Abstract
Osteoarthritis (OA) is an age-related disease characterized by cartilage degeneration. TNFR1-associated death domain protein (TRADD) is a key upstream molecule of TNF-α signals but its role in OA pathogenesis is unknown. This study aimed to verify that whether inhibition of TRADD could protect against chondrocyte necroptosis and OA, and further elucidate the underlying mechanism. We demonstrated that TNF-α-related OA-like phenotypes including inflammation response, extracellular matrix degradation, apoptosis, and necroptosis in chondrocytes were inhibited by TRADD deficiency. Furthermore, TRADD interacted with TRAF2 and knockdown of TRADD suppressed the activation of RIPK1-TAK1-NF-κB signals and restored impaired autophagy. ICCB-19, the selective inhibitor of TRADD, also attenuated necroptosis in chondrocytes. Mechanismly, ICCB-19 blocked the phosphorylation of TAK1-NF-κB signals and restored impaired autophagy, whereas inhibiting autophagic process with 3-Methyladenine compromised these effects of ICCB-19. The in vivo study showed that the intra-articular injection of ICCB-19 rescued the expression of collagen alpha-1(II) chain and LC3, and mitigated the cartilage degeneration of OA mice. This study demonstrates that TRADD mediates TNF-α-induced necroptosis and OA-like phenotypes of chondrocytes and suggests that ICCB-19 suppresses chondrocyte damage and cartilage degeneration by inhibiting TNF-α-TRADD-mediated signals and dysregulation of autophagy in chondrocytes. ICCB-19 may serve as an important option for OA therapy.
Similar content being viewed by others
Introduction
Osteoarthritis (OA) is a common disease that causes physical pain and even disability in patients. This disease affects the majority of older people in the world and brings huge economic burden to society [1, 2]. A variety of risk factors have been identified for OA [3], but the clear pathogenesis and effective treatment remain to be further studied. The pathological changes of OA are characterized by chondrocyte death and cartilage destruction, synovitis, and osteophyte formation [4]. Chondrocytes and their secreted extracellular matrix (ECM) are main components of articular cartilage [5]. Chondrocytes maintain physiological structure of articular cartilage and improve joint function by well-regulating the balance of anabolism and catabolism of ECM [4]. Tumor necrosis factor-α (TNF-α) is a key inflammatory mediator in OA progression and one of the leading cytokines giving rise to the imbalance of matrix synthesis and decomposition [6, 7]. Therefore, it is important to maintain homeostasis of chondrocytes by protecting chondrocytes against death and degradation of ECM under the pathological conditions.
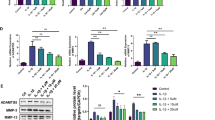
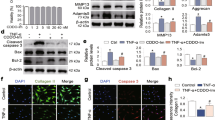
TNFR1-associated death domain protein (TRADD), is an adaptor molecule for the signaling downstream of tumor necrosis factor receptor 1 (TNFR1) [47]. The sections were incubated with anti-LC3, MMP13, P-RIPK3, and COL2A1 antibodies to observe expression changes of these markers.
Statistical analysis
Graph Pad Prism 8.0 software was used for statistical analysis. Results were revealed as mean ± standard deviation (SD). Student T test (unpaired, two-tailed) was used for statistical analysis between the two groups. Comparisons between more than two groups were performed by ANOVA and Tukey test. P values <0.05 was statistically significant. N = 3 means the in vitro experiments were performed at least three biological replicates.
Data availability
Full images of western blotting had been included in Supplementary Materials. Other data are available from the corresponding authors for reasonable grounds.
References
Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet. 2019;393:1745–59.
Mobasheri A, Batt M. An update on the pathophysiology of osteoarthritis. Ann Phys Rehabil Med. 2016;59:333–9.
Silverwood V, Blagojevic-Bucknall M, **ks C, Jordan JL, Protheroe J, Jordan KP. Current evidence on risk factors for knee osteoarthritis in older adults: a systematic review and meta-analysis. Osteoarthr Cartil. 2015;23:507–15.
Guilak F, Nims RJ, Dicks A, Wu C, Meulenbelt I. Osteoarthritis as a disease of the cartilage pericellular matrix. Matrix Biol. 2018;71-72:40–50.
Pitsillides AA, Beier F. Cartilage biology in osteoarthritis—lessons from developmental biology. Nat Rev Rheumatol. 2011;7:654–63.
Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier J, Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011;7:33–42.
Ma C, Wu C, Jou I, Tu Y, Hung C, Hsieh P, et al. PKR activation causes inflammation and MMP-13 secretion in human degenerated articular chondrocytes. Redox Biol. 2018;14:72–81.
Hsu H, **ong J, Goeddel DV. The TNF receptor 1-associated protein TRADD signals cell death and NF-kappa B activation. In, 1995:495-504.
