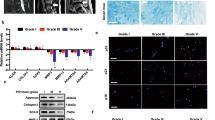
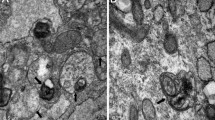
Abstract
Treatment of intervertebral disc degeneration (IDD) seeks to prevent senescence and death of nucleus pulposus (NP) cells. Previous studies have shown that sirt6 exerts potent anti-senescent and anti-apoptotic effects in models of age-related degenerative disease. However, it is not known whether sirt6 protects against IDD. Here, we explored whether sirt6 influenced IDD. The sirt6 level was reduced in senescent human NP cells. Sirt6 overexpression protected against apoptosis and both replicative and stress-induced premature senescence. Sirt6 also activated NP cell autophagy both in vivo and in vitro. 3-methyladenine (3-MA) and chloroquine (CQ)-mediated inhibition of autophagy partially reversed the anti-senescent and anti-apoptotic effects of sirt6, which regulated the expression of degeneration-associated proteins. In vivo, sirt6 overexpression attenuated IDD. Together, the data showed that sirt6 attenuated cell senescence, and reduced apoptosis, by triggering autophagy that ultimately ameliorated IDD. Thus, sirt6 may be a novel therapeutic target for IDD treatment.
Similar content being viewed by others
Introduction
Intervertebral disc degeneration (IDD) is an age-related degenerative disease and the major cause of low back pain, reducing quality-of-life and creating a large economic burden1. Various patient-specific and external factors, including age, a genetic predisposition, and mechanical stress, contribute to IDD initiation and progression2,Immunohistochemical examination All discs were embedded in paraffin and cut into sections (5 μm). The sections were deparaffinised in xylene, rehydrated in ethanol, and blocked by addition of 3% (v/v) H2O2 for 10 min followed by incubation in 5% BSA for 30 min. After incubation with primary antibodies (anti-LC3, anti-cleaved caspase 3), the sections were incubated with the respective second antibodies and counterstained with hematoxylin. Images were captured using a light microscope. To measure the extent of IDD after surgery, all rats were killed 8 weeks after surgery and disc tissues sections (5 µm) were cut and treated as described above for hematoxylin-eosin (HE) staining. Images were captured using a light microscope. Magnetic resonance imaging (MRI) of the coccyx was performed 8 weeks after surgery. All rats were anesthetised throughout the examinations, and their tails straightened. We subjected five rats of each group (total, 15) to sagittal and horizontal T2-weighted imaging with a 3.0-T clinical magnet (Philips Intera Achieva 3.0MR). T2-weighted sections were set as followed: a fast-spin echo sequence with a time-to-repetition of 5400 ms and a time-to-echo of 920 ms; a 320 (h) × 256 (v) matrix; a field of view of 260°; and four excitations. The section thickness was 2 mm and the gap 0 mm. All MR images were analyzed in a blinded manner using the IDD classification of Pfirrmann et al.49 All data are the means ± standard deviations (SDs). Results were compared via Graphpad Prism (USA) (one-way analysis of variance and Tukey’s post hoc test). P values <0.05 were considered statistically significant.Hematoxylin-eosin staining
Magnetic resonance imaging
Data analysis
References
Luoma, K. et al. Low back pain in relation to lumbar disc degeneration. Spine 25, 487–492 (2000).
Feng, Y., Egan, B. & Wang, J. Genetic factors in intervertebral disc degeneration. Genes Dis. 3, 178–185 (2016).
**a, D. D. et al. Effects of shear force on intervertebral disc: an in vivo rabbit study. Eur. Spine J. 24, 1711–1719 (2015).
Sakai, D. et al. Exhaustion of nucleus pulposus progenitor cells with ageing and degeneration of the intervertebral disc. Nat. Commun. 3, 1264 (2012).
Nasto, L. A. et al. Investigating the role of DNA damage in tobacco smoking-induced spine degeneration. Spine J. 14, 416–423 (2014).
**ng, Q. J. et al. Leg amputation accelerates senescence of rat lumbar intervertebral discs. Spine 35, E1253–E1261 (2010).
Adams, M. A. & Roughley, P. J. What is intervertebral disc degeneration, and what causes it? Spine 31, 2151–2161 (2006).
Zhao, C. Q., Wang, L. M., Jiang, L. S. & Dai, L. Y. The cell biology of intervertebral disc aging and degeneration. Ageing Res. Rev. 6, 247–261 (2007).
