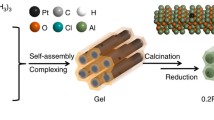
Abstract
The contribution of the reverse spillover effect to hydrogen generation reactions is still controversial. Herein, the promotion functions for reverse spillover in the ammonia borane hydrolysis reaction are proven by constructing a spatially separated NiO/Al2O3/Pt bicomponent catalyst via atomic layer deposition and performing in situ quick X-ray absorption near-edge structure (XANES) characterization. For the NiO/Al2O3/Pt catalyst, NiO and Pt nanoparticles are attached to the outer and inner surfaces of Al2O3 nanotubes, respectively. In situ XANES results reveal that for ammonia borane hydrolysis, the H species generated at NiO sites spill across the support to the Pt sites reversely. The reverse spillover effects account for enhanced H2 generation rates for NiO/Al2O3/Pt. For the CoOx/Al2O3/Pt and NiO/TiO2/Pt catalysts, reverse spillover effects are also confirmed. We believe that an in-depth understanding of the reverse effects will be helpful to clarify the catalytic mechanisms and provide a guide for designing highly efficient catalysts for hydrogen generation reactions.
Similar content being viewed by others
Introduction
The ever-increasing global energy demand and the detrimental effect of the CO2 product of fossil fuels have triggered a widespread search for alternative energy sources, which are effective and renewable and do not cause further environmental issues1. Because of its high energy density and renewability, H2 has been regarded as an attractive green fuel and a promising energy carrier for the future to meet increasing energy and environmental challenges2. Catalytic H2 generation from hydrogen storage materials is considered a potential method of H2 production if they can be effectively catalyzed3,4. The search for efficient catalytic systems would be greatly facilitated by a clearer understanding of the underlying chemical process.
Noble metal catalysts, such as Pt, Pd, and Ru, have been recognized as important classes of catalysts for hydrogen generation, due to their high catalytic activity and durability5,6,7,8,9,10,11. It is noted that coupling metal catalysts with secondary metals12,13,14,15 and/or transition metal oxides16,17,18,19,20,21,22,23,24,25 is an encouraging strategy to further enhance catalytic performance. In the past, various theories (e.g., the metal-oxide interfacial sites, electron interactions, or hydrogen reverse spillover effect) have been offered to explain the enhancement of H2 generation when different components are combined in a catalyst. For example, Francisco Zaera and coworkers argued that in the photocatalytic production of H2 from water with semiconductor catalysts, the role of metal additives is a reverse spillover effect, not to trap excited electrons26. Hydrogen reverse spillover, as a form of spillover, involves the migration of adsorbed hydrogen atoms from an oxide (or other nonmetal surface) to a metal, where they recombine to molecular hydrogen27,28,29. However, due to the lack of well-defined catalysts with clearly separated functional components and the difficulties in performing in situ characterization technologies, researchers have not formed an agreement on the enhancement mechanism. It is still a challenging issue to reveal the promotion effects of reverse spillover in H2 generation reactions.
In this work, taking the ammonia borane (NH3·BH3, AB) hydrolysis reaction as an example, the promotion functions of reverse spillover in this reaction are proven using a spatially separated NiO/Al2O3/Pt catalyst as a model catalyst, in combination with in situ quick XANES characterization. The NiO/Al2O3/Pt catalyst was prepared by a facile and general template-assisted atomic layer deposition (ALD) method30,31,1d, e) for NiO/Al2O3/Pt show that Ni and Pt are distributed on the outer and inner surfaces of Al2O3 nanotubes, respectively. The STEM image, EDX map**, and line-scanning profile for a cross-sectional specimen prepared by focused ion beam milling along the vertical direction of the Al2O3 nanotubes (Supplementary Fig. 3) further demonstrate the separated structure of NiO/Al2O3/Pt. TEM images of the Al2O3/Pt and NiO/Al2O3 catalysts are shown in Fig. 1b, c. There are no Pt particles on the outer surfaces of Al2O3 nanotubes for Al2O3/Pt (Supplementary Fig. 4). Due to the small size and low contrast of NiO nanoparticles, it is not straightforward to distinguish NiO nanoparticles in Fig. 1a, c. From the HRTEM image of NiO/Al2O3 (inset in Fig. 1c), NiO nanoparticles can be clearly observed. The Pt content in the catalysts was measured using inductively coupled plasma-atomic emission spectrometry (ICP-AES) to be 3.65 and 4.23% for NiO/Al2O3/Pt and Al2O3/Pt, and the Ni content was measured to be 8.05 and 8.71% for NiO/Al2O3/Pt and NiO/Al2O3, respectively. The N2 sorption isotherms for the NiO/Al2O3, Al2O3/Pt, and NiO/Al2O3/Pt catalysts almost overlap (Fig. 1f). The Brunauer–Emmett–Teller (BET) surface areas for the NiO/Al2O3, Al2O3/Pt, and NiO/Al2O3/Pt catalysts were calculated to be 95.4, 93.6, and 98.0 m2 g–1, respectively. Their pore volumes were 0.34, 0.34, and 0.39 cm3 g–1, respectively. The Barrett–Joiner–Halenda (BJH) pore size distribution curves (Fig. 1g) deduced from desorption branches of the N2 sorption isotherms confirm that NiO/Al2O3, Al2O3/Pt, and NiO/Al2O3/Pt samples are made up of pores with average sizes centred at 54.9, 54.4, and 57.0 nm, respectively. The pore sizes of these catalysts, i.e., the inner diameters of the Al2O3 nanotubes, correspond to the diameters of the CNC sacrificial templates. These results show that all the catalysts possess similar pore structures.
