 (BTO), Pb(Zr,Ti)O3 (PZT), polyvinylidene fluoride, etc.5,6,7,8,9. In recent years, molecular ferroelectrics emerge to be a focus of research, with the advantages of mechanical flexibility, lightweight, environmental friendliness, low cost, and ease of processing into films. Over a century has passed since the discovery of the molecular ferroelectric Rochelle salt. Recently, abundant progress has been made in molecular ferroelectrics due to the convenient modification and design of crystal engineering3). The typical ferroelectric J–V curve shows two opposite current peaks. According to the J–V curve, we obtained the perfect P–V hysteresis loop by integrating the current. With an applied voltage, the Ps value rapidly increases and reaches the maximum value of about 4.1 μC cm−2 for thin film, which is close to the estimated value of 4.21 μC cm−2 depending on the Berry phase method. The hysteresis loop is a significant feature of ferroelectric molecules, which proves (HaaOH)BF4 as a ferroelectric. In this respect, the modification of hydrogen bond is an effective strategy to successfully introduce ferroelectric polarization.
Ferroelectricity characterization was carried out on the film of (HaaOH)BF4 via the piezoresponse force microscopy (PFM) technology, realizing the polarization switching at micro and nano scales. We used the probe to scan across the surface of the thin film under the contact mode while applying an ac voltage to the ferroelectric samples simultaneously. Then polarization-dependent deformation occurred to the samples under the ac voltage due to the inverse piezoelectric effect. This can produce a nondestructive visualization of ferroelectric domains with ultra-high spatial resolution and help obtain the polarization information of these ferroelectric samples. PFM contains both lateral (LPFM) and vertical (VPFM) modes, corresponding to the in-plane and out-of-plane polarization components, respectively. According to the intensity and orientation of polarization components, the PFM amplitude and phase data can be obtained, respectively. Using the BFDH method, the crystal plane with dominant growth directions is predicted to be (110), and the polarization of (HaaOH)BF4 is distributed in an in-plane way along the c-axis. Therefore, the domain structure of the (HaaOH)BF4 film was detected by LPFM. Figure 4a, b show the as-grown domain structure of the (HaaOH)BF4 film. The PFM phase imaging (Fig. 4b) shows a clear domain structure with a 180° contrast, due to the different polarization directions on both sides of the PFM probe. As depicted in Fig. 4a, the PFM amplitude imaging shows clear domain walls, that is, the boundary between two domains, which conform to the domain structure of PFM phase imaging. The domain wall displays the lowest amplitude signals. Since the amplitude signals of different domains have no obvious differences, the domains show a 180° polarization distribution. This also indicates that (HaaOH)BF4 may be a uniaxial molecular ferroelectric. Besides, the clear distribution of as-grown domains has no correlation with the surface morphology of the thin film (Supplementary Fig. 12), demonstrating that (HaaOH)BF4 possesses spontaneous polarization in different directions.
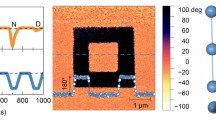
The lateral phase (a) and lateral amplitude (b) of the pristine domain on the as-grown thin film of (HaaOH)BF4 are displayed. The final state of LPFM amplitude (c), phase (d), and topography (e) images for the 90 × 90 μm2 region were observed after Vdc was applied in the order of +110 V and −110 V twice to switch fourfold box-in-box domains. f Phase and amplitude signals as functions of the tip voltage for a selected point in the off-field period, showing local PFM hysteresis loops.