Hall, J. A., Dominy, J. E., Lee, Y. & Puigserver, P. The sirtuin family’s role in aging and age-associated pathologies. J. Clin. Invest. 123, 973–979 (2013).
Sebastian, C., Satterstrom, F. K., Haigis, M. C. & Mostoslavsky, R. From sirtuin biology to human diseases: an update. J. Biol. Chem. 287, 42444–42452 (2012).
Kanfi, Y. et al. The sirtuin SIRT6 regulates lifespan in male mice. Nature 483, 218–221 (2012).
Tao, N. N. et al. Deacetylation of Ku70 by SIRT6 attenuates Bax-mediated apoptosis in hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 485, 713–719 (2017).
Kanfi, Y. et al. The sirtuin SIRT6 regulates lifespan in male mice. Nature 483, 218–221 (2012).
Snigdha, S., Smith, E. D., Prieto, G. A. & Cotman, C. W. Caspase-3 activation as a bifurcation point between plasticity and cell death. Neurosci. Bull. 28, 14–24 (2012).
Adams, J. M. & Cory, S. The Bcl-2 protein family: arbiters of cell survival. Science 281, 1322–1326 (1998).
Yang, Z. & Klionsky, D. J. Eaten alive: a history of macroautophagy. Nat. Cell Biol. 12, 814–822 (2010).
Boquoi, A. et al. Reversible cell cycle inhibition and premature aging features imposed by conditional expression of p16Ink4a. Aging Cell 14, 139–147 (2015).
Qian, Y. & Chen, X. Senescence regulation by the p53 protein family. Methods Mol. Biol. 965, 37–61 (2013).
Kletsas, D. Senescent cells in the intervertebral disc: numbers and mechanisms. Spine J. 9, 677–678 (2009).
Wang, F., Cai, F., Shi, R., Wang, X. H. & Wu, X. T. Aging and age related stresses: a senescence mechanism of intervertebral disc degeneration. Osteoarthritis Cartilage 24, 398–408 (2016).
Davalos, A. R., Coppe, J. P., Campisi, J. & Desprez, P. Y. Senescent cells as a source of inflammatory factors for tumor progression. Cancer Metastasis Rev. 29, 273–283 (2010).
van Deursen, J. M. The role of senescent cells in ageing. Nature 509, 439–446 (2014).
Risbud, M. V. & Shapiro, I. M. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat. Rev. Rheumatol. 10, 44–56 (2014).
Chung, S. A. et al. Nucleus pulposus cellular longevity by telomerase gene therapy. Spine 32, 1188–1196 (2007).
Nakai, T., Mochida, J., Sakai, D. Synergistic role of c-Myc and ERK1/2 in the mitogenic response to TGF beta-1 in cultured rat nucleus pulposus cells. Arthritis Res. Ther. 10, 1478–6354 (2008).
Chen, D. et al. Metformin protects against apoptosis and senescence in nucleus pulposus cells and ameliorates disc degeneration in vivo. Cell Death Dis. 7, e2441 (2016).
Kim, J. S. et al. Insulin-like growth factor 1 synergizes with bone morphogenetic protein 7-mediated anabolism in bovine intervertebral disc cells. Arthritis Rheum. 62, 3706–3715 (2010).
Kawahara, T. L. A. et al. SIRT6 links histone H3 lysine 9 deacetylation to NF-kappa B-dependent gene expression and organismal life span. Cell 136, 62–74 (2009).
Cardus, A., Uryga, A. K., Walters, G. & Erusalimsky, J. D. SIRT6 protects human endothelial cells from DNA damage, telomere dysfunction, and senescence. Cardiovasc. Res. 97, 571–579 (2013).
Maksin-Matveev, A. et al. Sirtuin 6 protects the heart from hypoxic damage. Exp. Cell Res. 330, 81–90 (2015).
Wu, Y. et al. Overexpression of Sirtuin 6 suppresses cellular senescence and NF-kappaB mediated inflammatory responses in osteoarthritis development. Sci. Rep. 5, 17602 (2015).
Kang, L., Hu, J., Weng, Y. X., Jia, J. & Zhang, Y. K. Sirtuin 6 prevents matrix degradation through inhibition of the NF-kappa B pathway in intervertebral disc degeneration. Exp. Cell Res. 352, 322–332 (2017).
Le Maitre, C. L., Freemont, A. J. & Hoyland, J. A. The role of interleukin-1 in the pathogenesis of human intervertebral disc degeneration. Arthritis Res. Ther. 7, R732–R745 (2005).