Xu D, Zhao H, ** M, Zhu H, Shan B, Geng J, et al. Modulating TRADD to restore cellular homeostasis and inhibit apoptosis. Nature .2020;587:133–8.
Pobezinskaya YL, Liu Z. The role of TRADD in death receptor signaling. Cell Cycle. 2014;11:871–6.
Wajant H, Scheurich P. TNFR1-induced activation of the classical NF-κB pathway. FEBS J. 2011;278:862–76.
Ea C, Deng L, **a Z, Pineda G, Chen ZJ. Activation of IKK by TNFalpha requires site-specific ubiquitination of RIP1 and polyubiquitin binding by NEMO. Mol Cell. 2006;22:245–57.
Zhang D, Shao J, Lin J, Zhang N, Lu B, Lin S, et al. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science. 2009;325:332–6.
Yamazaki K, Gohda J, Kanayama A, Miyamoto Y, Sakurai H, Yamamoto M, et al. Two mechanistically and temporally distinct NF-κB activation pathways in IL-1 signaling. Sci Signal. 2009;2:a66.
Liu WJ, Ye L, Huang WF, Guo LJ, Xu ZG, Wu HL, et al. p62 links the autophagy pathway and the ubiqutin–proteasome system upon ubiquitinated protein degradation. Cell Mol Biol Lett. 2016;21:29.
Liang S, Wang Z, Zhang Z, Chen K, Lv Z, Wang Y, et al. Decreased RIPK1 expression in chondrocytes alleviates osteoarthritis via the TRIF/MyD88-RIPK1-TRAF2 negative feedback loop. Aging 2019;11:8664–80.
Gaud G, Guillemot D, Jacob Y, Favre M, Vuillier F. EVER2 protein binds TRADD to promote TNF-α-induced apoptosis. Cell Death Dis. 2013;4:e499.
Pasparakis M, Vandenabeele P. Necroptosis and its role in inflammation. Nature.2015;517:311–20.
Lotz MK, Caramés B. Autophagy and cartilage homeostasis mechanisms in joint health, aging and OA. Nat Rev Rheumatol. 2011;7:579–87.
Sun K, **g X, Guo J, Yao X, Guo F. Mitophagy in degenerative joint diseases. Autophagy 2021;17:2082–92.
Boffa A, Merli G, Andriolo L, Lattermann C, Salzmann GM, Filardo G. Synovial fluid biomarkers in knee osteoarthritis: a systematic review and quantitative evaluation using BIPEDs criteria. Cartilage 2021;13:82S–103S.
Lee SW, Rho JH, Lee SY, Kim JH, Cheong JH, Kim HY, et al. Leptin protects rat articular chondrocytes from cytotoxicity induced by TNF-alpha in the presence of cyclohexamide. Osteoarthr Cartil. 2015;23:2269–78.
Sun K, Luo J, **g X, Guo J, Yao X, Hao X, et al. Astaxanthin protects against osteoarthritis via Nrf2: a guardian of cartilage homeostasis. Aging 2019;11:10513–31.
Riegger J, Brenner RE. Evidence of necroptosis in osteoarthritic disease: investigation of blunt mechanical impact as possible trigger in regulated necrosis. Cell Death Dis. 2019;10:683.
Zhang C, Lin S, Li T, Jiang Y, Huang Z, Wen J, et al. Mechanical force-mediated pathological cartilage thinning is regulated by necroptosis and apoptosis. Osteoarthr Cartil. 2017;25:1324–34.
Ogasawara M, Yano T, Tanno M, Abe K, Ishikawa S, Miki T, et al. Suppression of autophagic flux contributes to cardiomyocyte death by activation of necroptotic pathways. J Mol Cell Cardiol. 2017;108:203–13.
Horiuchi T, Mitoma H, Harashima SI, Tsukamoto H, Shimoda T. Transmembrane TNF-: structure, function and interaction with anti-TNF agents. Rheumatology 2010;49:1215–28.
Dowling JP, Alsabbagh M, Del Casale C, Liu Z, Zhang J. TRADD regulates perinatal development and adulthood survival in mice lacking RIPK1 and RIPK3. Nat Commun. 2019;10:705.
Ermolaeva MA, Michallet M, Papadopoulou N, Utermöhlen O, Kranidioti K, Kollias G, et al. Function of TRADD in tumor necrosis factor receptor 1 signaling and in TRIF-dependent inflammatory responses. Nat Immunol. 2008;9:1037–46.
Wang L, Chang X, Feng J, Yu J, Chen G. TRADD Mediates RIPK1-Independent Necroptosis Induced by Tumor Necrosis Factor. Front Cell Developmental Biol. 2020;7:393.
Sueishi T, Akasaki Y, Goto N, Kurakazu I, Toya M, Kuwahara M, et al. GRK 5 inhibition attenuates cartilage degradation via decreasedNF ‐κB signaling. Arthritis Rheumatol 2020;72:620–31.