TEM images of a NiO/Al2O3/Pt, b Al2O3/Pt, and c NiO/Al2O3 catalysts. Inset in c shows a HRTEM image of NiO/Al2O3. d HAADF-STEM image and e EDX elemental map** of the NiO/Al2O3/Pt catalyst. f N2 adsorption−desorption isotherms and g the corresponding pore size distributions of the catalysts. h XPS Ni 2p analysis of NiO/Al2O3 and NiO/Al2O3/Pt. i H2-TPR profiles of NiO/Al2O3, Al2O3/Pt, and NiO/Al2O3/Pt.
The X-ray photoelectron spectroscopy (XPS) results reveal the existence of Ni2+ species in NiO/Al2O3 and NiO/Al2O3/Pt (Fig. 1h). The XPS peaks for the two catalysts are similar. The XPS peaks located at binding energies of 856.1 and 874.0 eV are attributed to Ni 2p3/2 and Ni 2p1/2, respectively, and the peaks located at binding energies of 861.8 and 879.7 eV are attributed to satellite peaks. From the X-ray diffraction (XRD) patterns for the Al2O3/Pt and NiO/Al2O3/Pt catalysts (Supplementary Fig. 5), the presence of Pt nanoparticles can be confirmed. No diffraction peak assigned to NiO is detected from the XRD patterns for NiO/Al2O3 and NiO/Al2O3/Pt, which can be ascribed to the high dispersion of ALD-prepared nanoparticles. Hydrogen temperature programmed reduction (H2-TPR) was used to study the redox properties of the catalysts (Fig. 1i). The profile obtained for Al2O3/Pt displays a principal reduction peak at 385 °C, which can be attributed to Pt interacting with Al2O337. The NiO/Al2O3 catalyst exhibits a small shoulder peak at approximately 349 °C and a strong peak centred at 431 °C, corresponding to the reductions of bulk NiO and the NiO interacting with Al2O3. In contrast, for NiO/Al2O3/Pt, the first H2 consumption peak (corresponding to the reduction of bulk NiO) shifts from 349 to 326 °C and becomes obvious, and a broadened peak centred at 408 °C (corresponding to the reductions of Pt and NiO interacting with Al2O3) can be observed. Quantification of the H2-TPR curves (Supplementary Table 1) shows that the hydrogen consumed by NiO/Al2O3/Pt (1.86 mmol H2 g–1) is greater than the sum of the hydrogen consumed by Al2O3/Pt (0.31 mmol H2 g–1) and NiO/Al2O3 (1.29 mmol H2 g–1). These results demonstrate that the reduction of NiO species is promoted after Pt addition, which can be attributed to the hydrogen spillover effect38,39,56. The crystal structure of γ-Al2O3 proposed by Gutiérrez et al.57 was adopted in our model system. The most stable (100) surface of γ-Al2O3 with three alumina layers and a Ni4O4 cluster adsorbed onto it were used for the NiO/γ-Al2O3(100) slab model. The two bottom layers of the slab were kept fixed. The thickness of the vacuum region was 20 Å. A Monkhorst-Pack grid was used for Brillouin-zone integrations with 1 × 1 × 1 k-mesh (gamma point) sampling. The solvation effect was included with an implicit solvation solvent of water using the VASPsol tool58. The free energies at room temperature (298.15 K) were obtained by adding to the DFT electronic energy (E), the zero-point energy, enthalpy, and entropy contribution from the vibrational modes. The transition states (TS) were calculated using the climbing image nudged elastic band method59, and frequency analysis was confirmed to verify the TS.