In addition to the as-grown domains and domain walls, ferroelectrics are also characteristic of reversible polarization under an applied electric field. Thanks to the switchable polarization, arbitrary micron patterns of domains can be written on ferroelectrics. Based on this, we characterized the local polarization switching behavior on the (HaaOH)BF4 thin film through switching spectroscopy PFM (SSPFM) measurement. The reversal of polarization was recorded off-field by PFM phase and amplitude signals when the dc voltage (Vdc), a triangular pulse square wave, and superimposed ac voltage were applied to the film through the conductive tip (Fig. 4f). Evidently, the PFM phase curve shows a square-shaped hysteresis loop with a 180° contrast under the electric field, and the PFM amplitude curve shows a typical butterfly shape. These curves indicate the switching and hysteresis behaviors of ferroelectric polarization in (HaaOH)BF4, which are obtained in the off-field period. Meanwhile, temperature-dependent SSPFM measurements indicate that the required voltages for polarization switching decrease as the temperature increases and the polarization switching still occurs at 473 K (Supplementary Fig. 13). Furthermore, we selected a 90×90 μm2 region and applied the dc bias to the (HaaOH)BF4 crystal film through the PFM probe to achieve clear domain switching. The selected region on the film is in initial single-domain state (Supplementary Fig. 14). With the PFM probe, Vdc was applied to a selected rectangular region that became smaller and smaller, in the order of +110 V and −110 V twice. The domain in the same region of the film was switched four times under the applied bias at room temperature. Supplementary Fig. 14 vividly shows the meticulous PFM imaging of each polarization reversal process. Evidently, a significant 180° PFM phase contrast exists between different domains, and adjacent domains are separated by clear domain walls with weak amplitude signals. This proves that the polarization in the (HaaOH)BF4 film can be reversed back and forth, conforming to the principle of ferroelectric polarization. Figure 4c, d displays the fourfold box-in-box domain structures in the final state, which are almost the same as the preset ones. Because the new domain nucleation energy of (HaaOH)BF4 is lower than the domain growth energy, the domain is easier to nucleate and difficult to diffuse. No obvious change is observed in the morphology of the scanning region before and after domain switching, as shown in Supplementary Fig. 14. And the domain structure written by the electric field can remain stable until 443 K (Supplementary Fig. 15) and for over 2 years at room temperature (Supplementary Figs. 15 and 16). Meanwhile, the (HaaOH)BF4 film can still be switched under the external electric field at the temperature of 413 K (Supplementary Fig. 17). This rebuts the assumption that domain switching of (HaaOH)BF4 is caused by charge injection, and further corroborates its intrinsic ferroelectric switching. Furthermore, the written box-in-box domain pattern proves that (HaaOH)BF4 has switchable spontaneous polarization and can conduct the stable domain configuration on the micron scale.
The resonant PFM mode is widely used for the electromechanical coupling of ferroelectric materials, which detects the surface deformation excited by the electric field17,20. With PFM, the local piezoresponse of (HaaOH)BF4 and (Haa)BF4 thin films can be obtained. The two samples were both driven at the cantilever-sample resonance frequency of 10 V. The amplitude signal obtained is matched with the Damped Simple Harmonic Oscillator model (Supplementary Fig. 18), and the inherent piezoelectric response can be obtained through quality factor correction and resonant amplification. Supplementary Fig. 18 shows the obvious piezoelectric response of (HaaOH)BF4 and a positive linear relationship between the inherent piezoelectric and the driving voltage. On the contrary, (Haa)BF4 has no piezoelectric response along different crystal axis directions as expected (Supplementary Fig. 19). The piezoelectric coefficient d33 along the corresponding polarization direction of (HaaOH)BF4 is 22 pC N−1, according to the quasi-static method (Berlincourt method) (Fig. 5a). And the value of g33 can be evaluated through the formula of g33 = d33/ɛ33, in which the dielectric permittivity ɛ33 can be derived from ɛ’ = ɛ33/ɛ0 (ɛ’ = 21). Based on the results of d33 and ɛ’ (Supplementary Fig. 11, 19), the g33 of (HaaOH)BF4 is about 165.7 × 10−3 Vm N−1, which is higher than that of PZT-based piezoelectric ceramics (about 20 to 40 × 10−3 Vm N−1).
a Diagram of the piezoelectric coefficient d33 of the (HaaOH)BF4 crystal along the [001] direction using the quasi-static method. b Schematic illustration of piezoelectric energy-harvesting devices. c Generated VOC of energy-harvesting devices based on (HaaOH)BF4 and (Haa)BF4 polycrystalline samples under a periodical vertical force of 17 N at a frequency of 10 Hz, and the VOC of the device without samples under the same conditions. d VOC of the (HaaOH)BF4 energy-harvesting device under forward (blue line) and reverse (red line) electrical connections. e VOC of the energy-harvesting device containing (HaaOH)BF4 with different periodical vertical forces applied (3, 13, 22, and 31 N) at a frequency of 10 Hz. f Linear fitting of the VOC of the (HaaOH)BF4 energy-harvesting device as a function of the applied force.