Weiler, C., Nerlich, A. G., Bachmeier, B. E. & Boos, N. Expression and distribution of tumor necrosis factor alpha in human lumbar intervertebral discs: a study in surgical specimen and autopsy controls. Spine 30, 44–53 (2005).
Dinarello, C. A. Interleukin-1 in the pathogenesis and treatment of inflammatory diseases. Blood 117, 3720–3732 (2011).
Xu, D. et al. Hydrogen sulfide protects against endoplasmic reticulum stress and mitochondrial injury in nucleus pulposus cells and ameliorates intervertebral disc degeneration. Pharmacol. Res. 117, 357–369 (2017).
Dai, S. M. et al. Catabolic stress induces features of chondrocyte senescence through overexpression of caveolin 1 - possible involvement of caveolin 1-induced down-regulation of articular chondrocytes in the pathogenesis of osteoarthritis. Arthritis Rheum. 54, 818–831 (2006).
Sasaki, H. et al. Autophagy modulates osteoarthritis-related gene expression in human chondrocytes. Arthritis Rheum. 64, 1920–1928 (2012).
Ye, W. et al. Age-related increases of macroautophagy and chaperone-mediated autophagy in rat nucleus pulposus. Connect. Tissue Res. 52, 472–478 (2011).
Las, G. & Shirihai, O. S. The role of autophagy in beta-cell lipotoxicity and type 2 diabetes. Diabetes Obes. Metab. 12, 15–19 (2010).
Mizushima, N. & Komatsu, M. Autophagy: renovation of cells and tissues. Cell 147, 728–741 (2011).
Miyazaki, S. et al. Recombinant human SIRT1 protects against nutrient deprivation-induced mitochondrial apoptosis through autophagy induction in human intervertebral disc nucleus pulposus cells. Arthritis Res. Ther. 17, 253 (2015).
Takasaka, N. et al. Autophagy induction by SIRT6 through attenuation of insulin-like growth factor signaling is involved in the regulation of human bronchial epithelial cell senescence. J. Immunol. 192, 958–968 (2014).
He, J. P. et al. SIRT6 reduces macrophage foam cell formation by inducing autophagy and cholesterol efflux under ox-LDL condition. FEBS J. 284, 1324–1337 (2017).
Shao, J. X. et al. Autophagy induction by SIRT6 is involved in oxidative stress-induced neuronal damage (vol 7, pg 281, 2016). Protein Cell 8, 313–313 (2017).
Risbud, M. V. et al. Nucleus pulposus cells express HIF-1 alpha under normoxic culture conditions: a metabolic adaptation to the intervertebral disc microenvironment. J. Cell. Biochem. 98, 152–159 (2006).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 29, e45 (2001).
Han, B. et al. A simple disc degeneration model induced by percutaneous needle puncture in the rat tail. Spine 33, 1925–1934 (2008).
Pfirrmann, C. W., Metzdorf, A., Zanetti, M., Hodler, J. & Boos, N. Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine 26, 1873–1878 (2001).
Acknowledgements
This study was funded by the following foundations: Natural Science Foundation of Zhejiang Province (LY15H060008), National Natural Science Foundation of China (81501907, 81371988), and Wenzhou Municipal Science and Technology Bureau (Y20150063), Science and technology plan of Zhejiang medical and Health Department (2014KYB158), Zhejiang science and technology innovation program for College Students (2014R413021).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, J., **e, JJ., **, MY. et al. Sirt6 overexpression suppresses senescence and apoptosis of nucleus pulposus cells by inducing autophagy in a model of intervertebral disc degeneration. Cell Death Dis 9, 56 (2018). https://doi.org/10.1038/s41419-017-0085-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-017-0085-5
- Springer Nature Limited
This article is cited by
-
Sirtuins in intervertebral disc degeneration: current understanding
Molecular Medicine (2024)
-
S100A6 Regulates nucleus pulposus cell apoptosis via Wnt/β-catenin signaling pathway: an in vitro and in vivo study
Molecular Medicine (2024)
-
Overexpression of SIRT6 alleviates apoptosis and enhances cell viability and monoclonal antibody expression in CHO-K1 cells
Molecular Biology Reports (2023)
-
Serum SIRT6 Levels Are Associated with Frailty in Older Adults
The Journal of nutrition, health and aging (2023)
-
Molecular mechanisms of dietary restriction promoting health and longevity
Nature Reviews Molecular Cell Biology (2022)