Yan H, Duan X, Pan H, Holguin N, Rai MF, Akk A, et al. Suppression of NF-κB activity via nanoparticle-based siRNA delivery alters early cartilage responses to injury. Proc Natl Acad Sci. 2016;113:E6199–E6208.
Qiu X, Zhuang M, Lu Z, Liu Z, Cheng D, Zhu C, et al. RIPK1 suppresses apoptosis mediated by TNF and caspase-3 in intervertebral discs. J Transl Med. 2019;17:135.
Wang H, Sun L, Su L, Rizo J, Liu L, Wang L, et al. Mixed lineage kinase domain-like protein MLKL causes necrotic membrane disruption upon phosphorylation by RIP3. Mol Cell. 2014;54:133–46.
Bertheloot D, Latz E, Franklin BS. Necroptosis, pyroptosis and apoptosis: an intricate game of cell death. Cell Mol Immunol. 2021;18:1106–21.
Rockel JS, Kapoor M. Autophagy: controlling cell fate in rheumatic diseases. Nat Rev Rheumatol 2016;12:517–31.
Bouderlique T, Vuppalapati KK, Newton PT, Li L, Barenius B, Chagin AS. Targeted deletion of Atg5 in chondrocytes promotes age-related osteoarthritis. Ann Rheum Dis. 2016;75:627–31.
Zhang Y, Vasheghani F, Li Y, Blati M, Simeone K, Fahmi H, et al. Cartilage-specific deletion of mTOR upregulates autophagy and protects mice from osteoarthritis. Ann Rheum Dis. 2015;74:1432–40.
Caramés B, Hasegawa A, Taniguchi N, Miyaki S, Blanco FJ, Lotz M. Autophagy activation by rapamycin reduces severity of experimental osteoarthritis. Ann Rheum Dis. 2012;71:575–81.
Li J, Jiang M, Yu Z, **ong C, Pan J, Cai Z, et al. Artemisinin relieves osteoarthritis by activating mitochondrial autophagy through reducing TNFSF11 expression and inhibiting PI3K/AKT/mTOR signaling in cartilage. Cell Mol Biol Lett. 2022;27:62.
Matsuzawa-Ishimoto Y, Shono Y, Gomez LE, Hubbard-Lucey VM, Cammer M, Neil J, et al. Autophagy protein ATG16L1 prevents necroptosis in the intestinal epithelium. J Exp Med. 2017;214:3687–705.
Abe K, Yano T, Tanno M, Miki T, Kuno A, Sato T, et al. mTORC1 inhibition attenuates necroptosis through RIP1 inhibition-mediated TFEB activation. Biochimica et Biophysica Acta (BBA) - Mol Basis Dis. 2019;1865:165552.
Leidal AM, Levine B, Debnath J. Autophagy and the cell biology of age-related disease. Nat Cell Biol. 2018;20:1338–48.
Meckes JK, Caramés B, Olmer M, Kiosses WB, Grogan SP, Lotz MK, et al. Compromised autophagy precedes meniscus degeneration and cartilage damage in mice. Osteoarthr Cartil. 2017;25:1880–9.
Ma HL, Blanchet TJ, Peluso D, Hopkins B, Morris EA, Glasson SS. Osteoarthritis severity is sex dependent in a surgical mouse model. Osteoarthr Cartil. 2007;15:695–700.
Schmitz N, Laverty S, Kraus VB, Aigner T. Basic methods in histopathology of joint tissues. Osteoarthr Cartil. 2010;18:S113–S116.
Sun K, Luo J, **g X, **ang W, Guo J, Yao X, et al. Hyperoside ameliorates the progression of osteoarthritis: An in vitro and in vivo study. Phytomedicine 2021;80:153387.
Funding
This work was supported by the National Natural Science Foundation of China (82172498), the Project of Health Commission of Hubei Province (WJ2021M123), and the Knowledge Innovation Project of Wuhan (2022020801010442).
Author information
Authors and Affiliations
Contributions
WTZ Conception, design and obtaining of funding. FJG Conception and obtaining of funding. KS Data acquisition, analysis and drafting of the manuscript. ZG Data acquisition, analysis. JMZ Conception and design. LCH Data acquisition. SL: Technical support. FL Data acquisition and revision of the manuscript. GCW Analysis and interpretation of the data. JTX Technical support. XZ Technical support. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sun, K., Guo, Z., Zhang, J. et al. Inhibition of TRADD ameliorates chondrocyte necroptosis and osteoarthritis by blocking RIPK1-TAK1 pathway and restoring autophagy. Cell Death Discov. 9, 109 (2023). https://doi.org/10.1038/s41420-023-01406-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-023-01406-0
- Springer Nature Limited