To exploit the piezoelectric response with the energy-harvesting capability of (HaaOH)BF4, we fabricated a device with the structure of electrode-(HaaOH)BF4-electrode through the package of polydimethylsiloxane (PDMS) (Fig. 5b). The device exhibited an open-circuit voltage (VOC) of about 3.5 V under a 10 Hz periodic vertical pressure of 17 N. Under the same condition, the blank PDMS device and (Haa)BF4 device were tested, whose VOC were close to 0 and were lower than (HaaOH)BF4 device (Fig. 5c). This indicates that the VOC of the (HaaOH)BF4 device is induced by piezoelectricity. Similarly, switching-polarity tests were conducted to verify that the generated output signals indeed originate from the piezoelectric phenomenon. The reversal polarization test realized by electronic reverse connection also produces corresponding reverse transformation of VOC (Fig. 5d). The reversible electrical signals indicated that the detected outputs were generated by the compression-induced strain of the (HaaOH)BF4 devices37. And this rules out the possibility that VOC comes from the change of system capacitance38. Additionally, voltage peak values between compressing and releasing conditions were different and asymmetric. This can be explained by variations in the strain rate during the application and removal of stress on the devices39. It is obvious that the VOC of the (HaaOH)BF4 device increases gradually with the pressure rising from 3 N to 31 N at a stable frequency (Fig. 5e). Figure 5f displays a good linear relationship between the pressure and VOC. Meanwhile, the electrical VOC of the device were measured under various force frequencies (Supplementary Fig. 20). The generated electrical output performance is noticeable and capable of responding to different external force frequencies. The output current of the (HaaOH)BF4 device was measured under a pressure of 20 N, exhibiting a maximum of ~0.31 μA (Supplementary Fig. 21). And the piezoelectric device of (HaaOH)BF4 can realize long-term sensing with the voltage maintained for ~5 V after at least 7000 cycles (Supplementary Fig. 22). Furthermore, the output signals of the (HaaOH)BF4 device at high temperatures have also been verified, which maintained the output voltages of around 2 V at 503 K (Supplementary Fig. 23). This is consistent with the temperature-dependent piezoresponse measurements, which indicate that the sample can maintain piezoelectricity until Td (Supplementary Fig. 24). This indicates the potential of (HaaOH)BF4 device for monitoring the working status and vibrations of mechanical components in high-temperature environments. Evidently, the design strategy of hydrogen bond modification can successfully build intermolecular force networks in molecular materials to achieve an orderly molecule arrangement and introduce polarity. And the molecular ferroelectric (HaaOH)BF4, with a high Td and stable domains, will not only promote the conversion of mental-free materials into electromechanical converters but also be a promising source for flexible electronic devices.
Due to insufficient development in power and sensing technologies, operating long-duration missions in unstructured environments remain a difficult task for robots40,41. Non-metallic (HaaOH)BF4, possessing good mechanical-to-current conversion capabilities, can be potentially applied to robots to achieve mechanical energy harvesting and self-powered tactile sensing under external mechanical stimuli. Additionally, the output performance of the device was measured by connecting various external load resistors. The output voltage gradually rose as the external load resistance was increased, while the output current decreased (Supplementary Fig. 25a). The obtained maximum output power density was approximately 1.2 μW cm−2 at a load resistance of 4 × 107 Ω (Supplementary Fig. 25b). In our proof-of-concept study, the (HaaOH)BF4 device lit 9 blue LEDs (3.0-3.2 V, 60–64 mW) under the periodic mechanical tap** without using any external power unit (such as a capacitor) (Fig. 6a and Supplementary Movie 1). In addition, by housing the (HaaOH)BF4 device directly on the robot’s surface, it is readily available to detect external mechanical stimuli or collisions. The energy harvester consists of the top conductive adhesive tape, (HaaOH)BF4, and the bottom conductive adhesive tape, encapsulated by PDMS. The top conductive adhesive tape is electrically grounded (Supplementary Fig. 26). Through this configuration, charges originating from the human body and those induced by the triboelectric effect between the human body and the device can dissipate into the ground. Consequently, the impact of additional charges, apart from the piezoelectric effect, is eliminated. As shown in Fig. 6b, the output piezoelectric response signals vary a lot according to different tap** forces (Supplementary Movie 2), although light tap** can still produce distinct signal responses. Meanwhile, the piezoelectric sensor responded to the pressure quickly, with a fast response time of 10.24 ms under a pressure upon finger tap** (Supplementary Fig. 27). The two applications further prove non-metallic (HaaOH)BF4-based devices as reliable applicants for robots equipped with energy harvesting and mechanical stimuli sensing.