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Suh, M. P., Park, H. J., Prasad, T. K. & Lim, D. W. Hydrogen storage in metal-organic frameworks. Chem. Rev. 112, 782–835 (2012).
Dresselhaus, M. S. & Thomas, I. L. Alternative energy technologies. Nature 414, 332–337 (2001).
Sun, Q., Wang, N., Xu, Q. & Yu, J. Nanopore-supported metal nanocatalysts for efficient hydrogen generation from liquid-phase chemical hydrogen storage materials. Adv. Mater. 32, 2001818 (2020).
Schlapbach, L. & Züttel, A. Hydrogen-storage materials for mobile applications. Nature 414, 353–358 (2001).
Chen, W. et al. Mechanistic insight into size-dependent activity and durability in Pt/CNT catalyzed hydrolytic dehydrogenation of ammonia borane. J. Am. Chem. Soc. 136, 16736–16739 (2014).
Yang, J. et al. Atomic design and fine-tuning of subnanometric Pt catalysts to tame hydrogen generation. ACS Catal. 11, 4146–4156 (2021).
Bi, Q. et al. Dehydrogenation of formic acid at room temperature: boosting palladium nanoparticle efficiency by coupling with pyridinic-nitrogen-doped carbon. Angew. Chem. Int. Ed. 55, 11849–11853 (2016).
Tedsree, K. et al. Hydrogen production from formic acid decomposition at room temperature using a Ag−Pd core-shell nanocatalyst. Nat. Nanotechnol. 6, 302–307 (2011).
Wang, N. et al. In situ confinement of ultrasmall Pd clusters within nanosized silicalite-1 zeolite for highly efficient catalysis of hydrogen generation. J. Am. Chem. Soc. 138, 7484–7487 (2016).
Liu, Y. et al. Regulating electron–hole separation to promote photocatalytic H2 evolution activity of nanoconfined Ru/MXene/TiO2 catalysts. ACS Nano 14, 14181–14189 (2020).
Li, W. et al. Exploiting Ru-induced lattice strain in CoRu nanoalloys for robust bifunctional hydrogen production. Angew. Chem. Int. Ed. 60, 3290–3298 (2021).
Fu, F. et al. Highly selective and sharp volcano-type synergistic Ni2Pt@ZIF-8-catalyzed hydrogen evolution from ammonia borane hydrolysis. J. Am. Chem. Soc. 140, 10034–10042 (2018).
Karaca, T., Sevim, M. & Metin, Ö. Facile synthesis of monodisperse copper–platinum alloy nanoparticles and their superb catalysis in the hydrolytic dehydrogenation of ammonia borane and hydrazine borane. ChemCatChem 9, 4185–4190 (2017).
Singh, A. K. & Xu, Q. Synergistic catalysis over bimetallic alloy nanoparticles. ChemCatChem 5, 652–676 (2013).
Wang, S. et al. Aqueous solution synthesis of Pt–M (M = Fe, Co, Ni) bimetallic nanoparticles and their catalysis for the hydrolytic dehydrogenation of ammonia borane. ACS Appl. Mater. Interfaces 6, 12429–12435 (2014).
Li, J. et al. Highly active and stable metal single-atom catalysts achieved by strong electronic metal–support interactions. J. Am. Chem. Soc. 141, 14515–14519 (2019).
Chen, W. et al. Synergistic Pt-WO3 dual active sites to boost hydrogen production from ammonia borane. iScience 23, 100922 (2020).
Ren, X. et al. Promoting effect of heterostructured NiO/Ni on Pt nanocatalysts toward catalytic hydrolysis of ammonia borane. J. Phys. Chem. Lett. 10, 7374–7382 (2019).
Zhang, J. et al. Synergistic effects in atomic-layer-deposited PtCox/CNTs catalysts enhancing hydrolytic dehydrogenation of ammonia borane. Appl. Catal. B: Environ. 235, 256–263 (2018).
