Abstract
Background
We examined associations of CD44, CD24 and ALDH1A1 breast stem cell markers with mammographic breast density (MBD), a well-established breast cancer (BCa) risk factor.
Methods
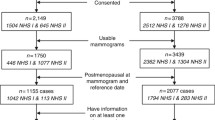
We included 218 cancer-free women with biopsy-confirmed benign breast disease within the Nurses’ Health Study (NHS) and NHSII. The data on BCa risk factors were obtained from biennial questionnaires. Immunohistochemistry (IHC) was done on tissue microarrays. For each core, the IHC expression was assessed using a semi-automated platform and expressed as percent of positively stained cells for each marker out of the total cell count. MBD was assessed with computer-assisted techniques. Generalised linear regression was used to examine the associations of each marker with square root-transformed percent density (PD), absolute dense and non-dense areas (NDA), adjusted for BCa risk factors.
Results
Stromal CD44 and ALDH1A1 expression was positively associated with PD (≥ 10% vs. <10% β = 0.56, 95% confidence interval [CI] [0.06; 1.07] and β = 0.81 [0.27; 1.34], respectively) and inversely associated with NDA (β per 10% increase = −0.17 [−0.34; −0.01] and β for ≥10% vs. <10% = −1.17 [−2.07; −0.28], respectively). Epithelial CD24 expression was inversely associated with PD (β per 10% increase = −0.14 [−0.28; −0.01]. Stromal and epithelial CD24 expression was positively associated with NDA (β per 10% increase = 0.35 [0.2 × 10−2; 0.70] and β per 10% increase = 0.34 [0.11; 0.57], respectively).
Conclusion
Expression of stem cell markers is associated with MBD.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the Nurses’ Health Studies, however they are not publicly available. Investigators interested in using the data can request access, and feasibility will be discussed at an investigators’ meeting. Limits are not placed on scientific questions or methods, and there is no requirement for co-authorship. Additional data-sharing information and policy details can be accessed at http://www.nurseshealthstudy.org/researchers.
Code availability
The underlying code for this study is not publicly available but may be made available to qualified researchers on reasonable request from the corresponding author.
References
McCormack VA, dos Santos Silva I. Breast density and parenchymal patterns as markers of breast cancer risk: a meta-analysis. Cancer Epidemiol Biomark Prev. 2006;15:1159–69.
Vachon CM, Kuni CC, Anderson K, Anderson VE, Sellers TA. Association of mammographically defined percent breast density with epidemiologic risk factors for breast cancer (United States). Cancer Causes Control. 2000;11:653–62.
Vachon CM, Sellers TA, Vierkant RA, Wu FF, Brandt KR. Case-control study of increased mammographic breast density response to hormone replacement therapy. Cancer Epidemiol Biomark Prev. 2002;11:1382–8.
Titus-Ernstoff L, Tosteson AN, Kasales C, Weiss J, Goodrich M, Hatch EE, et al. Breast cancer risk factors in relation to breast density (United States). Cancer Causes Control. 2006;17:1281–90.
Tamimi RM, Hankinson SE, Colditz GA, Byrne C. Endogenous sex hormone levels and mammographic density among postmenopausal women. Cancer Epidemiol Biomark Prev. 2005;14:2641–7.
Yaghjyan L, Colditz GA, Rosner B, Bertrand KA, Tamimi RM. Reproductive factors related to childbearing and mammographic breast density. Breast Cancer Res. Treat. 2016;158:351–9.
Cobaleda C, Cruz JJ, Gonzalez-Sarmiento R, Sanchez-Garcia I, Perez-Losada J. The emerging picture of human breast cancer as a stem cell-based disease. Stem Cell Rev. 2008;4:67–79.
Smalley M, Ashworth A. Stem cells and breast cancer: a field in transit. Nat Rev Cancer. 2003;3:832–44.
Chang C-C. Recent translational research: stem cells as the roots of breast cancer. Breast Cancer Res. 2006;8:103.
Dowsett M, Goldhirsch A, Hayes DF, Senn HJ, Wood W, Viale G. International Web-based consultation on priorities for translational breast cancer research. Breast Cancer Res. 2007;9:R81.