In summary, we successfully designed the non-ferroelectric (Haa)BF4 as the molecular ferroelectric (HaaOH)BF4 through hydrogen bond modification. The modification of the hydroxyl group has not only introduced ferroelectric polarization but also promoted Tc of (HaaOH)BF4 by at least 336 K. By analyzing the intermolecular force of two compounds’ structure, we found that (HaaOH)BF4 obtained more H⋯O hydrogen bonds and guest-guest interactions. Due to the lattice intermolecular force formed in the crystal, (HaaOH)BF4 has a higher Tc, which is even higher than its Td (528 K). And the ferroelectricity of (HaaOH)BF4 can be maintained until Td, which is close to the high Tc (534 K) as reported among molecular ferroelectrics. This proves hydrogen bond modification as an effective strategy for designing molecular ferroelectrics, stabling ferroelectric domain structure, and optimizing the phase transition temperature. Meanwhile, the fabrication of adamantane with so many modification sites can hopefully optimize the properties of molecular ferroelectrics. Furthermore, stable micron ferroelectric domain structures have been constructed on the film of molecular ferroelectric (HaaOH)BF4. These structures can retain stable until 443 K and for over 2 years at room temperature. The piezoelectric properties of the flexible sample were detected by the energy-harvesting device capable of mechanical energy harvesting and self-powered sensing. The precise molecular design strategy and crystal engineering are crucial to further optimize and promote the development of molecular ferroelectrics. Such an organic molecule with a variety of modification sites also provides opportunities and platforms to enhance modern energies and develop micro-nano electronic devices.
Methods
Materials
Synthesis of single crystals
All reagents and solvents in the syntheses were of reagent grade and used without further purification. Slight excess of tetrafluoroboric acid (48 wt. % in H2O, 20,12 g, 0.11 mol) and proper amount of deionized water (100 mL) was added to the beaker, and then adamantan-1-amine (15.13 g, 0.1 mol) or 3-amino-1-adamantanol (16.73 g, 0.1 mol) was added to the solution and stirred for 20 min at room temperature. Transparent and colorless crystal of 1-adamantanammonium tetrafluoroborate [(Haa)BF4] or 1-hydroxy-3-adamantanammonium tetrafluoroborate [(HaaOH)BF4] can be obtained by slowly evaporating at room temperature.
Thin film preparation
The thin films of [(Haa)BF4] and [(HaaOH)BF4] were prepared by drop-coating approach on ITO/glass substrate. The deionized aqueous solutions of [(Haa)BF4] and [(HaaOH)BF4] with 0.08 g/ml concentration were prepared. And then 15 μL of the solution was dropped on 1 × 1 μm2 ITO/glass at 353 K. The transparent films can be obtained after the solution has evaporated.
Measurements
Methods of single-crystal X-ray crystallography, powder X-ray diffraction, differential scanning calorimetry, thermogravimetric analysis, dielectric measurements, ferroelectric hysteresis loop measurements, piezoresponse force microscopy characterization, piezoelectric coefficient measurements, preparation and measurements of piezoelectric energy-harvesting devices, calculate condition, Hershefield surface analysis and energy framework analysis were described in the Supplementary Information.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data generated and analyzed in this study are included in the Article and its Supplementary Information. And all relevant data that support the findings of this study are available from the corresponding authors upon request. The crystal structures generated in this study have been deposited in the Cambridge Crystallographic Data Centre under accession code CCDC: 2192545, 2192429, and 2314039. The data can be obtained free of charge via www.ccdc.cam.ac.uk/data_request/cif, or by emailing data_request@ccdc.cam.ac.uk, or by contacting The Cambridge Crystallographic Data Centre, 12 Union Road, Cambridge CB2 1EZ, UK; fax: +44 1223 336033.
References
Lines M. E., Glass A. M. Principles and Applications of Ferroelectrics and Related Materials.(Oxford Univ. Press, 2001).
Bain A. K., Chand P. Ferroelectrics: Principles and Applications (John Wiley & Sons, 2017).