Ge, Y. et al. PtNi/NiO clusters coated by hollow sillica: novel design for highly efficient hydrogen production from ammonia−borane. ACS Appl. Mater. Interfaces 9, 3749–3756 (2017).
Yao, Q., Shi, Y., Zhang, X., Chen, X. & Lu, Z. Facile synthesis of platinum-cerium(IV) oxide hybrids arched on reduced graphene oxide catalyst in reverse micelles with high activity and durability for hydrolysis of ammonia borane. Chem. Asian J. 11, 3251–3257 (2016).
Chen, J., Lu, Z., Yao, Q., Feng, G. & Luo, Y. Complete dehydrogenation of N2H4BH3 with NiM-Cr2O3(M = Pt, Rh, and Ir) hybrid nanoparticles. J. Mater. Chem. A 6, 20746–20752 (2018).
Xu, D. et al. Pt nanoparticles immobilized in mesoporous silica-coated magnetic nanocapsules: a non-leaching catalyst for hydrogen generation from hydrolysis of ammonia borane. Int. J. Hydrog. Energy 42, 27034–27042 (2017).
Zhou, Q. & Xu, C. Nanoporous PtCo/Co3O4 composites with high catalytic activities toward hydrolytic dehydrogenation of ammonia borane. J. Colloid Interface Sci. 508, 542–550 (2017).
Zhao, B. et al. PtxNi10−xO nanoparticles supported on N-doped graphene oxide with a synergetic effect for highly efficient hydrolysis of ammonia borane. Catal. Sci. Technol. 7, 5135–5142 (2017).
Joo, J. B. et al. Promotion of atomic hydrogen recombination as an alternative to electron trap** for the role of metals in the photocatalytic production of H2. Proc. Natl Acad. Sci. USA 111, 7942–7947 (2014).
Roland, U., Braunschweig, T. & Roessner, F. On the nature of spilt-over hydrogen. J. Mol. Catal. A: Chem. 127, 61–84 (1997).
Conner, W. C. & Falconer, J. L. Spillover in heterogeneous catalysis. Chem. Rev. 95, 759–788 (1995).
Prins, R. Hydrogen spillover. Facts and fiction. Chem. Rev. 112, 2714–2738 (2012).
Gao, Z. & Qin, Y. Design and properties of confined nanocatalysts by atomic layer deposition. Acc. Chem. Res. 50, 2309–2316 (2017).
Zhang, J. et al. Origin of synergistic effects in bicomponent cobalt oxide-platinum catalysts for selective hydrogenation reaction. Nat. Commun. 10, 4166 (2019).
**ong, M. et al. In situ tuning of electronic structure of catalysts using controllable hydrogen spillover for enhanced selectivity. Nat. Commun. 11, 4773 (2020).
Liu, X. et al. Activation of subnanometric Pt on Cu-modified CeO2 via redox-coupled atomic layer deposition for CO oxidation. Nat. Commun. 11, 4240 (2020).
Lei, Y., Lee, S., Low, K. B., Marshall, C. L. & Elam, J. W. Combining electronic and geometric effects of ZnO-promoted Pt nanocatalysts for aqueous phase reforming of 1-propanol. ACS Catal. 6, 3457–3460 (2016).
Zhang, H. & Marshall, C. L. Atomic layer deposition: catalytic preparation and modification technique for the next generation. Chin. J. Catal. 40, 1311–1323 (2019).
Marichy, C. & Pinna, N. Atomic layer deposition to materials for gas sensing applications. Adv. Mater. Interfaces 3, 1600335 (2016).
Hu, Q. et al. The precise decoration of Pt nanoparticles with Fe oxide by atomic layer deposition for the selective hydrogenation of cinnamaldehyde. Appl. Catal. B: Environ. 218, 591–599 (2017).
Karim, W. et al. Catalyst support effects on hydrogen spillover. Nature 541, 68–71 (2017).
Jiang, L. et al. Facet engineering accelerates spillover hydrogenation on highly diluted metal nanocatalysts. Nat. Nanotechnol. 15, 848–853 (2020).
**ong, M., Gao, Z. & Qin, Y. Spillover in heterogeneous catalysis: new insights and opportunities. ACS Catal. 11, 3159–3172 (2021).