Giatromanolaki A, Sivridis E, Fiska A, Koukourakis M. The CD44+/CD24− phenotype relates to ‘triple-negative’ state and unfavorable prognosis in breast cancer patients. Med Oncol. 2010;28:745–52.
Abraham BK, Fritz P, Van der Kuip H, Buck M, Szabo Z, Athelogou M, et al. Evaluation of CD44+/CD24-/low cells in breast cancer and relevance for distant metastasis. AACR Meet Abstr. 2005;2005:481.
Neumeister V, Agarwal S, Bordeaux J, Camp RL, Rimm DL. In situ identification of putative cancer stem cells by multiplexing ALDH1, CD44, and cytokeratin identifies breast cancer patients with poor prognosis. Am J Pathol. 2010;176:2131–8.
Lee JH, Kim SH, Lee ES, Kim YS. CD24 overexpression in cancer development and progression: a meta-analysis. Oncol Rep. 2009;22:1149–56.
Eden JA. Human breast cancer stem cells and sex hormones-a narrative review. Menopause. 2010;17:801–10.
Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M, et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell. 2007;1:555–67.
Liu Y, Lv DL, Duan JJ, Xu SL, Zhang JF, Yang XJ, et al. ALDH1A1 expression correlates with clinicopathologic features and poor prognosis of breast cancer patients: a systematic review and meta-analysis. BMC Cancer. 2014;14:444.
Khoury T, Ademuyiwa FO, Chandraseekhar R, Jabbour M, DeLeo A, Ferrone S, et al. Aldehyde dehydrogenase 1A1 expression in breast cancer is associated with stage, triple negativity, and outcome to neoadjuvant chemotherapy. Mod Pathol. 2012;25:388–97.
Yao J, ** Q, Wang X-d, Zhu H-j, Ni Q-c. Aldehyde dehydrogenase 1 expression is correlated with poor prognosis in breast cancer. Medicine. 2017;96:e7171.
Ouhtit A, Abdraboh ME, Hollenbach AD, Zayed H, Raj MHG. CD146, a novel target of CD44-signaling, suppresses breast tumor cell invasion. Cell Commun Signal: CCS. 2017;15:45.
Kabel AM. Tumor markers of breast cancer: new prospectives. J Oncol Sci. 2017;3:5–11.
Sin WC, Lim CL. Breast cancer stem cells-from origins to targeted therapy. Stem cell Investig. 2017;4:96.
Yaghjyan L, Stoll E, Ghosh K, Scott CG, Jensen MR, Brandt KR, et al. Tissue-based associations of mammographic breast density with breast stem cell markers. Breast Cancer Res. 2017;19:100.
Yaghjyan L, Esnakula AK, Scott CG, Wijayabahu AT, Jensen MR, Vachon CM. Associations of mammographic breast density with breast stem cell marker-defined breast cancer subtypes. Cancer Causes Control. 2019;30:1103–11.
Tamimi RM, Colditz GA, Wang Y, Collins LC, Hu R, Rosner B, et al. Expression of IGF1R in normal breast tissue and subsequent risk of breast cancer. Breast Cancer Res Treat. 2011;128:243–50.
Collins LC, Baer HJ, Tamimi RM, Connolly JL, Colditz GA, Schnitt SJ. The influence of family history on breast cancer risk in women with biopsy-confirmed benign breast disease: results from the Nurses’ Health Study. Cancer. 2006;107:1240–7.
Tamimi RM, Byrne C, Colditz GA, Hankinson SE. Endogenous hormone levels, mammographic density, and subsequent risk of breast cancer in postmenopausal women. J Natl Cancer Inst. 2007;99:1178–87.
Yaghjyan L, Heng YJ, Baker GM, Murthy D, Mahoney MB, Rosner B, et al. Associations of stem cell markers in benign breast tissue with subsequent breast cancer risk. Am J Cancer Res. 2023;13:6280–9.
Tamimi RM, Rosner B, Colditz GA. Evaluation of a breast cancer risk prediction model expanded to include category of prior benign breast disease lesion. Cancer. 2010;116:4944–53.
Kensler KH, Beca F, Baker GM, Heng YJ, Beck AH, Schnitt SJ, et al. Androgen receptor expression in normal breast tissue and subsequent breast cancer risk. NPJ Breast Cancer. 2018;4:33.