Scott, J. F. Applications of modern ferroelectrics. Science 315, 954–959 (2007).
Dragan, D. Ferroelectric, dielectric and piezoelectric properties of ferroelectric thin films and ceramics. Rep. Prog. Phys. 61, 1267 (1998).
Liu, Y. et al. Ferroelectric polymers exhibiting behaviour reminiscent of a morphotropic phase boundary. Nature 562, 96–100 (2018).
Haertling, G. H. Ferroelectric ceramics: history and technology. J. Am. Ceram. Soc. 82, 797–818 (1999).
Li, F. et al. Ultrahigh piezoelectricity in ferroelectric ceramics by design. Nat. Mater. 17, 349–354 (2018).
Lovinger, A. J. Ferroelectric polymers. Science 220, 1115–1121 (1983).
Qiu, C. et al. Transparent ferroelectric crystals with ultrahigh piezoelectricity. Nature 577, 350–354 (2020).
Whatmore, R. W., You, Y.-M., **ong, R.-G. & Eom, C.-B. 100 years of ferroelectricity—a celebration. APL Mater. 9, 070401 (2021).
Shi, P.-P. et al. Symmetry breaking in molecular ferroelectrics. Chem. Soc. Rev. 45, 3811–3827 (2016).
Liu, H.-Y., Zhang, H.-Y., Chen, X.-G. & **ong, R.-G. Molecular design principles for ferroelectrics: ferroelectrochemistry. J. Am. Chem. Soc. 142, 15205–15218 (2020).
Pan, Q., **ong, Y.-A., Sha, T.-T. & You, Y.-M. Recent progress in the piezoelectricity of molecular ferroelectrics. Mater. Chem. Front. 5, 44–59 (2021).
**ong, Y.-A. et al. Recent progress in molecular ferroelectrics with perovskite structure. Chin. Sci. Bull. 65, 916–930 (2020).
Fu, D.-W. et al. Diisopropylammonium bromide is a high-temperature molecular ferroelectric crystal. Science 339, 425–428 (2013).
Ai, Y., Zeng, Y.-L., He, W.-H., Huang, X.-Q. & Tang, Y.-Y. Six-fold vertices in a single-component organic ferroelectric with most equivalent polarization directions. J. Am. Chem. Soc. 142, 13989–13995 (2020).
You, Y.-M. et al. An organic-inorganic perovskite ferroelectric with large piezoelectric response. Science 357, 306–309 (2017).
Lv, H.-P., Liao, W.-Q., You, Y.-M. & **ong, R.-G. Inch-size molecular ferroelectric crystal with a large electromechanical coupling factor on par with barium titanate. J. Am. Chem. Soc. 144, 22325–22331 (2022).
Wang, B. et al. Achievement of a giant piezoelectric coefficient and piezoelectric voltage coefficient through plastic molecular-based ferroelectric materials. Matter 5, 1296–1304 (2022).
Liao, W.-Q. et al. A molecular perovskite solid solution with piezoelectricity stronger than lead zirconate titanate. Science 363, 1206–1210 (2019).
Yao, J. et al. Hybrid organic–inorganic perovskite ferroelectrics bring light to semiconducting applications: Bandgap engineering as a starting point. APL Mater. 9, 040901 (2021).
Ye, H.-Y. et al. Bandgap engineering of lead-halide perovskite-type ferroelectrics. Adv. Mater. 28, 2579–2586 (2016).
Zhang, H.-Y. et al. Observation of vortex domains in a two-dimensional lead iodide perovskite ferroelectric. J. Am. Chem. Soc. 142, 4925–4931 (2020).
Tang, Y.-Y. et al. Organic ferroelectric vortex–antivortex domain structure. J. Am. Chem. Soc. 142, 21932–21937 (2020).
Song X.-J. et al. The first demonstration of strain-controlled periodic ferroelectric domains with superior piezoelectric response in molecular materials. Adv. Mater. 35, 2211584 (2023).
**ong, Y.-A. et al. Rational design of molecular ferroelectrics with negatively charged domain walls. J. Am. Chem. Soc. 144, 13806–13814 (2022).
Li, P.-F. et al. Organic enantiomeric high-Tc ferroelectrics. Proc. Natl Acad. Sci. USA 116, 5878–5885 (2019).