Wei, J. et al. In situ Raman monitoring and manipulating of interfacial hydrogen spillover by precise fabrication of Au/TiO2/Pt sandwich structures. Angew. Chem. Int. Ed. 59, 10343–10347 (2020).
Kalidindi, S. B., Indirani, M. & Jagirdar, B. R. First row transition metal ion-assisted ammonia−borane hydrolysis for hydrogen generation. Inorg. Chem. 47, 7424–7429 (2008).
Yamada, Y., Yano, K., Xu, Q. & Fukuzumi, S. Cu/Co3O4 nanoparticles as catalysts for hydrogen evolution from ammonia borane by hydrolysis. J. Phys. Chem. C. 114, 16456–16462 (2010).
Lu, D. et al. A simple and scalable route to synthesize CoxCu1−xCo2O4@CoyCu1−yCo2O4 yolk–shell microspheres, a high-performance catalyst to hydrolyze ammonia borane for hydrogen production. Small 15, 1805460 (2019).
Hess, N. J. et al. Spectroscopic studies of the phase transition in ammonia borane: Raman spectroscopy of single crystal NH3BH3 as a function of temperature from 88 to 330 K. J. Chem. Phys. 128, 034508 (2008).
Bart, J. C. J. & Vlaic, G. Extended X-ray absorption fine structure studies in catalysis. Adv. Catal. 35, 1–138 (1987).
Shido, T. & Prins, R. Application of synchrotron radiation to in situ characterization catalysts. Curr. Opin. Solid State Mater. Sci. 3, 330–335 (1998).
Zhan, W., Zhu, Q. & Xu, Q. Dehydrogenation of ammonia borane by metal nanoparticle catalysts. ACS Catal. 6, 6892–6905 (2016).
Gudmundsdottir, S., Skulason, E. & Jonsson, H. Reentrant mechanism for associative desorption: H2/Pt(110)-(1 x 2). Phys. Rev. Lett. 108, 156101 (2012).
Yu, C. et al. H2 thermal desorption spectra on Pt(111): a density functional theory and kinetic Monte Carlo simulation study. Catalysts 8, 450 (2018).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Hafner, J. Ab-initio simulations of materials using VASP: density-functional theory and beyond. J. Comput. Chem. 29, 2044–2078 (2008).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Csonka, G. I. et al. Assessing the performance of recent density functionals for bulk solids. Phys. Rev. B 79, 155107 (2009).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the dam** function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Gutiérrez, G., Taga, A. & Johansson, B. Theoretical structure determination of γ-Al2O3. Phys. Rev. B 65, 012101 (2001).
Mathew, K., Sundararaman, R., Letchworth-Weaver, K., Arias, T. A. & Hennig, R. G. Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J. Chem. Phys. 140, 084106 (2014).
Jónsson, H., Mills, G. & Jacobsen, K. W. in Nudged Elastic Band Method for Finding Minimum Energy Paths of Transitions 385−404 (World Scientific, 1998).
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (21773282, Z.G.; U1932131, Z.G.; and U1832208, Y.Q.), National Science Fund for Distinguished Young Scholars (21825204, Y.Q.), the National Key R&D Programme of China (2017YFA0700101, Y.Q.; and 2020YFA0210902, Y.Q.), and Youth Innovation Promotion Association of the Chinese Academy of Sciences (2018208, Z.G.). We are grateful to all staff at the 1W1B beamline of the Bei**g Synchrotron Radiation Facility, Institute of High Energy Physics, Chinese Academy of Sciences, and at BL14W1 and BL11B beamlines of the Shanghai Synchrotron Radiation Facility, Shanghai Advanced Research Institute, Chinese Academy of Sciences.
Author information
Authors and Affiliations
Contributions
Z.G. synthesized the catalysts and performed the activity tests. G.W., Z.L., M.X., J.M., and Z.J. helped to perform or provide the XANES measurement. T.L. performed the theoretical calculation. L.W. and S.X. assisted in the synthesis and characterizations of the catalysts. Z.G. and Y.Q. conceived the idea, supervised the work, and wrote the manuscript. All authors contributed to the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review information
Nature Communications thanks Niall English and the other, anonymous, reviewer for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, Z., Wang, G., Lei, T. et al. Enhanced hydrogen generation by reverse spillover effects over bicomponent catalysts. Nat Commun 13, 118 (2022). https://doi.org/10.1038/s41467-021-27785-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-021-27785-5
- Springer Nature Limited