Malkov S, Shepherd JA, Scott CG, Tamimi RM, Ma L, Bertrand KA, et al. Mammographic texture and risk of breast cancer by tumor type and estrogen receptor status. Breast Cancer Res. 2016;18:122.
Yaghjyan L, Colditz G, Eliassen H, Rosner B, Gasparova A, Tamimi RM. Interactions of alcohol and postmenopausal hormone use in regards to mammographic breast density. Cancer Causes Control. 2018;29:751–8.
Byng JW, Boyd NF, Little L, Lockwood G, Fishell E, Jong RA, et al. Symmetry of projection in the quantitative analysis of mammographic images. Eur J Cancer Prev. 1996;5:319–27.
Jacobs TW, Byrne C, Colditz G, Connolly JL, Schnitt SJ. Pathologic features of breast cancers in women with previous benign breast disease. Am J Clin Pathol. 2001;115:362–9.
Tamimi RM, Byrne C, Baer HJ, Rosner B, Schnitt SJ, Connolly JL, et al. Benign breast disease, recent alcohol consumption, and risk of breast cancer: a nested case–control study. Breast Cancer Res. 2005;7:R555.
Page DL, Dupont WD, Rogers LW, Rados MS. Atypical hyperplastic lesions of the female breast. A long-term follow-up study. Cancer. 1985;55:2698–708.
Collins LC, Wang Y, Connolly JL, Baer HJ, Hu R, Schnitt SJ, et al. Potential role of tissue microarrays for the study of biomarker expression in benign breast disease and normal breast tissue. Appl Immunohistochem Mol Morphol. 2009;17:438–41.
Yaghjyan L, Heng YJ, Baker GM, Bret-Mounet V, Murthy D, Mahoney MB, et al. Reliability of CD44, CD24, and ALDH1A1 immunohistochemical staining: pathologist assessment compared to quantitative image analysis. Front Med. 2022;9:1040061.
Turashvili G, Leung S, Turbin D, Montgomery K, Gilks B, West R, et al. Inter-observer reproducibility of HER2 immunohistochemical assessment and concordance with fluorescent in situ hybridization (FISH): pathologist assessment compared to quantitative image analysis. BMC Cancer. 2009;9:165.
Pettersson A, Hankinson SE, Willett WC, Lagiou P, Trichopoulos D, Tamimi RM. Nondense mammographic area and risk of breast cancer. Breast Cancer Res. 2011;13:R100.
Schernhammer ES, Tworoger SS, Eliassen AH, Missmer SA, Holly JM, Pollak MN, et al. Body shape throughout life and correlations with IGFs and GH. Endocr-Relat Cancer. 2007;14:721–32.
Farabaugh SM, Boone DN, Lee AV. Role of IGF1R in breast cancer subtypes, stemness, and lineage differentiation. Front Endocrinol. 2015;6:59.
Isfoss B, Holmqvist B, Jernström H, Alm P, Olsson H. Women with familial risk for breast cancer have an increased frequency of aldehyde dehydrogenase expressing cells in breast ductules. BMC Clin Pathol. 2013;13:1–9.
Britt K, Ashworth A, Smalley M. Pregnancy and the risk of breast cancer. Endocr Relat Cancer. 2007;14:907–33.
Dall G, Risbridger G, Britt K. Mammary stem cells and parity-induced breast cancer protection- new insights. J Steroid Biochem Mol Biol. 2017;170:54–60.
Baer HJ, Colditz GA, Willett WC, Dorgan JF. Adiposity and sex hormones in girls. Cancer Epidemiol Biomark Prev. 2007;16:1880–8.
Bertrand KA, Baer HJ, Orav EJ, Klifa C, Shepherd JA, Van Horn L, et al. Body fatness during childhood and adolescence and breast density in young women: a prospective analysis. Breast Cancer Res. 2015;17:1–10.
Poole EM, Tworoger SS, Hankinson SE, Schernhammer ES, Pollak MN, Baer HJ. Body size in early life and adult levels of insulin-like growth factor 1 and insulin-like growth factor binding protein 3. Am J Epidemiol. 2011;174:642–51.
Warner ET, Eliassen AH, Chen WY, Holmes MD, Willett WC, Tamimi RM. Abstract 130: childhood and adolescent body size and risk of fatal breast cancer. Cancer Res. 2013;73:130.