Li, P.-F. et al. Anomalously rotary polarization discovered in homochiral organic ferroelectrics. Nat. Commun. 7, 13635 (2016).
**ong, Y.-A. et al. A nickel(II) nitrite based molecular perovskite ferroelectric. Angew. Chem. Int. Ed. 58, 8857–8861 (2019).
Zhang, H.-Y. et al. Large electrostrictive coefficient in a two-dimensional hybrid perovskite ferroelectric. J. Am. Chem. Soc. 143, 1664–1672 (2021).
King-Smith, R. D. & Vanderbilt, D. Theory of polarization of crystalline solids. Phys. Rev. B 47, 1651–1654 (1993).
Vanderbilt, D. & King-Smith, R. D. Electric polarization as a bulk quantity and its relation to surface charge. Phys. Rev. B 48, 4442–4455 (1993).
Spackman, M. A. & Jayatilaka, D. Hirshfeld surface analysis. CrystEngComm 11, 19–32 (2009).
Turner, M. J., Grabowsky, S., Jayatilaka, D. & Spackman, M. A. Accurate and efficient model energies for exploring intermolecular interactions in molecular crystals. J. Phys. Chem. Lett. 5, 4249–4255 (2014).
Turner, M. J., Thomas, S. P., Shi, M. W., Jayatilaka, D. & Spackman, M. A. Energy frameworks: insights into interaction anisotropy and the mechanical properties of molecular crystals. Chem. Commun. 51, 3735–3738 (2015).
Song, X.-J. et al. Record enhancement of curie temperature in host–guest inclusion ferroelectrics. J. Am. Chem. Soc. 143, 5091–5098 (2021).
Ding, R. et al. High-performance piezoelectric nanogenerators composed of formamidinium lead halide perovskite nanoparticles and poly(vinylidene fluoride). Nano Energy 37, 126–135 (2017).
Yang, R., Qin, Y., Li, C., Dai, L. & Wang, Z. L. Characteristics of output voltage and current of integrated nanogenerators. Appl. Phys. Lett. 94, 022905 (2009).
Chen, H. et al. Piezoelectric nanogenerator based on in situ growth all-inorganic CsPbBr3 perovskite nanocrystals in PVDF fibers with long-term stability. Adv. Funct. Mater. 31, 2011073 (2021).
Aubin, C. A. et al. Towards enduring autonomous robots via embodied energy. Nature 602, 393–402 (2022).
Duan, S. et al. Water-modulated biomimetic hyper-attribute-gel electronic skin for robotics and skin-attachable wearables. ACS Nano 17, 1355–1371 (2023).
Acknowledgements
This work was supported by the National Key R&D Program of China (Grant No. 2021YFA1200700 (Y.-M.Y.)), the National Natural Science Foundation of China (Grant No. 21925502 (Y.-M.Y.), and 223B2502 (Y.-A.X.)), Postgraduate Research & Practice Innovation Program of Jiangsu Province (No. KYCX23_0224 (Y.-A.X.)), the China Scholarship Council program (Project ID: 202306090116 (Y.-A.X.)) and “the Fundamental Research Funds for the Central Universities, China”.
Author information
Authors and Affiliations
Contributions
Y.-M.Y. and J.W. conceived and supervised the project. Y.-A.X. prepared the samples and performed the PFM measurements, piezoelectric energy-harvesting devices, and analysis. S.-S.D. prepared and test the power supply and stimuli sensing of piezoelectric devices. H.-H.H. measured the thermodynamic properties and sorted out the article. J.Y. performed the single-crystal measurement and analysis. Q.P. contributed to P–V loop measurements. T.-T.S., X.W., and H.-R.J. participated in the production and performance testing of piezoelectric devices. Y.-M.Y., J.W., Y.-A.X., S.-S.D., and H.-H.H. analyzed the data and results. Y.-A.X., S.-S.D., and H.-H.H. wrote the manuscript with input from all the other authors. All authors discussed the results and contributed to the manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks **angyu Gao, Kanghua Li, and Guifu Zou for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
**ong, YA., Duan, SS., Hu, HH. et al. Enhancement of phase transition temperature through hydrogen bond modification in molecular ferroelectrics. Nat Commun 15, 4470 (2024). https://doi.org/10.1038/s41467-024-48948-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-48948-0
- Springer Nature Limited