Baer HJ, Colditz GA, Rosner B, Michels KB, Rich-Edwards JW, Hunter DJ, et al. Body fatness during childhood and adolescence and incidence of breast cancer in premenopausal women: a prospective cohort study. Breast Cancer Res. 2005;7:1–12.
Palmer JR, Adams-Campbell LL, Boggs DA, Wise LA, Rosenberg L. A prospective study of body size and breast cancer in black women. Cancer Epidemiol Biomark Prev. 2007;16:1795–802.
Frazier AL, Rosenberg SM. Pre-adolescent and adolescent risk factors for benign breast disease. J Adolesc Health. 2013;52:S36–S40.
Warner ET, Hu R, Collins LC, Beck AH, Schnitt S, Rosner B, et al. Height and body size in childhood, adolescence, and young adulthood and breast cancer risk according to molecular subtype in the Nurses’ Health Studies. Cancer Prev Res. 2016;9:732–8.
Woolcott CG, Koga K, Conroy SM, Byrne C, Nagata C, Ursin G, et al. Mammographic density, parity and age at first birth, and risk of breast cancer: an analysis of four case-control studies. Breast Cancer Res Treat. 2012;132:1163–71.
Yaghjyan L, Mahoney MC, Succop P, Wones R, Buckholz J, Pinney SM. Relationship between breast cancer risk factors and mammographic breast density in the Fernald Community Cohort. Br J Cancer. 2012;106:996–1003.
Colacino JA, Azizi E, Brooks MD, Harouaka R, Fouladdel S, McDermott SP, et al. Heterogeneity of human breast stem and progenitor cells as revealed by transcriptional profiling. Stem Cell Rep. 2018;10:1596–609.
Ricardo S, Vieira AF, Gerhard R, Leitão D, Pinto R, Cameselle-Teijeiro JF, et al. Breast cancer stem cell markers CD44, CD24 and ALDH1: expression distribution within intrinsic molecular subtype. J Clin Pathol. 2011;64:937–46.
Escudero Mendez L, Srinivasan M, Hamouda RK, Ambedkar B, Arzoun H, Sahib I, et al. Evaluation of CD44+/CD24- and aldehyde dehydrogenase enzyme markers in cancer stem cells as prognostic indicators for triple-negative breast cancer. Cureus. 2022;14:e28056.
Wang QA, Scherer PE. Remodeling of murine mammary adipose tissue during pregnancy, lactation, and involution. J Mammary Gland Biol Neoplasia. 2019;24:207–12.
Radisky DC, Hartmann LC. Mammary involution and breast cancer risk: transgenic models and clinical studies. J Mammary Gland Biol Neoplasia. 2009;14:181–91.
Colleluori G, Perugini J, Barbatelli G, Cinti S. Mammary gland adipocytes in lactation cycle, obesity and breast cancer. Rev Endocr Metab Disord. 2021;22:241–55.
Maskarinec G, Ju D, Horio D, Loo LW, Hernandez BY. Involution of breast tissue and mammographic density. Breast cancer Res: BCR. 2016;18:128.
Gierach GL, Patel DA, Pfeiffer RM, Figueroa JD, Linville L, Papathomas D, et al. Relationship of terminal duct lobular unit involution of the breast with area and volume mammographic densities. Cancer Prev Res. 2016;9:149–58.
Baker GM, Bret-Mounet VC, Wang T, Veta M, Zheng H, Collins LC, et al. Immunohistochemistry scoring of breast tumor tissue microarrays: a comparison study across three software applications. J Pathol Inf. 2022;13:100118.
Rice MS, Tamimi RM, Connolly JL, Collins LC, Shen D, Pollak MN, et al. Insulin-like growth factor-1, insulin-like growth factor binding protein-3 and lobule type in the Nurses’ Health Study II. Breast Cancer Res. 2012;14:1–7.
Huh SJ, Oh H, Peterson MA, Almendro V, Hu R, Bowden M, et al. The proliferative activity of mammary epithelial cells in normal tissue predicts breast cancer risk in premenopausal women. Cancer Res. 2016;76:1926–34.
Oh H, Eliassen AH, Wang M, Smith-Warner SA, Beck AH, Schnitt SJ, et al. Expression of estrogen receptor, progesterone receptor, and Ki67 in normal breast tissue in relation to subsequent risk of breast cancer. NPJ Breast Cancer. 2016;2:16032.
Oh H, Eliassen AH, Beck AH, Rosner B, Schnitt SJ, Collins LC, et al. Breast cancer risk factors in relation to estrogen receptor, progesterone receptor, insulin-like growth factor-1 receptor, and Ki67 expression in normal breast tissue. NPJ breast cancer. 2017;3:39.
Ghosh K, Hartmann LC, Reynolds C, Visscher DW, Brandt KR, Vierkant RA, et al. Association between mammographic density and age-related lobular involution of the breast. J Clin Oncol. 2010;28:2207–12.
Krishnan K, Baglietto L, Stone J, Simpson JA, Severi G, Evans CF, et al. Longitudinal study of mammographic density measures that predict breast cancer risk. Cancer Epidemiol, Biomark Prev. 2017;26:651–60.
Economopoulou P, Kaklamani VG, Siziopikou K. The role of cancer stem cells in breast cancer initiation and progression: potential cancer stem cell-directed therapies. Oncologist. 2012;17:1394–401.
Grudzien P, Lo S, Albain KS, Robinson P, Rajan P, Strack PR, et al. Inhibition of Notch signaling reduces the stem-like population of breast cancer cells and prevents mammosphere formation. Anticancer Res. 2010;30:3853–67.
Lamb R, Ablett MP, Spence K, Landberg G, Sims AH, Clarke RB. Wnt pathway activity in breast cancer sub-types and stem-like cells. PLoS ONE. 2013;8:e67811.
Lombardo Y, Filipović A, Molyneux G, Periyasamy M, Giamas G, Hu Y, et al. Nicastrin regulates breast cancer stem cell properties and tumor growth in vitro and in vivo. Proc Natl Acad Sci USA. 2012;109:16558–63.
Harrison H, Farnie G, Howell SJ, Rock RE, Stylianou S, Brennan KR, et al. Regulation of breast cancer stem cell activity by signaling through the Notch4 receptor. Cancer Res. 2010;70:709–18.
Louderbough JMV, Schroeder JA. Understanding the dual nature of CD44 in breast cancer progression. Mol Cancer Res. 2011;9:1573–86.
Acknowledgements
The authors would like to acknowledge the contribution to this study from central cancer registries supported through the Centers for Disease Control and Prevention’s National Program of Cancer Registries (NPCR) and/or the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) Program. Central registries may also be supported by state agencies, universities, and cancer centres. Participating central cancer registries include the following: Alabama, Alaska, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Florida, Georgia, Hawaii, Idaho, Indiana, Iowa, Kentucky, Louisiana, Massachusetts, Maine, Maryland, Michigan, Mississippi, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Puerto Rico, Rhode Island, Seattle SEER Registry, South Carolina, Tennessee, Texas, Utah, Virginia, West Virginia, Wyoming.
Funding
This work was supported by the National Cancer Institute at the National Institutes of Health [CA240341 to LY, CA131332, CA175080, P01 CA087969 to RMT, UM1 CA186107 and, to MS, U01 CA176726 to WW], Avon Foundation for Women, Susan G. Komen for the Cure®, and Breast Cancer Research Foundation.
Author information
Authors and Affiliations
Contributions
LY and RT conceived of and designed the study, directed statistical analyses, interpreted results, substantially revised initial drafts of the paper and provided final review and approval. LY performed statistical analyses. YH assessed IHC results. LY wrote the first draft of the manuscript which was revised with contribution from YH, GB, DM, MM, BR and RT. LY supervised the overall study progress. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was performed in accordance with the Declaration of Helsinki. The study protocol was approved by the institutional review boards of the Brigham and Women’s Hospital and Harvard T.H. Chan School of Public Health, and those of participating registries as required, and University of Florida Institutional Review Boards. Consent was obtained or implied by the return of questionnaires.
Consent for publication
Not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yaghjyan, L., Heng, Y.J., Baker, G.M. et al. Associations of stem cell markers CD44, CD24 and ALDH1A1 with mammographic breast density in women with benign breast biopsies. Br J Cancer (2024). https://doi.org/10.1038/s41416-024-02743-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41416-024-02743-2
- Springer Nature Limited